By Annie Stuart, Contributing Writer, interviewing Guillermo Amescua, MD, Ali Djalilian, MD, and Bennie H. Jeng, MD
Download PDF
A novel treatment for unilateral limbal stem cell deficiency (LSCD), simple limbal epithelial transplant (SLET) involves transplanting a small number of limbal stem cells from the contralateral eye.
The procedure has its origins in India. After using ex vivo cultivated limbal epithelial cells for more than 1,000 unilateral transplants, Virender S. Sangwan, MBBS, and colleagues at the L.V. Prasad Eye Institute in Hyderabad began to explore the idea of in vivo expansion, said Guillermo Amescua, MD, from the University of Miami.
“I traveled to India in 2011 to learn about ex vivo expansion but found the lab nearly empty,” said Dr. Amescua. Instead, he shadowed surgeons performing SLET and observed postop results in the clinic. After seeing favorable results in India, he decided to pursue SLET at home. Dr. Amescua is a pioneer of this procedure in the United States, which is now beginning to capture the attention of other surgeons.
 |
|
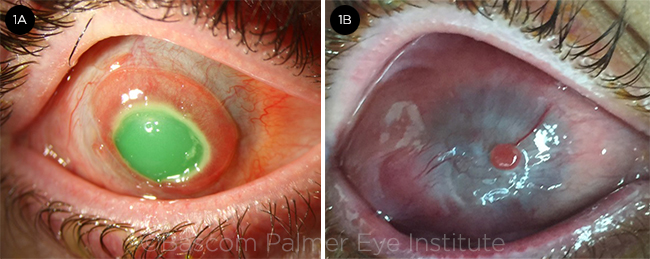
PREOP. (1A) This patient with an alkali injury to the right eye has a central and paracentral epithelial defect with early conjunctivalization of the peripheral cornea. Vision is hand motions. (1B) Patient 6 months after the injury.
|
Patient Selection
“SLET works really well for patients with unilateral chemical or thermal injuries and iatrogenic LSCD from multiple surgeries,” said Dr. Amescua. It could also be used for unilateral LSCD caused by toxicity from medications or preservatives, said Bennie H. Jeng, MD, from the University of Maryland, Baltimore.
Not always ideal. “SLET is not a solution for all unilateral stem cell deficiencies,” said Dr. Amescua. “I’ve tried it in patients with immunological diagnoses and it doesn’t work as well.”
Contact lens wearers may also develop unilateral LSCD, said Dr. Jeng, but most wear contact lenses in both eyes. “And even if one eye looks okay,” he said, “it can have subclinical problems. Therefore, we are generally unwilling to remove stem cells from the seemingly good eye because this could result in stem cell deficiency.”
In addition, patients with significant stromal scarring or thinning are less favorable candidates because they will most likely require a future corneal transplant, said Ali Djalilian, MD, from the University of Illinois, Chicago. “This may result in removal of transplanted limbal tissues within the central 8 mm area.”
Dr. Djalilian reported that he has had trouble finding the right candidates. “I have my first patient ready to go,” he said, “but I more often see patients with bilateral disease.”
 |
|
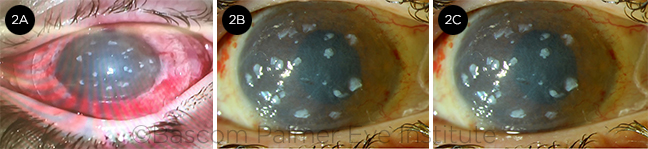
POSTOP. (2A) Immediate postop photo. Notice the small limbal explants placed under the bandage contact lens. The cornea is clear after removal of the conjunctival pannus covering the whole cornea. (2B) Postop week 2. The bandage contact lens has been removed. Amniotic membrane graft and limbal explants are still present. (2C) Postop month 3. The amniotic membrane is almost completely resorbed, and the limbal explant is slowly decreasing in size. Uncorrected vision is 20/60.
|
How SLET Stacks Up
How does SLET compare with other procedures used for unilateral LSCD?
CLAU—hard on donor eye. Conjunctival limbal autograft (CLAU) involves removing a relatively large piece of conjunctiva and limbus and transplanting these into the injured eye. “With CLAU, it was generally thought you needed to transfer a total of about 4 clock hours of limbal stem cells for the surgery to be successful,” said Dr. Jeng. Although CLAU is a great procedure, removing so much tissue can prompt LSCD in the donor eye, said Dr. Amescua, and it may not be necessary to do so—as evidenced by CLET and SLET.
