By Michael Mott, Contributing Writer, interviewing Ahmad A. Aref, MD, MBA; Aleksandra V. Rachitskaya, MD; and Elizabeth Yeu, MD
Download PDF
Elevated intraocular pressure (IOP) is not uncommon after ocular surgeries—from phacoemulsification to keratoplasty to vitrectomy. For most eyes, moderate spikes are often transient and don’t pose a significant threat to long-term visual health. However, extremely high IOP or IOP elevation in patients with certain preexisting conditions, such as optic nerve damage, can significantly threaten vision following an otherwise successful surgery.
To complicate matters, ophthalmologists aren’t always able to predict which patients will experience a spike. That’s why it’s valuable to know how to both minimize the risk of IOP elevation and address high pressure after it occurs.
Cataract: Vexing Viscoelastic
Elevated IOP is the most frequent complication that requires treatment following phacoemulsification, said Ahmad A. Aref, MD, MBA, at the University of Illinois in Chicago. And its most common cause is retained viscoelastic material, which inhibits aqueous outflow through the trabecular meshwork. The surgeon should therefore be aggressive in removing as much viscoelastic as possible before completing the procedure, he said.
Dr. Aref also relies on two pharmacological interventions to preemptively manage the possibility of post-op IOP spikes in certain cases.
Acetazolamide. Cataract surgeons who administer oral acetazolamide for IOP control commonly do so right after completing surgery, said Dr. Aref. Recent research, however, shows that acetazolamide’s effects are superior when given to the patient prior to surgery.1
“These results changed my practice patterns,” said Dr. Aref. “Instead of using the acetazolamide as a post-op treatment in higher-risk eyes, I’m now administering one hour before and seeing fewer IOP events for one to 24 hours after surgery.”
Despite that success, Dr. Aref doesn’t use acetazolamide in every case. He typically uses it only for patients with advanced preexisting glaucoma or those who have a known outflow obstruction. This is because, in addition to a host of potential systemic side effects, the medication can, in rare instances, induce acute bilateral angle-closure glaucoma with choroidal detachment, he said.
Acetylcholine. Dr. Aref saves his second pharmacological intervention for surgeries that require manipulation of the iris or other complex anterior segment procedures. Injected during the cataract procedure, intracameral acetylcholine is a miotic that pulls the iris away from the trabecular meshwork, improving outflow and helping to decrease IOP. And it’s safe to use in combination with acetazolamide. However, acetylcholine also comes with some risk that may limit routine use. In rare cases, for example, acetylcholine injections have resulted in aqueous misdirection, which can cause a postoperative angle closure event and uncontrolled IOP, said Dr. Aref.
Burping the wound. If a pressure spike is evident on the first postoperative day and the patient is deemed to be at risk for secondary complications, Dr. Aref moves on to “burping” the wound. “With a sterile cotton tip applicator, I apply direct pressure next to the corneal paracentesis site to release the remnant viscoelastic and aqueous from inside the eye,” he said. “This decompression is immediate and can be performed repeatedly on an as-needed basis.”
In some cases, a short course of topical IOP-lowering agents may also be indicated in order to keep the pressure controlled over the first postoperative week, he said.
Cornea: Creating and Taming Glaucoma
Keratoplasty procedures can induce elevated IOP—which in turn can accelerate graft failure and endothelial cell loss, said Elizabeth Yeu, MD, at Virginia Eye Consultants in Norfolk, Virginia. In addition, management of corneal disease typically requires a long course of anti-inflammatory steroids, which can cause elevated IOP, she said. “As a result, cornea surgeons create a lot of secondary glaucoma.”
Pre-op. When candidates for lamellar transplantation present with very poorly controlled IOP, Dr. Yeu refers them to a glaucoma specialist to stabilize that pressure.
Immediately post-op. Cornea surgery can cause IOP spikes even in relatively healthy eyes. What should you watch for during the immediate post-op period?
Viscoelastic. The typical culprit in IOP rise after a full-thickness PK is the leftover viscoelastic that’s used to maintain adequate separation between the transplanted cornea and the iris and lens, said Dr. Yeu. “As in cataract surgery, this material increases the risk of blocking the trabecular meshwork.” An IOP spike can be treated shortly after surgery, by burping out some of the viscoelastic between two sutures at the graft-host junction, she said.
Bubble-induced IOP elevation. For lamellar endothelial keratoplasty, increased post-op pressure is often due to the introduction of gas in the anterior chamber that helps hold the graft tightly to the posterior stroma of the host. Although a large gas bubble minimizes the risk of graft detachment, overfilling the chamber can prevent aqueous from entering the anterior chamber, resulting in a pupillary block.
