Download PDF
Hydroxychloroquine (HCQ; Plaquenil) is widely used for the treatment of rheumatoid arthritis and other connective tissue diseases. Excessive dosages of HCQ, however, can result in HCQ retinopathy, a potentially blinding disease. In an effort to minimize the risk and simplify the estimations of HCQ dosages in the clinic, a team of ophthalmologists created a free smartphone app for calculating optimal weekly dosages.1 However, as initially described,1 the app deviated from current screening recommendations,2 thus leading to a revision.
How it works. The original DoseChecker app combined 2 approaches to HCQ dosing. The developers used both ideal and actual body weight as methods for determining the maximum dose. After a physician entered the patient’s height and weight, the app selected the method that recommends the lower dose—under the assumption that the lowest dose is the safest dose to avoid any toxic effects. After making the calculation, the app then suggested a dosing schedule that divided the total weekly doses into a combination of 400- and 200-mg daily doses of hydroxychloroquine.
The developers advised that physicians will need to take other risk factors into consideration when using the app, including systemic disease, concomitant retinal disease, and tamoxifen usage.1
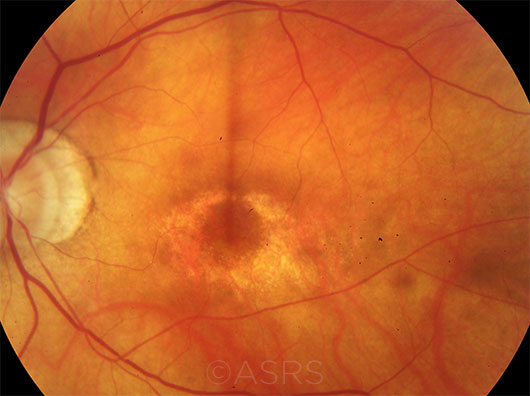 |
TOXICITY. A case of HCQ retinopathy in a 68-year-old woman with a 15-year history of HCQ use.This image was originally published in the ASRS Retina Image Bank. Joshua O. Mall, MD. Plaquenil (Hydroxycholoroquine) Toxicity Fundus Photo. Retina Image Bank. 2016; Image Number 26378. © The American Society of Retina Specialists.
|
Potential problems. Such an app would be very helpful, said Michael F. Marmor, MD, of Stanford University in Palo Alto, California, and the lead author of the Academy’s guidelines. However, the original DoseChecker’s use of both actual and ideal body weight contradicts the Academy’s guidelines2 for calculating optimal daily dosage. “On the basis of a recent study of 2,361 long-term HCQ users,3 the Academy now recommends that all patients using HCQ keep daily dosage less than 5.0 mg/kg actual body weight—not ideal body weight,” said Dr. Marmor. “Older recommendations once advised calculating dosage as 6.5 mg/kg ideal body weight, but that conclusion was based on 50-year-old studies about HCQ and fat-using animals. We really should follow the most current human data.”
Ideal body weight formulas tend to overdose slight individuals, especially women, Dr. Marmor added, whereas real weight predicts risk accurately and evenly across all body types.3 As initially constructed, “the DoseChecker app selects actual body weight for thin individuals but switches to ideal body weight for heavier individuals, as it calculates a lower dose. However, ‘lowest is safest’ is only true for drugs that are equally effective at both doses, and there is no evidence to date that low HCQ doses are still therapeutic for heavy patients, or why physicians should give one group of patients a different dose than another,” he said.
But there is good news, Dr. Marmor said. “As we speak, the developers are changing the app to use the Academy’s dosing recommendations. When this revised app is available, it can be heartily recommended to simplify the calculation of daily dose and of the schedule of tablets needed to provide a proper weekly dose.” He concluded, “This device has promise to aid rheumatologists as well as ophthalmologists in providing the latest and safest guidelines for prescribing HCQ.”
—Mike Mott
___________________________
1 Perlman EM et al. JAMA Ophthalmol. 2018;136(2):218-219.
2 Marmor MF et al. Ophthalmology. 2016;123(6):1386-1394.
3 Melles RB, Marmor MF. JAMA Ophthalmol. 2014;132(12):1453-1460.
___________________________
Relevant financial disclosures: Dr. Marmor—None.
For full disclosures and the disclosure key, see below.
Full Financial Disclosures
Dr. Bachmann Oculocare Medical: E,O.
Dr. Davis AbbVie: C; Allergan: C.
Dr. Doan NEI: S; Research to Prevent Blindness: S; Silicon Valley Community/Huang Pacific Foundation: S; UCSF Resource Allocation Program: S.
Dr. Gonzales NEI: S.
Dr. Marmor None.
Disclosure Category
|
Code
|
Description
|
| Consultant/Advisor |
C |
Consultant fee, paid advisory boards, or fees for attending a meeting. |
| Employee |
E |
Employed by a commercial company. |
| Speakers bureau |
L |
Lecture fees or honoraria, travel fees or reimbursements when speaking at the invitation of a commercial company. |
| Equity owner |
O |
Equity ownership/stock options in publicly or privately traded firms, excluding mutual funds. |
| Patents/Royalty |
P |
Patents and/or royalties for intellectual property. |
| Grant support |
S |
Grant support or other financial support to the investigator from all sources, including research support from government agencies (e.g., NIH), foundations, device manufacturers, and/or pharmaceutical companies. |
|
More from this month’s News in Review