By M. Edward Wilson, MD
Introduction and epidemiology
In children, cataract causes more visual disability than any other form of treatable blindness. Children with untreated, visually significant cataracts face a lifetime of blindness at tremendous quality of life and socioeconomic costs to the child, the family, and the society. More than 200,000 children are blind from unoperated cataract, from complications of cataract surgery, or from ocular anomalies associated with cataracts.1 Many more children suffer from partial cataracts that may slowly progress over time, increasing the visual difficulties as the child grows. The cumulative risk of cataract during the growing years is as high as 1 per 1000.2
The management of cataracts in childhood is tedious and often difficult, requiring many visits over many years. Success requires a dedicated team effort that often involves parents, primary care pediatricians, surgeons, anesthesiologists, technicians, orthoptists, low vision rehabilitation specialists, and community health workers.
Classification (Categorization)
Cataracts in children can be classified using a number of methods including age of onset, etiology, and morphology.
Age of onset
Congenital/Infantile
While the presence of lens opacities at birth indicates a congenital onset, the diagnosis and recognition of a lens opacity at a later age does not exclude a congenital onset. It is critical to provide a detailed description of the type of lens opacities before the cataract is extracted and in the operative note so the type can be determined and any later study correlating genetic etiology or associated systemic disease can be done more accurately. Some morphological categories of cataracts such as anterior polar, central fetal nuclear, and posterior polar clearly indicate a congenital onset, while others such as cortical or lamellar may be associated either with a later onset or be congenital in nature.
Acquired/Juvenile
This category can be confusing. Strictly speaking, an acquired cataract is one from an external cause, as opposed to one in which the cause is genetically determined, such as a mutation in one of the crystalline genes. However, some would use acquired to indicate an onset after infancy, which does not necessarily indicate a non-genetic cause. Juvenile cataracts are by definition those with an onset in childhood, after infancy, irrespective of underlying etiology.
Etiology
Genetic
Approximately 50% of childhood cataracts are caused by mutations in genes that code for proteins involved in lens structure or clarity. Table 1 lists genes in which mutations can cause cataracts.3 While many of these genes are dominantly inherited, others are autosomal recessive or X-linked. Drawing a pedigree and recognizing some cataract and ocular phenotypes that are associated with specific mutations will help determine the probable mode of inheritance and the possible underlying syndrome. Recent advances in genetic testing, including next generation sequencing, allow the determination of the precise genetic cause of isolated congenital cataracts in 75% of individual families and 63% of those with syndromic congenital cataracts.4 Mutations in crystallins account for 50% of isolated (no associated systemic abnormalities) cataracts, while mutations in the gap junction protein connexins account for 25% of cases and mutations in genes for heat shock transcription factor-4, aquaporin-0, and beaded filament structural protein-2 account for the remaining 25%.
Metabolic disorders can cause cataracts, which may have particular morphologies that point to the underlying cause. Next generation sequencing of genes associated with syndromic or metabolic cataracts can provide a precise diagnosis if the systemic findings do not allow recognition of the metabolic or systemic illness. Table 1 summarizes findings in some of the main diseases associated with acquired syndromic cataracts.
Trauma remains a major cause of acquired cataracts in children. Traumatic cataracts are more common in boys and can be the result of penetrating or blunt injuries to the eye. One has to be careful in ruling in or out the presence of an intraocular or intraorbital foreign body, hence the importance of a detailed physical examination and of imaging studies such as ultrasonography and computed tomography. Magnetic resonance imaging (MRI) studies are contraindicated if the foreign body is suspected to be metallic.
