Download PDF
Using a murine model of early-onset Fuchs corneal dystrophy, researchers found that a single instance of CRISPR/Cas9 gene editing can protect endothelial cells indefinitely from the degeneration that would otherwise cloud the cornea.1
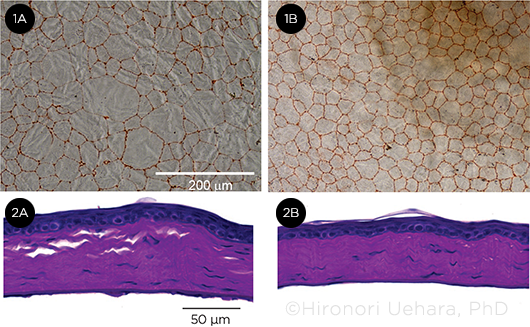 |
ENDOTHELIAL RESCUE. Comparison of the (1A) nontreated and (1B) treated endothelial cells in mice. Representative corneal endothelium in each group stained with alizarin red. The intraocular injection of an adenovirus encoding both the Cas9 gene and guide RNA also reduced guttae-like structures on the corneal endothelium, as shown in the periodic acid-Schiff—stained corneas of (2A) nontreated and (2B) treated mice. Photo courtesy of Hironori Uehara, PhD. Adapted from: Uehara H et al. Start cordon disruption with CRISPR/Cas9 prevents murine Fuchs endothelial corneal dystrophy. eLife. 2021;10:e55637. This is an open access article under the CC Attribution 4.0 International license (http://creativecommons. org/licenses/by/4.0/).
|
Novel approach. The scientists accomplished this feat by taking a different approach to ocular gene therapy, said study coauthor Balamurali K. Ambati, MD, PhD, at the University of Oregon in Eugene.
Their target: COL8A2. Normally, this collagen gene is not essential to healthy endothelial cell function—but in its mutated form, the gene leads to the cellular dysfunction and death found in Fuchs, Dr. Ambati noted.
At that point, because the endothelial cells are postmitotic, editing the mutated gene sequence with CRISPR is not an option, he said. Instead of trying to edit out the single point mutation, Dr. Ambati said, “What we did with CRISPR was to use a technique called insertion/deletion to disrupt the start codon of the mutant protein. And by knocking down the synthesis of this mutant protein, we were able to ameliorate all the different adverse effects of it, restoring endothelial cell density and function.”
Study in mice. Dr. Ambati and his colleagues worked with gene knockout mice that develop a disease which closely resembles early-onset Fuchs.
The mice were given a single anterior chamber injection of a recombinant adenovirus, which carried an RNA snippet designed to inactivate the faulty gene’s “start” codon. This prevented production of the mutant protein, and the therapeutic effect lasted at least 10 months after injection, the researchers said.
“Once you disrupt the gene in these postmitotic cells, you’ve fixed the problem, because there will be no new cells, since endothelial cells do not reproduce,” Dr. Ambati said.
With regard to safety, because adenoviruses can induce inflammation and cell toxicity, the researchers tested a range of titers of the adenoviral vector. Corneal transparency, corneal thickness, and histopathology were normal at low titers, they reported. They also confirmed that the vector did not suppress retinal function or damage the retinal structure, nor did it induce liver or kidney damage or inflammation.
Looking ahead. If this CRISPR-based therapy works in humans, it would reduce or perhaps eliminate the need for corneal transplants in patients diagnosed with Fuchs, Dr. Ambati said. Start codon disruption also might be applicable to other adult-onset diseases caused by missense mutations, he added.
In the next year, the researchers plan to begin testing the therapeutic adenovirus in donor cadaver corneas with and without guttae (a hallmark of Fuchs). If those studies show that the technique works and is not toxic to human cells, tests in larger animals will follow, Dr. Ambati said.
—Linda Roach
___________________________
1 Uehara H et al. eLife. 2021;10:e55637.
___________________________
Relevant financial disclosures: Dr. Ambati—NEI: S; Research to Prevent Blindness: S.
For full disclosures and the disclosure key, see below.
Full Financial Disclosures
Dr. Ambati iVeena: O; NEI: S; Research to Prevent Blindness: S.
Dr. Bursztyn None.
Dr. Hunter Lippincott Williams & Wilkins: P; Luminopia: C,O; Rebion: C,O,P; Slack: P.
Dr. Shen American Glaucoma Society: S; Topcon: S.
Disclosure Category
|
Code
|
Description
|
| Consultant/Advisor |
C |
Consultant fee, paid advisory boards, or fees for attending a meeting. |
| Employee |
E |
Employed by a commercial company. |
| Speakers bureau |
L |
Lecture fees or honoraria, travel fees or reimbursements when speaking at the invitation of a commercial company. |
| Equity owner |
O |
Equity ownership/stock options in publicly or privately traded firms, excluding mutual funds. |
| Patents/Royalty |
P |
Patents and/or royalties for intellectual property. |
| Grant support |
S |
Grant support or other financial support to the investigator from all sources, including research support from government agencies (e.g., NIH), foundations, device manufacturers, and/or pharmaceutical companies. |
|
More from this month’s News in Review