By James Reidy, MD, FACS, with Kathryn Hatch, MD, and Erin Stahl, MD
Download PDF
Read Part 1 of this roundtable discussion at “MD Roundtable, Part 1: Corneal Cross-Linking for Pediatric Keratoconus.”
Corneal collagen cross-linking (CXL) techniques can be distinguished by the amount of corneal epithelium that is removed. The standard Dresden protocol1 entails an “epi-off” treatment, whereas transepithelial, or “epi-on,” procedures minimize removal of the epithelium. In the final segment of this two-part series, James Reidy, MD, FACS, at the University of Chicago, continues a roundtable discussion with Kathryn Hatch, MD, of Massachusetts Eye and Ear Waltham and Harvard Medical School, and Erin Stahl, MD, of Children’s Mercy Hospital at the University of Missouri, Kansas City School of Medicine in Kansas City. Focusing on CXL in the pediatric age group, the experts share their thoughts on epi-on and epi-off techniques, indicators of surgical success and failure, and the future of treatment for children with keratoconus.
 |
|
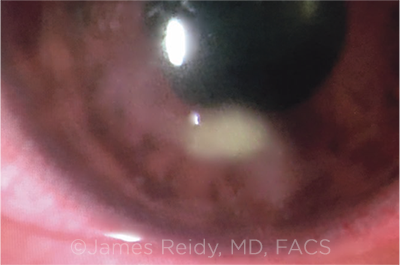
INFILTRATES. A minority of pediatric patients who undergo epi-off CXL may develop sterile corneal infiltrates. If this complication develops, it typically occurs within one week postoperatively.
|
Stromal Demarcation Line
Dr. Reidy: How much importance do you give to the stromal demarcation line—that is, the boundary between the cross-linked stroma of the anterior cornea and the untreated posterior cornea?
Dr. Stahl: I perform epi-off CXL by the Dresden protocol in a pediatric practice. My patients require general anesthesia for this surgery and sometimes are uncooperative with our testing. I do look at the cornea in every case, but I don’t always get to study it for long. When I can visualize the demarcation line in my patients, I notice that it slowly fades over time.
Dr. Hatch: I don’t routinely see a stromal demarcation line in every patient. I sometimes see either this demarcation line or a reticular haze pattern that subsequently fades. I don’t, however, put a lot of emphasis on the presence or absence of these findings, as a deep line does not necessarily predict better outcomes.2
Epi-off Versus Epi-on
Dr. Reidy: When a patient undergoes epi-off CXL, he or she typically will have blurry vision and a hazy cornea for approximately a month after surgery. Do epi-on techniques avoid these effects?
Dr. Hatch: The epi-off procedure results in hypertrophy of the epithelium in the first postoperative month. During this time, the keratometry becomes steeper, visual acuity often decreases, and corneal haze is present. Patients also avoid using contact lenses while the cornea heals. Epi-on procedures avoid many of these drawbacks to a greater extent.
The hybrid or transepithelial procedure that I perform is off-label, but it allows patients to return to contact lens use and to see well in the early postoperative period. Some patients even experience better vision early on.
Progression and Failure
Dr. Reidy: In a prospective study, Godefrooij and colleagues3 evaluated 36 pediatric patients with keratoconus (54 eyes) who received up to five years of follow-up. They found disease progression in 22% of patients after epi-off CXL. In an earlier retrospective study, Chatzis and Hafezi4 found an even higher rate of post-CXL progression: 88% in 42 patients (59 eyes) with a mean follow-up of approximately two years.
How frequently do you observe postoperative disease progression in your patients, and what constitutes failure of the procedure?
Dr. Hatch: There are multiple ways to assess progression, such as change in Kmax, worsening topography and thinning, as well as a change in corrective prescription and loss of best-corrected visual acuity. I think it’s crucial that we agree on how we define progression.