|
Table 1. Common causes of congenital or early acquired cataracts3
|
|
Disease
|
Location
|
Gene
|
Phenotype
|
OMIM number
|
|
AUTOSOMAL DOMINANT
|
|
Hyperferritinemia-cataract syndrome
|
19q13.33
|
FTL
|
Congenital nuclear cataract and elevated serum ferritin
|
600886
|
|
Coppock-like cataracts
|
2q33.3
|
CRYGC
|
Dusty opacity of the fetal nucleus with frequent involvement of the zonular lens
|
604307
|
|
22q11.23
|
CRYBB2
|
|
Volkmann type congenital cataract
|
1p36
|
Unknown
|
Central and zonular cataract
|
115665
|
|
Zonular with sutural opacities
|
17q11.2
|
CRYBA1
|
Zonular cataracts with sutural opacities
|
600881
|
|
Posterior polar 1 (CTPP1)
|
1p36.13
|
EPHA2
|
Opacity located at back of lens
|
116600
|
|
Posterior polar 2 (CTPP2)
|
11q23.1
|
CRYAB
|
Single well-defined plaque in posterior pole of lens; bilateral
|
613763
|
|
Posterior polar 3 (CTPP3)
|
20q11.22
|
CHMP4B
|
Progressive, disc-shaped, posterior subcapsular opacity
|
605387
|
|
Posterior pole 4 (CTPP4)
|
10q24.32
|
PITX3
|
Single well-defined plaque in posterior pole of lens
|
610623
|
|
Posterior pole 5 (CTPP5)
|
14q22-q23
|
Unknown
|
Mat reflex of posterior capsule that progresses into well-demarcated disc in posterior pole, forming opaque plaque
|
610634
|
|
Zonular pulverulent 1 (CZP1)
|
1q21.2
|
GJA8
|
Lenticular opacities located in the fetal nucleus with scattered, fine, diffuse cortical opacities and incomplete cortical 'riders'
|
116200
|
|
Zonular pulverulent 3 (CZP3)
|
13q12.11
|
GJA3
|
Central pulverulent opacity surrounded by snowflake-like opacities in anterior and posterior cortical regions of the lens
|
601885
|
|
Anterior polar cataract 1
|
14q24-qter
|
Unknown
|
Small opacities on anterior surface of lens
|
115650
|
|
Anterior polar cataract 2
|
17q13
|
Unknown
|
Small opacities on anterior surface of lens
|
601202
|
|
Cerulean type 1 (CCA1)
|
17q24
|
Unknown
|
Peripheral blue and white opacities in concentric circles
|
115660
|
|
Cerulean type 2 (CCA2)
|
22q11.23
|
CRYBB2
|
Numerous peripheral blue flakes and occasional spoke-like central opacities
|
601547
|
|
Cerulean type 3 (CCA3)
|
2q33.3
|
CRYGD
|
Progressive blue dot opacities
|
608983
|
|
Crystalline aculeiform cataract
|
2q33.3
|
CRYGD
|
Needle-like crystals projecting in different directions, through or close to the axial region of the lens
|
115700
|
|
Nonnuclear polymorphic congenital cataract
|
2q33.3
|
CRYGD
|
Opacities between the fetal nucleus and the cortex of the lens
|
601286
|
|
Sutural cataract with punctate and cerulean opacities
|
22q11.23
|
CRYBB2
|
Dense, white opacification around the anterior and posterior Y sutures, oval punctate and cerulean opacities of various sizes arranged in lamellar form
|
607133
|
|
Myotonic dystrophy 1 (DM1)
|
19q13.32
|
DMPK
|
Myotonia, muscular dystrophy, cataracts, hypogonadism, frontal balding, and ECG changes
|
160900
|
|
Polymorphic and lamellar cataracts
|
12q13.3
|
MIP
|
Lamellar, sutural, polar and cortical opacities
|
604219
|
|
Cataract, autosomal dominant, multiple types 1
|
3q22.1
|
BFSP2
|
Nuclear and sutural opacities.
