By Rebecca Taylor, interviewing Zaina N. Al-Mohtaseb, MD, Bennie H. Jeng, MD, Thomas M. Lietman, MD, and Jennifer Rose-Nussbaumer, MD
Download PDF
Fungal keratitis is notoriously challenging to diagnose and treat, and aggressive cases can perforate through the cornea and spread inside of the eye, making early diagnosis critical. “The keys to being able to successfully manage these cases are first having the suspicion of a fungal infection, then being able to confirm the diagnosis quickly,” said Bennie H. Jeng, MD, at the University of Maryland School of Medicine in Baltimore.
“Current management is still based on figuring out that it’s fungus as quickly as possible and using traditional antifungal treatment, which includes topical natamycin,” said Thomas M. Lietman, MD, at the University of California, San Francisco (UCSF). But topical natamycin penetrates poorly into the corneal stroma, making deep ulcers hard to treat,1 and a new “wonder drug” for fungal keratitis remains elusive, Dr. Leitman said. In the Mycotic Ulcer Treatment Trials (MUTT) I and II, even the newest drug, voriconazole, failed to show significant benefit, either in outperforming topical natamycin (in MUTT I) or as an adjuvant oral therapy to antifungal topicals (in MUTT II), he said.
Fortunately, there are some bright lights on the horizon: Metagenomic deep sequencing (MDS) and repeat cultures raise the promise of an earlier diagnosis. And corneal cross-linking (CXL) may improve treatment outcomes.
 |
|
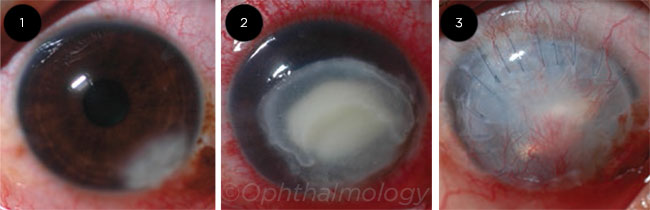
PROGRESSION. This patient’s ulcer was culture-positive for Fusarium. At enrollment in MUTT I (Fig. 1), his VA was 20/25; after three weeks of topical voriconazole, his VA had declined to hand motions (Fig. 2). He experienced perforation and required a TPK, and at three months (Fig. 3), his VA was light perception.
|
Devastating Infections
“These devastating fungal infections are very difficult to treat,” said Jennifer Rose-Nussbaumer, MD, also at UCSF. “In one of our studies, we gave patients everything we had: eyedrops, oral medications, and surgery—and about 50% still needed therapeutic penetrating keratoplasty [TPK].”
Diagnostic challenge. A key issue with fungal keratitis is the time lag to an accurate diagnosis. “We always jump to thinking about the best way to treat fungal keratitis,” Dr. Lietman said, “but if patients are not coming in for two or three weeks and the damage is already done, then early diagnosis is where we’re going to make a big leap forward.”
With a fungal infection of the cornea, the corneal epithelium is often intact, so the infection is harder to catch with traditional cultures. And, as fungus can occur in a wound and penetrate deeper into the cornea, it’s less amenable to the scraping techniques used for bacteria. Moreover, the rate of culture-positive scraping is low, even in academic settings.
“I am at a tertiary center, and we sometimes only see the infection once it has resulted in a corneal melt,” said Zaina N. Al-Mohtaseb, MD, at Baylor College of Medicine in Houston. “Many referral physicians don’t have the capabilities we have at academic institutions [e.g., a good microbiology lab and confocal microscopy]. But even with these, the sensitivity and specificity of these tests for fungal keratitis is low.”
Treatment attempts. Furthermore, attempted treatment can backfire. “Given the difficulty with diagnosis, some of the patients sent to our practice are on multiple therapies, such as antibacterials, antivirals, and steroids,” said Dr. Al-Mohtaseb. “Unfortunately, these can worsen the infection—especially the topical steroids.” As a result, she said that she has had to re-culture some patients. “Others have needed a corneal biopsy for diagnosis, and, at times—although this is rare—the diagnosis occurs after a TPK.”