I assess progression, in part, by monitoring maximum keratometry readings (Kmax), but I do not use this measurement in isolation. I have seen post-op changes in Kmax that are concerning (such as an increase of more than 1.0 D5), but if the patient has stable vision or unchanged refractive properties, I wouldn’t necessarily consider the disease to be progressing. Occasionally, I obtain slightly different results from repeat topography testing that same day. In such cases, I ask the patient to return (for instance, in six to eight weeks) for serial topographic analysis. Sometimes the culprit is dry eye or how the patient wears contacts and there is resultant warpage on the scans rather than true disease progression. A patient’s Kmax results certainly can indicate progression. When this result is substantiated by other visual and topographic results, I would regard this as progression and re-treat accordingly.
I’ve been using the Photrexa system (Avedro) since 2016, and my retreatment rate has been, in general, less than 5% after two years. I also was part of the CXLUSA Study Group from 2011 to 2016, and most of our data corresponded to epi-on patients with a 0% rate of progression6 in the first two years. Among my patients, the eyes necessitating retreatments were eyes with advanced keratoconus at presentation.
Dr. Stahl: I’ve performed the Dresden CXL procedure on 36 eyes since January 2017. A few of my patients have shown fluctuation on follow-up tests, but only one patient has had definitive progression. He experienced hydrops after cross-linking, possibly because of eye rubbing.
Follow-up Routine
Dr. Reidy: How often do you follow up with patients during the first year?
Dr. Hatch: I carefully observe patients, usually every one to two days, until re-epithelialization occurs. If I notice stromal corneal haze in the first few weeks, I follow up at six weeks to confirm that the haze has cleared. From there, I monitor patients routinely at three to four months, at six to nine months, and at one year after surgery. I like to see visual function stabilized and stable scan results longitudinally.
I don’t examine the patient for topographic changes until the three- to four-month visit. I tell the patient, “If you wear contact lenses, please don’t wear them for the week before your appointment,” particularly if the patient’s initial screening had been done without contact lenses in. We want to ensure that the contact lens doesn’t cause warpage and affect our scan findings.
Dr. Stahl: We fit our patients’ soft contacts and scleral contacts in the office, which gives me the opportunity to monitor patients frequently during their first year of visual rehabilitation. We perform scans as part of their contact lens fitting, and we end up with a lot of data on these children.
Sterile Infiltrates
Dr. Reidy: Do you find that sterile epithelial infiltrates are a common occurrence after cross-linking in the pediatric population?
Dr. Stahl: Sterile infiltrates have developed in three of the 36 eyes I have treated. In these cases, I have adjusted the patient’s steroid regimen accordingly. I think this rate of infiltrates is consistent with other findings.
Dr. Reidy: I agree. Your rate of 8.3% (3/36 eyes) is similar to the infiltrate rate of 7.6% found by Koller and colleagues7 in their study of 99 patients (117 eyes) with primary keratectasia who underwent CXL.
Dr. Stahl: Children seem to have a more vigorous healing response than adults after CXL. One patient even had a significant central healing response in which the cornea flattened, thinned, and left a scar. Fortunately, this patient’s results have been improving, but I wouldn’t be surprised if sterile infiltrates occur a bit more frequently in children than in adults.
Dr. Reidy: In my experience, when a child develops an infiltrate, it occurs within the first week, typically by three or four days post-op. Sometimes these patients don’t present with an infiltrate until the epithelium has already healed.
Dr. Stahl: Yes, the sterile infiltrates I’ve seen usually develop in healed epithelium on post-op day 2. All but one of the children I’ve treated have had complete healing of the epithelium on post-op day 3, and I also remove the bandage contact lens that day.
Dr. Reidy: I believe that sterile infiltrates are the biggest problem because they often result in corneal scarring and subsequent thinning. In epi-on CXL, sterile infiltrates do not occur, correct?
Dr. Hatch: That’s correct.
Future of CXL in Children
Dr. Reidy: How do you think our techniques of CXL will change in the next few years?