|
611597
|
|
AUTOSOMAL RECESSIVE
|
|
Congenital cataracts, facial dysmorphism, and neuropathy (CCFDN)
|
18q23
|
CTDP1
|
Congenital cataracts, facial dysmorphism, neuropathy, delayed psychomotor development, skeletal anomalies, microcornea and hypogonadism
|
604168
|
|
Marinesco-Sjögren syndrome
|
5q31.2
|
SIL1
|
Congenital cataracts, cerebellar ataxia, muscle weakness, delayed psychomotor development, short stature, hypergonadotrophic hypogonadism, and skeletal deformities
|
248800
|
|
Warburg micro syndrome 1
|
2q21.3
|
RAB3GAP1
|
Microcephaly, microphthalmia, microcornea, optic atrophy, cortical dysplasia, in particular corpus callosum hypoplasia, severe mental retardation, spastic diplegia, and hypogonadism
|
600118
|
|
Warburg micro syndrome 2
|
1q41
|
RAB3GAP2
|
614225
|
|
Warburg micro syndrome 3
|
10p12.1
|
RAB18
|
614222
|
|
Martsolf syndrome
|
1q41
|
RAB3GAP2
|
Mental retardation, hypogonadism, microcephaly
|
212720
|
|
Hallermann-Streiff syndrome (Francois dyscephalic syndrome)
|
6q22.31
|
GJA1
|
Brachycephaly, hypotrichosis, microphthalmia, beaked nose, skin atrophy, dental anomalies, short stature
|
234100
|
|
Rothmund-Thomson syndrome
|
8q24.3
|
RECQL4
|
Skin atrophy, telangiectasia, hyper- and hypopigmentation, congenital skeletal abnormalities, premature aging, increased risk of malignant disease
|
268400
|
|
Smith-Lemli-Opitz syndrome
|
11q13.4
|
DHCR7
|
Microcephaly, mental retardation, hypotonia, , polydactyly, cleft palate
|
270400
|
|
Congenital nuclear cataracts 2
|
22q11.23
|
CRYBB3
|
Nuclear cataract with cortical riders
|
609741
|
|
X-LINKED
|
|
Norrie disease
|
Xp11.3
|
NDP
|
Early childhood blindness, mental disorder, sensorineural deafness
|
310600
|
|
Nance Horan syndrome
|
Xp22.13
|
NHS
|
Males have dense nuclear cataracts, microcornea, dental abnormalities, and developmental delay. Carrier females have posterior Y-sutural cataracts with small corneas
|
302350
|
Xu LT, Traboulsi EI. Genetics of congenital cataracts. In: Wilson ME, Trivedi RH, editors. Pediatric Cataract Surgery: Lippincott, Walters Kluwer 2014. p. 1-8.
Secondary
Uveitis – Cataracts develop in patients with uveitis as a result of the chronic ocular inflammation or secondary to the chronic use of steroids. Surgery for such cataracts can be complicated by severe postoperative inflammation, hence the need for absence of preoperative inflammation in the anterior segment of the eye and the pre-, intra-, and post-operative use of various combinations of topical, subconjunctival, intracameral, and sometimes systemic steroids. Many patients will have a pupillary membrane that covers the lens and attaches to the iris, making surgery more difficult. Such membranes can be peeled off of the anterior lens capsule at the time of surgery to facilitate lens removal. The use of an intraocular lens (IOL) is left to the discretion of the individual surgeon.
- Juvenile idiopathic arthritis: One of the more common causes of anterior uveitis in children. The use of systemic antimetabolites in recent years has led to better control of uveitis in such patients and to a reduction in the incidence of cataracts.
- Other types of uveitis can also cause cataracts either because of the inflammation or as a complication of steroid use.
Intraocular tumors – It is very uncommon for cataracts to develop as a consequence of intraocular tumors. The lens is characteristically clear in patients with untreated retinoblastoma. Treatments of the tumor such as radiotherapy may lead to the development of cataracts, in which case timing of cataract removal has to be very carefully considered and surgery only performed when all tumor in the eye has been eradicated. Patients with radiation cataracts can have significant ocular surface dryness and will not tolerate contact lenses, hence the need for intraocular lens (IOL) implantation.
Chronic retinal detachment – These cataracts are seen in the setting of injuries or in association with Stickler syndrome. If the lens is totally opaque, preoperative ultrasonography should be performed to rule out a chronic retinal detachment. The presence of an afferent pupillary defect is a poor prognostic sign.
Maternal infection (rubella) – This type of cataract has not been seen in countries where rubella has been eradicated, but continues to occur in some parts of the world.
Iatrogenic
- Radiation – External beam radiation is avoided in patients with retinoblastoma. The eye is typically shielded if radiation is given to the brain or other parts of the head and neck.
- Systemic steroids are very rare causes of cataracts in children. Inhaled steroids for asthma do not cause cataracts. The typical steroid-induced cataract is posterior subcapsular.
- Vitrectomy – A large percentage of children who undergo vitrectomy develop cataracts. These are mostly posterior subcapsular.
- Laser for retinopathy of prematurity – Cataracts can develop from thermal injury to the lens when a prominent tunica vasculosa lentis is present.