CLET—not FDA approved. Cultivated limbal epithelial transplantation (CLET) involves ex vivo expansion of limbal stem cells. “The beauty of CLET is you take only a 1- to 2-mm punch of limbus, which minimizes trauma to the donor eye, and you expand it to a large number of limbal stem cells,” said Dr. Amescua. However, the FDA has not approved CLET, which requires a clean-room facility to grow the stem cells, and this is very costly.
SLET—best of both worlds. “The novelty of the SLET procedure is you take a very small number of stem cells, just as with CLET; ‘sprinkle’ them on a piece of amniotic membrane that covers the surface of the cornea; and allow it to re-epithelialize,” said Dr. Jeng. This doesn’t require supplying all the cells the eye needs, added Dr. Djalilian, “just enough to improve the health of the epithelium, which changes the environment so that healthy stem cells that survived the injury can start to recover.”
This produces sustained improvement in the surface phenotype of the cells, said Dr. Djalilian. “Results are comparable to CLET, but SLET is less expensive.”
“The beauty of SLET is that it can be done anywhere in the world as a same-day surgery,” said Dr. Amescua. “You don’t need fancy instruments or ex vivo expansion in a lab.” One of the most impressive results Dr. Amescua has noted in his patients is the resolution of symptoms such as pain and redness. “Even though 3 of my patients went from 20/400 to 20/40,” he said, “they thanked me more effusively for a renewed level of comfort in the eye.”
Evidence for SLET. Compared with CLAU, which has 25-plus years of follow-up study to show safety and efficacy, SLET has much shorter follow-up. Currently, a multicenter trial comparing CLET and SLET is under way; meanwhile, a recent paper published in Ophthalmology by Basu et al. has reported clinical outcomes with SLET in 125 cases of unilateral chronic ocular surface burns.1 At a median postop follow-up of 1.5 years, 76% of patients maintained a positive outcome. More than 75% saw a 2-line improvement in visual acuity, with 67% of successful cases attaining vision of 20/60 or better.
“Importantly, nothing yet has been published about SLET inducing problems in the donor eye,” said Dr. Amescua, who added that his group is studying SLET in rabbits to help determine why it performs better than CLET in pediatric populations—as was recently reported in the British Journal of Ophthalmology.2
Tips for Best Results
Dr. Amescua recommends trying at least 10 to 15 cases before deciding whether or not SLET works well for you and your patients. “It’s important to remember that we’re dealing with difficult pathologies and that CLET is also not 100% successful. Given that SLET has a 20% failure rate overall, you can consider 9 successful cases out of 12 a good result.”
Enhance the environment. If the patient has suffered a chemical injury, said Dr. Djalilian, wait as long as you can for the inflammation to subside. “You want to put healthy cells in the best possible environment,” he said. “If you put them in a harsh environment—whether it’s inflamed or dry, or the lids are turning in so lashes constantly touch the eye—you are not setting those cells up for long-term survival.”
Take care during surgery. When doing the surgery, said Dr. Amescua, try to minimize cautery to decrease postoperative inflammation. And, he added, “After the procedure, look through the microscope while you carefully remove the drapes to avoid removing the contact lens and cells.”
Stage it. In patients with LSCD and some degree of corneal scarring, said Dr. Amescua, it’s better to perform the SLET first, and not jump right into a corneal transplant. “I have done combined SLET [with] penetrating keratoplasty or lamellar keratoplasty for logistical reasons, but it is always better to do it as a staged procedure.”
|
Customizing SLET
Mini-SLET. Dr. Amescua, in collaboration with the cornea group from Conde de la Valenciana in Mexico City, helped pioneer “mini-SLET.” This is a novel procedure used after pterygium removal and capitalizes on the properties of the AM and limbal epithelial stem cells.1
Primary SLET. Publishing favorable results in the journal Cornea, another group in India has used primary SLET to treat mitomycin-C–induced LSCD in a patient with extensive ocular surface squamous neoplasia, said Dr. Amescua.2
Other variations. Researchers are trying a variety of techniques, said Dr. Amescua, such as putting the stem cells directly on top of the cornea without the amniotic membrane, then protecting them with the contact lens. It’s too soon to know how well this technique will work.
___________________________
1 Hernandez-Bogantes E et al. Br J Ophthalmol. 2015;99(12):1598-1600.
2 Mittal V et al. Cornea. Published online July 20, 2016.
|
How to Perform SLET
Before the surgery. Make sure to select patients carefully, said Dr. Jeng. Confirm that the donor eye is truly normal and that the recipient eye has a wet ocular surface and good eyelid closure. “Otherwise, the procedure is sure to fail,” he said.