Dr. Hatch: I would like to see pediatric keratoconus treatment evolve toward earlier screening and earlier detection. It would be very helpful if we had a way to inexpensively screen children—in schools, for instance, as part of a vision screening. The goal would be to perform transepithelial cross-linking very early—even before the disease becomes clinical—to prevent progressive ectasia.
Additionally, practitioners outside of the United States are performing modified CXL protocols, including accelerated cross-linking with more intense ultraviolet A (UVA) light for a shorter amount of time. I hope those techniques will eventually be incorporated into the array of keratoconus treatments in the United States.
Dr. Stahl: I agree that we need earlier corneal screening in children. By the time they present, these patients usually have one eye with moderate keratoconus and one with advanced disease.
I also would like the future of keratoconus treatment to avoid general anesthesia. I’d like to see a trend toward reduced operating time and accelerated cross-linking techniques that aren’t nearly as painful and avoid the risk of infiltrates and small scars.
Dr. Reidy: We have yet to find the optimal combination of riboflavin formulations and maneuvers that works best and is safest for treating our patients. Epi-on procedures, with some improvements, may be the general preference in the future. Stulting and colleagues6 have described brushing the cornea with a sterile sponge containing proparacaine to increase permeability of the intact epithelium. Other investigators have used pulsed, rather than continuous, UVA light, which decreases the amount of oxygen that’s consumed. It is possible that the concentration and formulation of riboflavin applied during CXL could be optimized to achieve increased penetration through intact corneal epithelium.
Pediatric patients are probably the most difficult to treat, but they’re also the ones who may benefit the most from CXL. Early screening is crucial, and it doesn’t necessarily have to be high tech. I find that examining the cornea with retroillumination, using a retinoscope or a direct ophthalmoscope, enables visualization of the cone—often before the patient is symptomatic.
___________________________
1 Wollensak G et al. Am J Ophthalmol. 2003;135(5):620-627.
2 Gatzioufas Z et al. J Cataract Refract Surg. 2016;42(5):804.
3 Godefrooij DA et al. Cornea. 2016;35(7):954-958.
4 Chatzis N, Hafezi F. J Refract Surg. 2012;28(11):753-758.
5 Evangelista CB, Hatch KM. Semin Ophthalmol. 2018;33(1):29-35.
6 Stulting RD et al. J Cataract Refract Surg. 2018;44(11):1363-1370.
7 Koller T et al. J Cataract Refract Surg. 2009;35(8):1358-1362.
___________________________
Dr. Hatch is the director of refractive surgery service at Massachusetts Eye and Ear, site director of Mass Eye and Ear Waltham, and assistant professor of ophthalmology at Harvard Medical School in Boston. Relevant financial disclosures: Avedro: C.
Dr. Reidy is associate professor of ophthalmology and visual science and vice chair for clinical operations at the University of Chicago. Relevant financial disclosures: None.
Dr. Stahl is associate professor of ophthalmology at the University of Missouri, Kansas City School of Medicine. Relevant financial disclosures: Avedro: C.
For full disclosures and the disclosure key, see below.
Full Financial Disclosures
Dr. Hatch Avedro: C; Carl Zeiss: C; EyePoint: C; Eyevance: C; Johnson & Johnson: C; Shire: C.
Dr. Reidy None.
Dr. Stahl Avedro: C; Retrophin: C,S; Treehouse Health: C,O.
Disclosure Category
|
Code
|
Description
|
| Consultant/Advisor |
C |
Consultant fee, paid advisory boards, or fees for attending a meeting. |
| Employee |
E |
Employed by a commercial company. |
| Speakers bureau |
L |
Lecture fees or honoraria, travel fees or reimbursements when speaking at the invitation of a commercial company. |
| Equity owner |
O |
Equity ownership/stock options in publicly or privately traded firms, excluding mutual funds. |
| Patents/Royalty |
P |
Patents and/or royalties for intellectual property. |
| Grant support |
S |
Grant support or other financial support to the investigator from all sources, including research support from government agencies (e.g., NIH), foundations, device manufacturers, and/or pharmaceutical companies. |
|