Morphology
As mentioned above, it is important to utilize the appropriate terminology to describe pediatric cataracts. The morphology can give a clue to the underlying etiology (isolated or associated with systemic disease), and possibly to the visual prognosis following surgery.
Diffuse/Total
This is not an uncommon type of congenital cataract. There are no specific causes of diffuse or total cataracts.
Anterior
Anterior polar – The opacity is in the capsule itself and can protrude into the anterior chamber as a small mammillation. There may be an underlying circular layer of cortical opacity slightly larger than the white polar opacity. While the majority are stable and do not interfere with vision, some can progress and require surgical removal. They can be dominantly inherited, especially in bilateral cases. Unilateral cases can be associated with anisometropia (astigmatism or hyperopia), which if left untreated can cause amblyopia, even if the cataract itself is not visually significant.
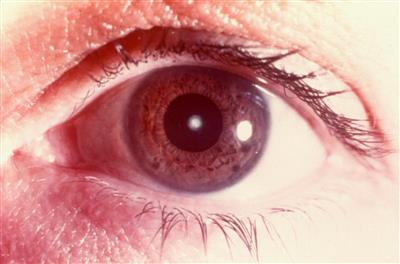
Figure 1. Anterior polar cataract.
Pyramidal – These are usually larger than polar cataracts and more likely to progress to visual significance. They are difficult to remove with a vitrectomy instrument and may require excision and removal with forceps before the rest of the lens is aspirated.
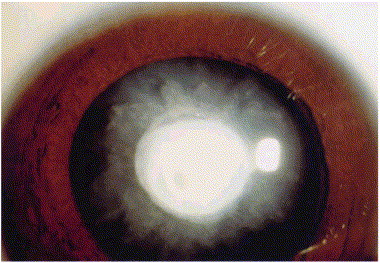
Figure 2. Pyramidal cataract.
Anterior lenticonus – This refers to a thinned-out central anterior capsule with or without anterior cortical opacities. Anterior lenticonus is said to be characteristic of Alport syndrome. Spontaneous rupture of the lens can occur, resulting in a hydrated total cataract.
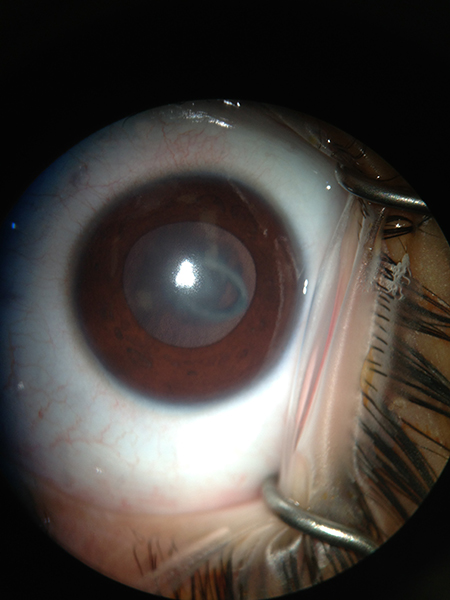
Figure 3. Anterior lenticonus (Courtesy of K. David Epley).
Cortical lamellar
In this type of cataract, the opacification is of a lamella (an ovoid layer of cortex) that can be visualized between adjacent clear lamellae. These are frequently associated with radial “rider” opacities. Familial lamellar cataracts are mostly autosomal dominant and are generally associated with a good visual prognosis after their removal. They can be stable or may be associated with progressive opacification of intervening cortex, necessitating removal.