To prevent pupillary block, cornea surgeons have traditionally used a peripheral iridotomy (PI) inferiorly, around the 6-o’clock position, said Dr. Yeu. But she cautioned that the PI can be rendered useless if the bubble occludes the iridotomy opening. “I’ll use more than one PI to give the patient two shots at the goal,” said Dr. Yeu. “That way, when they do sit upright following the keratoplasty, the gas elevates and the aqueous can travel through multiple sites.” In addition, she dilates the patient at the end of the surgery with a few drops of atropine 1%.
She advised that patients be sent home with instructions to avoid activity and to lie face up for much of the first 24 to 48 hours to help the graft adhere. However, if a patient experiences acute signs of pupillary block—including worsening headache, nausea, or vomiting—they should sit or stand upright to make sure the intraocular bubble can rise above the PI site and/or the inferior pupillary margin. For severe, unremitting symptoms, they should be seen emergently, she said.
Late post-op. In most cases, after the one-week post-op period, a significant volume of the gas will have dissipated. Any remaining IOP elevation is likely a response to the prescribed steroids, said Dr. Yeu.
To address this issue, she includes topical medications, including beta blockers and adrenergic agonists. Dr. Yeu avoids topical and oral carbonic anhydrase inhibitors because they may stun the active sodium pumps within the endothelium (which can reduce graft adhesion). She also avoids prostaglandin analogs because of their proinflammatory qualities.
If postkeratoplasty glaucoma does not respond adequately to medication, selective laser trabeculoplasty or more traditional glaucoma treatment might be necessary, she said. But, she added, the risk of corneal graft failure increases significantly when any hardware, like a tube shunt, is placed in the anterior chamber.
For patients who require glaucoma surgery up to four weeks after keratoplasty, Dr. Yeu always asks that subsequent eye care teams perform a pachymetry at every visit to identify any corneal thickness changes that might indicate early graft rejection. “I also want the patient back in my chair afterward,” she said, “because, at this point, it’s still a very vulnerable cornea and any surgery can beat up the ocular surface.”
 |
|
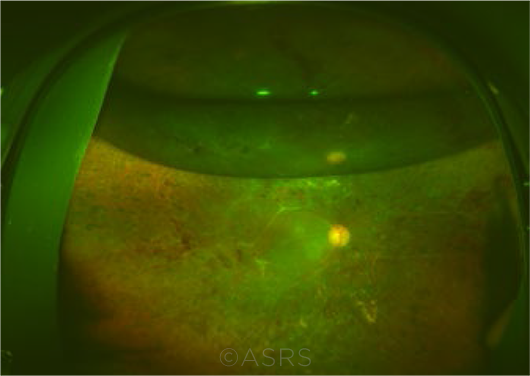
TAMPONADE. Post-op eye after 25-gauge vitrectomy and C3F8 gas to repair traction retinal detachment due to proliferative diabetic retinopathy. This image was originally published in the ASRS Retina Image Bank. Yusuke Oshima, MD, PhD. Retinal Attachment After Vitrectomy. Retina Image Bank. 2013; Image Number 8646. ©The American Society of Retina Specialists.
|
Retina: Buckles, Gas, and Oil
Acute increases in postoperative IOP can also occur during or after some vitreoretinal procedures. In most cases, the spikes can be managed with oral or topical pressure-lowering medications. In more complex patients, however, additional surgery might be required.
Scleral buckling. “Initial placement of a scleral buckle can cause increased IOP intraoperatively,” said Aleksandra V. Rachitskaya, MD, at the Cleveland Clinic in Ohio. She said that the retina surgeon should always check for optic nerve pulsation when using a buckle. After tightening the band, visible pulsation indicates that IOP is on the higher side. The absence of pulsation can signify that either perfusion is normal or the pressure is elevated. “To distinguish between the two scenarios, we usually take a cotton tip applicator and press on the eye,” said Dr. Rachitskaya. “If the additional pressure results in pulsations, we know that the IOP was normal.” Anterior chamber tap might be used to decrease the pressure if indicated, she said.
Even after successful repair of a retinal detachment, scleral buckling surgery can impair venous drainage from the vortex veins. “A high scleral buckle, for example, can lead to congestion and swelling of the ciliary body,” said Dr. Rachitskaya. “This forward rotation of the ciliary body can also shift the lens-iris diaphragm forward, resulting in a shallow anterior chamber or even angle closure.”
Typically, mildly elevated IOP following scleral buckling resolves within weeks, said Dr. Rachitskaya. If not, additional medical treatment includes cycloplegics, corticosteroids, and oral or topical pressure-lowering medications. She cautions that performing traditional glaucoma surgery on these eyes can be difficult due to the excessive scarring caused by the original buckle surgery.
