Download PDF
In a systematic review, the Complications of Retinal Detachment Surgery (CORDS) Study Group evaluated procedures for the repair of rhegmatogenous retinal detachment (RRD). They found that “the reporting of harms was inadequate and required improvement,” said Noemi Lois, MD, PhD, at Queens University in Belfast, Northern Ireland. Even when the frequency of complications was recorded, the severity was rarely noted, making it difficult to compare different interventions.1
Clinical trials “are often good at presenting efficacy of new treatments tested, but they are not as good at reporting complications (harms) in a systematic and quantifiable manner,” Dr. Lois said.
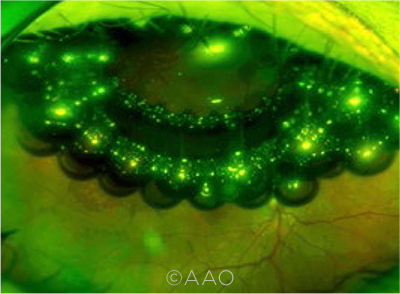 |
FISH EGGS. The formation of gas bubbles (fish-egg phenomenon) during pneumatic retinopexy is included in the new classification system.
|
Classifying complications. The CORDS Study Group first developed a comprehensive list of 87 complications associated with retinal repair procedures, including general intra- and postoperative surgical complications as well as those specific to scleral buckling, pars plana vitrectomy (PPV), and pneumatic retinopexy. Seventy surgeons from 17 countries were invited to participate in ranking these harms; of these, 43 completed the process.2
Participants were asked to assign a score, ranging from 1 (no harm to patient or vision) to 10 (worst possible harm to patient or vision, e.g., permanent loss of vision or painful eye) for each item on the list. The study group then applied the Delphi method to compile the anonymous responses, present the summary results of the first round to the participants, and allow them to either maintain or modify their own rankings for the next round.
Achieving international consensus. The group reached consensus on 84 (97%) of the complications.2 “It was very good indeed to see that this large group of surgeons from all continents graded complications in such a homogeneous manner and achieved consensus in only two rounds of the Delphi survey,” said Dr. Lois. She added, “In my mind, this supports the generalizability of the CORDS results.”
Dr. Lois attributed the lack of consensus on the three remaining complications to the “very strict criteria we set” of an interquartile range (IQR) of ≤2 on a 10-point scale, while “many consensus studies set the consensus criteria at an IQR of ≤3 on a 9-point scale.” The three outliers—suprachoroidal hemorrhage, not kissing and not involving the macula; subretinal infusion in the context of PPV; and early migration of the scleral buckle—each had an IQR of 2.75.
Looking ahead. To be useful, the classification must be easy for surgeons to consult, said Dr. Lois. “For this reason, we are working on an app that would be freely accessible through mobile phones and computers.” This would “greatly facilitate [the classification’s] introduction in clinical practice, not only for its use in clinical trials but also for auditing our surgical results.”
—Peggy Denny
___________________________
1 Xu ZY et al; CORDS Study Group. JAMA Ophthalmol. Published online June 17, 2021.
2 Xu ZY et al JAMA Ophthalmol. 2021;139(8):857-864.
___________________________
Relevant financial disclosures—Dr. Lois: None.
For full disclosures and the disclosure key, see below.
Full Financial Disclosures
Dr. Chen None.
Dr. Jain Foundation Fighting Blindness: S.
Dr. Lois None.
Dr. Sarraf Amgen: C; Bayer: C; Genentech: C,S; Heidelberg: S; Janssen: C; Novartis: L; Optovue: C,S; Regeneron: S; Topcon: S.
Dr. Shousha Heru: O; NEI: S; Resolve Ophthalmics: O,P.
Disclosure Category
|
Code
|
Description
|
| Consultant/Advisor |
C |
Consultant fee, paid advisory boards, or fees for attending a meeting. |
| Employee |
E |
Employed by a commercial company. |
| Speakers bureau |
L |
Lecture fees or honoraria, travel fees or reimbursements when speaking at the invitation of a commercial company. |
| Equity owner |
O |
Equity ownership/stock options in publicly or privately traded firms, excluding mutual funds. |
| Patents/Royalty |
P |
Patents and/or royalties for intellectual property. |
| Grant support |
S |
Grant support or other financial support to the investigator from all sources, including research support from government agencies (e.g., NIH), foundations, device manufacturers, and/or pharmaceutical companies. |
|
More from this month’s News in Review