Download PDF
The typical patient with idiopathic intracranial hypertension (IIH), also known as pseudotumor cerebri, is diagnosed relatively easily and accurately, based on the Modified Dandy Criteria. When it comes to treatment, however, things get more complicated.1
A variety of therapies are used in clinical practice, including weight reduction, carbonic anhydrase inhibitors (acetazolamide), diuretics (furosemide), cerebrospinal fluid shunting, optic nerve sheath fenestration, and, most recently, venous sinus stenting. Unfortunately, indications for each intervention are poorly defined, so treatment of IIH has been all over the map.2
The desperate need for evidence-based guidelines for medical and surgical management of IIH3 spurred the Neuro-Ophthalmology Research Disease Investigator Consortium (NORDIC) IIH Study Group to launch the Idiopathic Intracranial Hypertension Treatment Trial (IIHTT)—the first prospective, multicenter, double-blind, placebo-controlled study to evaluate treatment strategies and possible etiologies of IIH in patients with mild visual loss.2,4
The first results are in.
|
High Grade
|
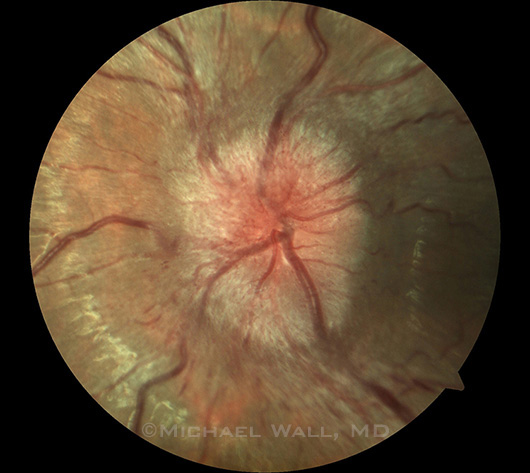 |
The effect of acetazolamide was greater in patients with a baseline papilledema grade of 3 to 5 than in those with lower-grade disease.
|
Finally, a Proven Therapy
The IIHTT, which compared the effects of acetazolamide plus a low-sodium, weight-reduction diet against placebo plus the diet, showed that acetazolamide combined with weight loss not only improved vision but also lowered cerebrospinal fluid pressure, reduced papilledema grade, and improved quality of life significantly more than weight loss alone.4 “All of the primary and secondary outcome measures were statistically significant, indicating that this drug is effective for IIH,” said lead author Michael Wall, MD, at the University of Iowa.
The results were not a given, the experts said. “Going into the trial, the evidence for acetazolamide’s effect on intracranial pressure was actually quite weak,” said Prem S. Subramanian, MD, PhD, at Johns Hopkins and a principal investigator. And coauthor Deborah I. Friedman, MD, MPH, at University of Texas Southwestern Medical Center, said, “When we were designing the study, I was not convinced that acetazolamide would show superiority to placebo. I truly had equipoise.”
Dr. Wall went in thinking that acetazolamide might prove effective, but he didn’t know if it worked by promoting weight loss or by an independent effect. “Our patients on acetazolamide plus diet lost twice as much weight as those on placebo plus diet,” said Dr. Wall. “We did a mediation analysis to see if the effect of acetazolamide was due to the weight loss. What we found was that the effects of acetazolamide and weight loss were independent, so it looks like we have two independent treatments.”
The trial showed that the benefit of acetazolamide occurs primarily during the first four to six weeks of treatment, while diet has a more gradual onset. The effect of the drug was faster than many experts had expected. “We also hadn’t realized that acetazolamide is so effective for higher grades of papilledema,” said Dr. Wall. In fact, the drug was shown to be much more effective in improving vision for higher grades than lower grades. “The biggest impact of this study is that it cements acetazolamide as a standard first-line treatment for IIH,” he said.
|
Web Extra
|
 |
Medical Management
Weight loss alone. The experts typically recommend diet alone for new patients who are asymptomatic or for patients with prior IIH who have stable vision and apparently quiescent disease with minimal or no papilledema. Studies of weight loss in obese patients with IIH have demonstrated a reduction in both papilledema and cerebrospinal fluid pressure.5,6 “To be effective, weight loss only has to be in the five to 10 percent range, and it can be a long-term solution to IIH,” said Dr. Subramanian.
Drug therapy. Dr. Friedman initiates medications to lower intracranial pressure when there is 1) visual field loss on automated perimetry, 2) transient visual obscurations, 3) binocular diplopia, or 4) pulsatile tinnitus. She also recommends headache treatment, generally topiramate, if there is no medical contraindication and headache is a significant problem.