Dr. Jeng also pointed out that “different fungi may respond differently to the various antifungal medications. Personally, my go-to antifungal for Candida is amphotericin B, which needs to be extemporaneously compounded.”
Dr. Mohtaseb added, “Cases that result in corneal perforation or fail to progress despite maximal appropriate antifungal therapy might require TPK to prevent scleral involvement and endophthalmitis and to preserve the globe.” With regard to therapeutic grafts, she said, “the risk of recurrence is as high as 50%2—so, again, early diagnosis and treatment is of utmost importance.”
Improving Diagnosis
Metagenomic deep seqencing. MDS is at the cutting edge of new diagnostic technology for fungal corneal ulcers. With a targeted test like polymerase chain reaction (PCR), clinicians must know what they’re looking for. In contrast, MDS is considered unbiased, as it will identify any organism in the clinical sample.
“With next-generation sequencing, the gain is in quicker, more accurate diagnosis because we don’t have to wait for the fungus to grow,” said Dr. Lietman. “MDS can tell us whether it’s a bacteria, a fungus, a parasite like Acanthamoeba, or another organism. This should allow for very rapid diagnosis.”
Clinical benefit. “Sequencing technology is being used more and more to help clinicians make diagnoses, and in the setting of fungal infections, this technology has huge potential in making a positive clinical impact,” said Dr. Jeng.
Scientific benefit. “The technology is also scientifically interesting for us because there are new organisms we didn’t know that cause keratitis,” Dr. Lietman said. As a result, he said, “next-generation sequencing is good for pathogen discovery.”
Not FDA approved. However, it should be noted that MDS is neither approved nor in mainstream use. Once it is, the hope is that any ophthalmologist will be able to take a swab, preserve it in nucleic acid–stabilizing media, and ship it off to a lab. The sample will be sequenced, and the treating clinician will get the results faster than the cultures could grow.
Repeat cultures. Another new diagnostic—and prognostic—tool is the use of repeat cultures after initiating treatment.
“A really interesting subfinding of MUTT I and II came from repeat cultures at six days,” said Dr. Rose-Nussbaumer. “If patients were positive on that repeat culture, they had a much higher risk of going on to need surgery, having worse three-month visual acuity, and having a larger scar size. It correlated with every single negative outcome.”
Identifying TPK need. Repeat cultures identified patients who would ultimately need TPK, said Dr. Rose-Nussbaumer. “In MUTT II, we found that the vast majority of patients who met certain criteria, such as large ulcers that were deep and culture positive, needed TPK. Repeat cultures are a very useful tool for prognosis and helping identify patient populations, such as those who might benefit from a TPK early on when the chances of eliminating the infection are higher.”
Adapting treatment. When six-day repeat cultures are positive, clinicians can increase the drug dosage, add a different topical antifungal, try an oral antifungal, and more closely monitor patients who are at higher risk of corneal perforation or may need TPK to excise the infection.3
Use of repeat cultures also helps determine which antimicrobials are working.4 “If we only use healing time or vision as outcomes, we don’t have much hope of distinguishing different antimicrobial agents,” Dr. Lietman said. In contrast, he said, “repeat cultures are an excellent way to distinguish new antimicrobials, because we’re directly testing how well we killed the fungus. And the fact that six-day culture correlates with vision gives more credibility to using repeat cultures in future clinical trials.”
New Use of CXL?
Although CXL has been well studied for ectasia, “it hasn’t been very well studied as a useful adjuvant for people with infections and corneal ulcers,” said Dr. Rose-Nussbaumer.
After the MUTT II trial, Dr. Rose-Nussbaumer said, she went back to look at patients’ charts, and many needed multiple surgeries. “The fungal infection came back, or they had a retinal detachment; it looked like almost all those eyes were lost. That’s why we started looking at cross-linking as a potential therapy for fungal keratitis: We’ve run out of medical therapies, and intrastromal injection of antifungals doesn’t really work.”
CLAIR trial. Dr. Rose-Nussbaumer’s current trial, Cross-Linking–Assisted Infection Reduction (CLAIR), is now in its follow-up phase. “We randomized patients to medical therapy alone versus medical therapy and adjuvant CXL,” she said. “We enrolled 111 patients with moderate fungal keratitis, and our primary outcome measure will be culture positivity after CXL.” The researchers will also look at clinical measures such as visual acuity, scar size, the need for TPK, and other complications.