A wet ocular surface is essential, Dr. Amescua said. “Just as with a garden, nothing will grow without moisture and nutrients.”
Harvest limbal stem cells. Dr. Amescua uses a retrobulbar block on the diseased eye and topical anesthesia on the donor eye. “I take 2 to 3 mm of the limbus from the donor eye,” he said, “and give it to the nurse who puts it in balanced salt solution to ensure it stays moist.” It’s important to be careful handling the stem cells, which are very fragile, said Dr. Jeng.
(To ensure that all is well with the recipient eye, Dr. Djalilian prefers to prepare it first before harvesting cells from the donor eye.)
Prepare recipient eye. It’s very important to do a very good superficial keratectomy, said Dr. Amescua. Consider using preop high-resolution corneal optical coherence tomography (OCT) to plan the surgery well, or use live OCT so you can see how deep the incision is. “If you go too deep, you may end up causing a small perforation, and a patch graft or corneal transplant may be necessary. In most cases, the stroma of the recipient eye is clear, so we just need to find the plane of scar tissue, peel it off like a sticker, and send a sample to pathology.”
After a 360-degree conjunctival peritomy, secure a piece of amniotic membrane (AM)—stromal side down—with fibrin glue, said Dr. Djalilian. “The AM should be large enough to cover the entire cornea. Tuck it in under the host conjunctiva, and secure it with glue or sutures or a combination.”
Transplant limbal cells. After cutting the donor tissue into 12 to 15 pieces, spread them on the amniotic membrane, sparing the visual axis, said Dr. Amescua.
The original protocol, said Dr. Djalilian, was to put the tissue in a paracentral area. “My preference would be to put the tissues a little closer to the limbus,” he said. This helps create a larger transplant-tissue–free zone that won’t be excised if a keratoplasty is required in the future.
After the glue is dry, said Dr. Amescua, place a 16- to 20-mm bandage contact lens over the transplanted cells. “Make sure that transplant tissues and AM are well secured and adherent and that the contact lens sits well and is secure,” said Dr. Djalilian. “If you have any concern about the lens falling out, do a temporary tarsorrhaphy to keep it in place.”
Postop care. “We see the patient the day after surgery,” said Dr. Amescua. “Normally, we keep the contact lens in place for about a week to 10 days, during which time the patient is also on topical steroids and topical antibiotics. Patients are on steroids for 6 to 8 weeks and tapered off slowly. You should start seeing healing of the corneal epithelium within the first 1 to 3 weeks.”
Potential complications. Although not common, complications can occur. “I’ve had patients who took a while to recover vision because the amniotic membrane was thicker than average and never melted,” said Dr. Amescua. “In one such patient, I performed a phototherapeutic keratectomy—polishing the cornea with the excimer laser—and the patient epithelialized very well.”
Another of his patients with a unilateral chemical injury had significant postop inflammation. Clinically, it behaved like herpetic corneal disease. Although the patient had no history of herpetic disease, the inflammation responded very well to topical steroids and oral acyclovir.
Recurrence of progressive conjunctivalization may also occur, said Dr. Amescua, but often a second procedure will succeed after an initial failure. Hemorrhage under the graft is a more common problem, he said. “You want to ensure that there is no active bleeding when you place the amniotic membrane, because if it doesn’t attach perfectly, the procedure doesn’t work well.”
Dr. Jeng has performed SLET twice, but in both cases, the stem cells sloughed off. He implanted a keratoprosthesis in one patient, and he opted for observation in the other. “I’ve not had good success, but I will certainly try it again,” he said, adding that in the future he will take steps to protect the stem cells on the surface—possibly with a temporary tarsorrhaphy.
___________________________
1 Basu S et al. Ophthalmology. 2016;123(5):1000-1010.
2 Mittal V et at. Br J Ophthalmol. 2016;100(8):1102-1108.
___________________________
Dr. Amescua is assistant professor of clinical ophthalmology at the University of Miami in Miami, Fla. Relevant financial disclosures: None.
Dr. Djalilian is associate professor of ophthalmology and director of the corneal stem cell and tissue engineering laboratory at the University of Illinois in Chicago. Relevant financial disclosures: None.
Dr. Jeng is professor and chair of the department of ophthalmology and visual sciences at the University of Maryland School of Medicine in Baltimore. Relevant financial disclosures: None.
For full disclosures and the disclosure key, see below.