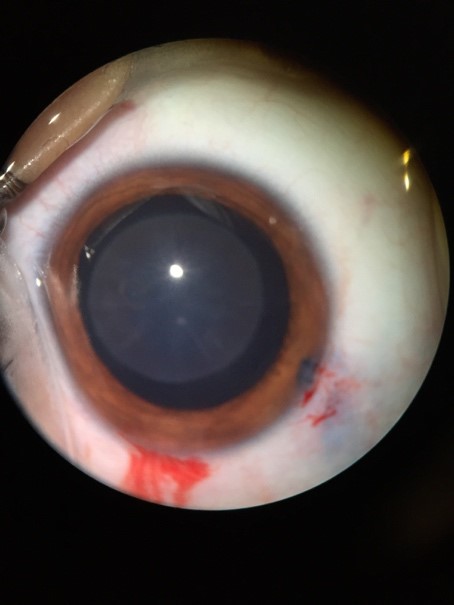
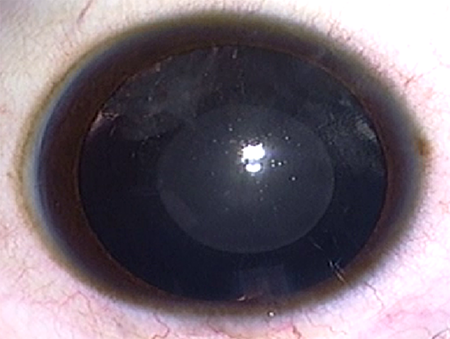
Figure 4. Lamellar cataracts (Top: Courtesy of K. David Epley, MD. Bottom: Courtesy of Faruk H. Örge, MD).
Fetal nuclear
These opacities occupy the central-most part of the lens. They can be dot-like or can be quite dense. They generally measure 2-3.5 mm and can be associated with microphthalmia. They are said to be associated with a higher incidence of postoperative glaucoma because of associated microphthalmia and the need for surgery early in infancy.
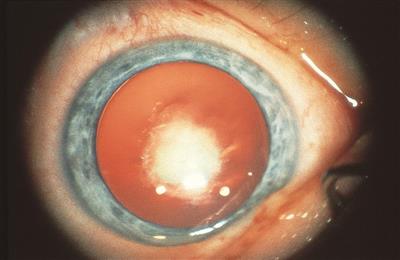
Figure 5. Congenital nuclear cataract.
Posterior polar
In this type of cataract, the opacity is in the capsule itself. It is necessary to differentiate posterior polar from posterior subcapsular cataracts. Posterior polar cataracts are genetically determined and some have been associated with mutations in PITX3.
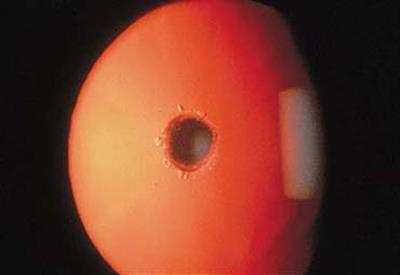
Figure 6. Posterior polar cataract.
Posterior lentiglobus (lenticonus)
In this group of conditions, the central and sometimes paracentral posterior capsule is thin and bulges posteriorly. This usually occurs at the location where the hyaloid system attaches to the eye. The distortion can cause a localized area of extreme myopic refraction. There may or may not be subcapsular cortical opacification. Interference with vision can be the result of optical distortion or of capsular opacification. Most cases are unilateral, although bilateral and familial cases have been reported. Surgery is associated with good visual outcomes in most cases. Spontaneous rupture of the lens can rarely occur, leading to abrupt progression to total cataract.
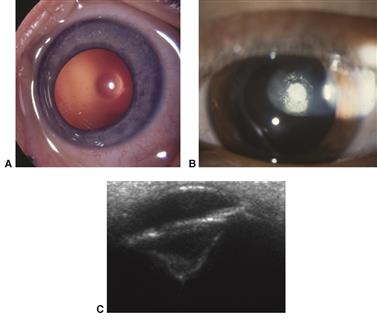
Figure 7. Posterior lentiglobus (lenticonus) cataract. (A) Early clear defect in central posterior capsule and (B) early opacification of central defect. (C) Ultrasound biomicroscopy of advanced posterior lenticonus.
Posterior subcapsular
These can be congenital but are more commonly acquired as a result of injury or steroid use. The opacities are cortical and do not involve the capsule proper.
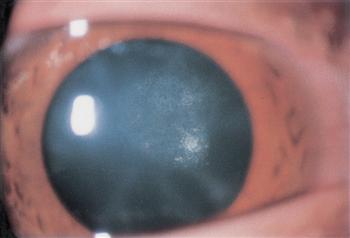
Figure 8. Posterior subcapsular cataract.
Persistent fetal vasculature (PFV) (severe varieties are still referred to as persistent hyperplastic primary vitreous)
The lens opacities in patients with PFV are generally capsular and can be associated with shrinkage, thickening, and vascularization of the capsule. There may be a posterior plaque outside or involving the lens capsule with a clear lens that nonetheless must be treated as a cataract.