CXL caveats. There are still unknowns in using CXL to treat corneal infections. It’s possible, for instance, that CXL “has an antiseptic quality that treats the infection but also causes a lot of abnormalities in the shape of the cornea that could negatively affect vision,” Dr. Rose-Nussbaumer hypothesized. “For patients with keratoconus, for instance, we counsel that they may lose a line of best-corrected vision after CXL, because some opacity could form in the cornea that might be a result of cross-linking.”
Evolving Epidemiology
In the United States, fungal keratitis is most often seen in warm, humid regions. “Some of the most devastating infections that I saw in Miami and now see in Texas are due to fungal keratitis,” Dr. Al-Mohtaseb said.
There is some speculation that climate change may be driving an increase in fungal ulcers, as they tend to occur in patients who live in tropical areas. “In these areas, filamentous fungi such as Fusarium predominate,” said Dr. Jeng.
However, he added, “Even in temperate areas of the world, fungal keratitis is still seen, and it needs to be suspected in certain cases.” In temperate climates, he noted, “yeasts such as Candida are most frequently seen. In either case, successful treatment of fungal keratitis still depends on accurate and timely diagnosis.”
What about bacterial keratitis? “Bacterial ulcers seem to be becoming less common because people can quickly access antibiotic drops anywhere in the world,” Dr. Lietman said. Given the prevalence of antibiotics, by the time a patient comes to the doctor’s office, the bacteria may already be dead. “That may be why, from the ophthalmologists’ perspective, the ulcers they see—particularly in tertiary settings—are often not bacterial; they’re now often fungal or Acanthamoeba infections.”
___________________________
1 Austin A et al. Ophthalmology. 2017;124(11):1678-1689.
2 Sharma N et al. Curr Opinion Ophthalmol. 2010;21(4):293-300.
3 Ray KJ et al, for the Mycotic Ulcer Treatment Trial Group. Am J Ophthalmol. 2017;178:157-162.
4 Ray KJ et al, for the Mycotic Ulcer Treatment Trial Group. Am J Ophthalmol. 2018;189:41-46.
___________________________
Dr. Al-Mohtaseb is assistant professor of ophthalmology and associate director of the Residency Program at Baylor College of Medicine in Houston. Relevant financial disclosures: None.
Dr. Jeng is professor of ophthalmology and chair of the Department of Ophthalmology & Visual Sciences at the University of Maryland School of Medicine in Baltimore. Relevant financial disclosures: None.
Dr. Lietman is director of the Francis I. Proctor Foundation for Research in Ophthalmology and professor of ophthalmology, epidemiology, and biostatistics at UCSF. Relevant financial disclosures: None.
Dr. Rose-Nussbaumer is a cornea and external disease specialist and assistant professor of ophthalmology at UCSF. Relevant financial disclosures: NEI: S.
For full disclosures and the disclosure key, see below.
Full Financial Disclosures
Dr. Al-Mohtaseb Alcon: C; Allergan: C; Bausch + Lomb: C.
Dr. Jeng Aquinox: C; CoDa Therapeutics: C; EyeGate Pharmaceuticals: O; Jade Therapeutics: C; Kedrion: C; Santen: C; TissueTech: C.
Dr. Leitman None.
Dr. Rose-Nussbaumer NEI: S.
Disclosure Category
|
Code
|
Description
|
| Consultant/Advisor |
C |
Consultant fee, paid advisory boards, or fees for attending a meeting. |
| Employee |
E |
Employed by a commercial company. |
| Speakers bureau |
L |
Lecture fees or honoraria, travel fees or reimbursements when speaking at the invitation of a commercial company. |
| Equity owner |
O |
Equity ownership/stock options in publicly or privately traded firms, excluding mutual funds. |
| Patents/Royalty |
P |
Patents and/or royalties for intellectual property. |
| Grant support |
S |
Grant support or other financial support to the investigator from all sources, including research support from government agencies (e.g., NIH), foundations, device manufacturers, and/or pharmaceutical companies. |
|