NOV 08, 2015
By Elias I. Traboulsi, MD
A Compendium of Inherited Disorders and the Eye, Oxford University Press
Genetics
Also called Bloch-Sulzberger syndrome
OMIM Numbers
Inheritance
Gene/Gene Map
- The disease is caused by mutations in the NEMO (NF-Kappa-B Essential Modulator) gene and is referred to as IP2, or "classical" incontinentia pigmenti. Sporadic incontinentia pigmenti, originally called IP1, maps to Xp11 and is categorized as similar to hypomelanosis of Ito.
- An 870-bp region of identity corresponding to an MER67B repeat exists in the NEMO gene both in intron 3 and 3’to exon 10; recombinations between the regions of identity delete exons 4 though 10 of NEMO. Accounting for 80% of new mutations, this rearrangement would result in a truncated molecule carrying the 133 N-terminal amino acids of NEMO plus at least 26 novel amino acids. It was predicted that this molecule would contain part of the first coiled-coil domain and may still interact with IKK2, but would be unlikely to respond to upstream signals.
Epidemiology
Clinical Findings
- This genodermatosis is lethal prenatally in males, with rare exceptions.
- In affected females, it causes highly variable abnormalities of the skin, hair, nails, teeth, eyes, and central nervous system. The prominent skin signs occur in 4 classic cutaneous stages: perinatal inflammatory vesicles, verrucous patches, a distinctive pattern of hyperpigmentation, and dermal scarring. The erythematous eruption with linear vesiculation in the newborn period is followed by a verrucous stage. After a few months, the verrucous growth is replaced by hyperpigmented areas, which disappear at age 20 years.
- 20% of patients have skeletal abnormalities, including hemivertebrae, kyphoscoliosis, syndactyly, and leg length discrepancies.
- Hypodontia, delayed tooth eruption, and conical teeth are frequent.
- 15% of patients show central nervous system involvement, developing mental deficiency, microcephaly, spasticity, or seizures.
- The diagnosis is based on clinical features and family history.
- Gene testing is possible.
Ocular Findings
- 35% of patients have significant ocular abnormalities, which are generally unilateral or markedly asymmetric.
- 20% of patients have significant visual loss, mostly from retinal disease.
- The development of a retrolental mass of glial tissue in rare cases may lead to confusion with retinoblastoma or retinopathy of prematurity.
- Abnormal vascular anastomoses with peripheral zones of decreased perfusion and preretinal fibrosis are frequently present.
- Less common ocular findings include cataracts (4%, and only if there is a retinal detachment), uveitis (2%), and blue sclerae (2%). Other abnormalities include nystagmus, strabismus, microphthalmos, pigmentation of the conjunctiva, corneal scarring, absence of the anterior chamber, optic atrophy, persistence of the hyaloid artery, and myopia.
- Goldberg reported on the nature of the ocular complications of incontinentia pigmenti. He emphasized that the ocular and cerebral abnormalities associated with this disease are far worse than the name would indicate. Although some patients have normal vision, total blindness or permanent visual deficiency may occur. Retinal vascular abnormalities, involving the periphery as well as the macula, appear to represent the primary disease process in the eye. Retinal detachment may ensue, due to mechanisms that seem analogous to those of retinopathy of prematurity. Optic nerve atrophy and occipital lobe infarction are additional causes of severe visual dysfunction in some patients. He reported, for the first time, neonatal infarction of the macula in this disease.
- When the eye is involved, retinopathy of prematurity and retinoblastoma are frequent misdiagnoses.
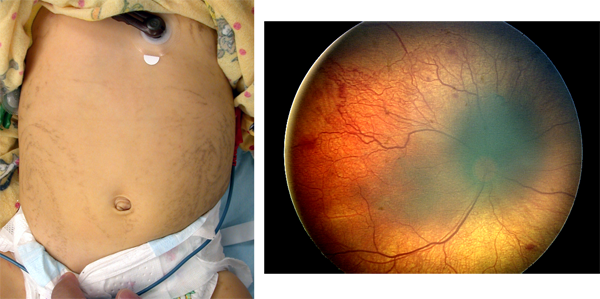
Figure 1. Whorly linear skin lesions (left) and optic atrophy (right), in a patient with incontinentia pigmenti.
Therapeutic Aspects
- There is no specific therapy for the skin disorder.
- Cryotherapy and laser photocoagulation have been used with some success in patients in whom the retinal disease was detected early.
References
- Goldberg MF: The blinding mechanisms of incontinentia pigmenti. Ophthalmic Genet. 1994; 15:69-76.
- International Incontinentia Pigmenti Consortium: Genomic rearrangement in NEMO impairs NF-kappa-B activation and is a cause of incontinentia pigmenti. Nature, 2000; 405:466-472.
- Minic S, Trpinac D, Obradovic,M. Incontinentia pigmenti diagnostic criteria update. Clin Genet. 2014; 85: 536-542.
- Phan TA, Wargon O, Turner AM. Incontinentia pigmenti case series: clinical spectrum of incontinentia pigmenti in 53 female patients and their relatives. Clin Exp Dermatol. 2005; 30:474-80.
- Swinney CC, Han DP, Karth PA. Incontinentia Pigmenti: A Comprehensive Review and Update. Ophthalmic Surg Lasers Imaging Retina. 2015; 46:650-657.
Traboulsi EI. Compendium of Inherited Disorders and the Eye. New York: Oxford University Press; 2005. Adapted with permission.