Vitrectomy. IOP rise associated with vitrectomy largely stems from the use of sulfur hexafluoride (SF6) and perfluoropropane (C3F8) gases as well as silicone oil. Although these tamponade agents are necessary for successful retinal reattachment, there are several caveats, said Dr. Rachitskaya.
SF6 and C3F8 gas. Unlike silicone oil, gas at 100% concentration is highly expansive. “Determining the appropriate gas concentration is extremely important,” said Dr. Rachitskaya. “I make it a habit of verbally confirming with my team the gas that is being used before mixing it myself. My go-to is 20% SF6 and 14% C3F8.”
Most IOP elevation following vitrectomy surgery is benign and the result of gas overfill. This can be remedied by topical pressure-lowering drops or oral carbonic anhydrase inhibitors as well as aspiration of a small amount of gas via a pars plana tap, if needed, she said. However, she added, in more serious cases of uncontrolled IOP stemming from incorrect gas concentration, there should be no delay in surgically replacing the gas tamponade, as prolonged elevation can cause permanent perfusion issues and optic nerve damage.
Patient warning. Since intraocular gas can expand with changes in atmospheric pressure, patients should abstain from air travel or exposure to significant changes in altitude until the bubble resorbs. Patients should also avoid postoperative exposure to nitrous oxide gas until resorption. The gas is highly soluble and quickly diffuses into the intraocular bubble, which can dangerously expand and lead to permanent vision loss, said Dr. Rachitskaya. As a result, it’s common practice to place a bracelet on patients’ arms indicating that they recently received vitrectomy surgery in case they need to undergo any subsequent surgery with general anesthesia.
Silicone oil. An oil tamponade doesn’t expand like its gas counter-part, but oil overfill is still possible, said Dr. Rachitskaya. When issues do arise, they’re typically seen in aphakic patients. “Without a lens to keep back the silicone oil, pupillary block can occur,” said Dr. Rachitskaya. “To prevent this, I perform a prophylactic peripheral iridotomy in the 6-o’clock position intraoperatively. Because the oil floats, blockage of an iridotomy can occur if you choose a more superior position.”
In more challenging retinal surgeries, including in patients with refractory glaucoma or with complete synechial angle closure, glaucoma surgery may be necessary—which raises an important point, said Dr. Rachitskaya. “Post-op IOP events often occur in complex patients requiring multiple treatments that span subspecialties. So regardless of expertise, all ophthalmologists should be aware of how common ocular surgeries can induce elevated pressure.”
__________________________
1 Hayashi K et al. Ophthalmology. 2017;124(5):701-708.
__________________________
Dr. Aref is an associate professor of ophthalmology, vice chair for clinical affairs, and director of the glaucoma fellowship training program at the University of Illinois College of Medicine, Chicago. Relevant financial disclosures: None.
Dr. Rachitskaya is an assistant professor of ophthalmology at the Cleveland Clinic, Cole Eye Institute. Relevant financial disclosures: None.
Dr. Yeu is a cornea and cataract surgeon at Virginia Eye Consultants in Norfolk. Relevant financial disclosures: None.
For full disclosures and the disclosure key, see below.
Full Financial Disclosures
Dr. Aref Aerie: L.
Dr. Rachitskaya Alcon: C; Allergan: C; Genentech: C; Novartis: C; Regeneron: C; Samsara: C; Zeiss: C.
Dr. Yeu Alcon: C,L; Allergan: C,L; ArcScan: C; Bausch + Lomb: C; Bio-Tissue: C,L; Carl Zeiss Meditec: C,L; Glaukos: C,L; Innovation Labs: O; iOptics: C,S; Johnson & Johnson Vision: C,L; Kala: C,S; Katena: C; Melt: C,O; Modernizing Medicine: O; Novartis, Alcon: C; Ocular Science: C,O; OcuSoft: C; Omeros: L; ScienceBased Health: C; Sight Sciences: C; Sun Ophthalmics: L; Surface: C; Tarsus: C,O; TearLab: C; TearScience: C; TissueTech: C,L; Topcon: S.
Disclosure Category
|
Code
|
Description
|
| Consultant/Advisor |
C |
Consultant fee, paid advisory boards, or fees for attending a meeting. |
| Employee |
E |
Employed by a commercial company. |
| Speakers bureau |
L |
Lecture fees or honoraria, travel fees or reimbursements when speaking at the invitation of a commercial company. |
| Equity owner |
O |
Equity ownership/stock options in publicly or privately traded firms, excluding mutual funds. |
| Patents/Royalty |
P |
Patents and/or royalties for intellectual property. |
| Grant support |
S |
Grant support or other financial support to the investigator from all sources, including research support from government agencies (e.g., NIH), foundations, device manufacturers, and/or pharmaceutical companies. |
|