The regimen used in the IIHTT began therapy with acetazolamide 500 mg twice a day, increasing the dosage as needed/tolerated, up to a total of 4 g daily. “I did not previously use dosages as high as 4 g daily but will do so now that there is evidence,” said Dr. Friedman. Furosemide (with potassium supplementation) is occasionally used as an adjunct to acetazolamide when single-drug therapy has not alleviated visual symptoms, but there is no proof yet that it works, said Dr. Wall.
Frequency of monitoring. For patients who present with mild visual loss and low-grade papilledema, Dr. Wall starts them on acetazolamide and sees them back in about six weeks. “I vary that frequency depending on the grade of papilledema and the degree of visual loss,” he said. “A patient with high-grade papilledema, for example, I’d see back within one to three weeks.” The IIHTT has reinforced these time points as a reasonable follow-up to look for an effect or monitor for progressive visual loss in the case of high-grade papilledema.
Once the papilledema has resolved, Dr. Wall starts tapering patients off acetazolamide to see how they do. Dr. Subramanian waits for patients to lose weight and keeps them on acetazolamide for a few more months to make sure the weight loss is durable before tapering off the acetazolamide. “At least 20 percent of patients need to go back on acetazolamide,” said Dr. Subramanian. “It’s related to their weight going back up.”
“After resolution, I caution patients to come back in if any symptoms reappear,” said Dr. Wall. Otherwise, he sees them every one to two years. “I follow people lifelong, especially because we see the disease recur with weight gain.”
|
Web Extra: Watch for IIH in Children
|
 |
Threshold for Surgery
“It’s the acuteness of disease onset and the severity of vision loss that guide initial management of IIH,” said Dr. Subramanian. “It’s almost a binary pathway. You have a group of patients who need surgery and need it now, and then a group of patients you have more time to work with to get the disease under control.”
Without guidelines, the threshold for surgery differs among doctors. In general, progressive visual loss that’s not responding to medical therapy, especially in patients with high-grade papilledema, indicates a need for surgery, said Dr. Wall. Dr. Friedman also cited central acuity loss at presentation (20/60 to 20/80 or worse) or marked visual field loss at presentation.
“If a patient presents with grade 5 papilledema with nerve fiber layer infarcts and a hemorrhagic optic nerve, and her medical history paints a picture of her disc swelling coming on very acutely, then my threshold for surgery is lower than for people who don’t fit that profile,” said Dr. Subramanian. “Here in Baltimore, the patients that worry me most are African-American teenagers, who for unclear reasons are more likely to have this fulminant type of disease.”
Type of Surgery
Fenestration vs. shunting. The two most common surgeries for IIH in the United States are optic nerve sheath fenestration (ONSF) and cerebrospinal fluid shunting. Opinions vary greatly regarding the best approach, with the decision often based on local preferences and the availability of specific surgeons.7
Dr. Wall has no preference, given that retrospective research has found no difference in outcomes between fenestration and shunting.7 Dr. Friedman prefers ONSF. “It is a less stressful operation for the patient, and there is no hardware in the body to fail in the future,” she said. Dr. Subramanian favors ONSF for visual loss and shunts for headaches.
“Ultimately, both surgeries have a high failure rate because you’re trying to create a fistula that the body doesn’t want, so it closes it off,” explained Dr. Subramanian. “I think of surgery as a temporizing measure, not a permanent cure, whether you do fenestration or shunting. It’s a means to protect vision and reduce symptoms while patients lose weight or address whatever the underlying problem is.”
|
Web Extra
|
 |
___________________________
1 Volpe NJ. JAMA Neurol. 2014;71(6):678-680.
2 Friedman DI et al; for the NORDIC IIHTT Study Group. J Neuroophthalmol. 2014;34(2):107-117.
3 Lueck CJ, McIlwaine GG. Cochrane Database Syst Rev. 2005;(3):CD003434.
4 Wall M et al. JAMA. 2014;311(16):1641-1651.
5 Newborg B. Arch Intern Med. 1974;133(5):802-807.
6 Sinclair AJ et al. Br Med J. 2010;340:c2701.
7 Fonseca PL et al. Br J Ophthalmol. 2014;98(10):1360-1363.
___________________________
Deborah I. Friedman, MD, MPH, is professor of neurology and neurotherapeutics and ophthalmology, University of Texas Southwestern Medical School, Dallas. Disclosure: None. Prem S. Subramanian, MD, PhD, is director of the Thyroid Eye Disease Center; associate professor of ophthalmology, neurology, neurosurgery, Johns Hopkins School of Medicine, Wilmer Eye Institute; and associate professor of surgery, division of ophthalmology, Uniformed Services University of the Health Sciences, Bethesda, Md. Disclosure: None. Michael Wall, MD, is professor of neurology and professor of ophthalmology and visual sciences, University of Iowa, Iowa City, and Iowa City Veterans Affairs Health Care System. Disclosure: None.