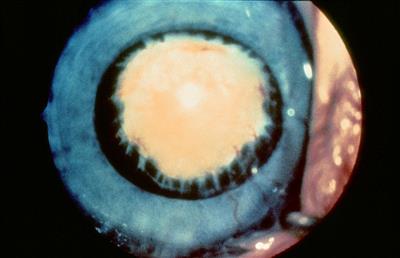
Figure 9. Persistent fetal vasculature.
Traumatic disruption of lens
In children, traumatic anterior lens capsule rupture quickly results in a hydrated white cataract. However, in children, lens cortex in the anterior chamber may be well tolerated without an intraocular pressure (IOP) rise. Cataract surgery can often be delayed for a few days or up to 3 or 4 weeks to allow the traumatic iritis to subside before the cataract and IOL surgery.
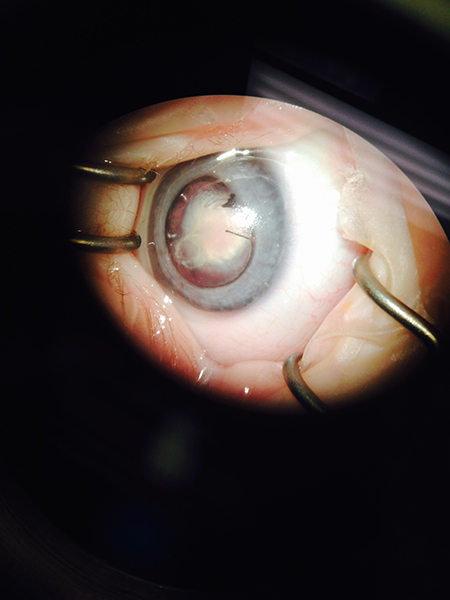
Figure 10. Traumatic disruption of lens (Courtesy of K. David Epley).
Evaluation and work-up
Role of vision screening
Vision screening is mandatory to detect cataracts as soon as possible. Late detection may result in poor visual outcomes. All newborns must have red reflex screening, ideally followed by another red reflex examination at the 6-8 week neonatal checkup. Red reflex testing is done by using direct ophthalmoscope from a distance of 1-2 feet in a darkened room. Preschool vision screening (at 3 and 5 years) is often done in the community. Photo screeners are used in preverbal and verbal children. These may help the pediatrician save time in screening. They work by a computer analyzing the red reflex for inequality in color, intensity, or clarity. New screeners utilizing polarized laser light are more accurate at detecting decreased vision. The presence of any opacities, an absent red reflex, or leukocoria should prompt an urgent referral to a pediatric ophthalmologist.
Evaluation by the ophthalmologist
A detailed history is taken that includes asking about the child’s developmental milestones, and about health problems in the siblings and parents. Visual assessment is conducted by using age-appropriate testing. When the child is two months old, vision assessment can be done with forced preferential looking techniques (eg, Teller acuity cards, Cardiff cards), fixation and following evaluation, and assessing objection to occlusion of each eye. The presence or absence of nystagmus is noted. Subjective visual testing (HOTV matching, LEA symbols, or tumbling Es) is done as soon as the child is able to play a matching game or identify the symbols and letters. These tests can usually be done at age 3 years and above.
Biomicroscopy (standard or portable slit lamp examination) is completed. Severity and morphology of the cataract and any associated abnormalities of cornea or anterior segment are documented. Examination of siblings and parents might indicate inherited cataracts. Intraocular pressure is checked if possible.
If there is a view of the retina, full retinal examination documenting optic nerves, retina, and fovea is performed. If there is no view, ultrasonography (B-scan) is carried out. If there is trauma, then child abuse must be ruled out. In unilateral cataracts, laboratory tests are not needed.
For bilateral cataracts, if there is family history of childhood cataracts, the child has no other medical problems, and the parents have lens opacities, then systemic and laboratory evaluations are not needed. If there is no family history of cataracts, a pediatric systemic evaluation is required because these cataracts may be associated with systemic or metabolic disease. Laboratory tests may also be needed. The ophthalmologist often works in conjunction with a pediatrician and/or a clinical geneticist when directing the laboratory work-up. A urine test for reducing sugars, TORCH (toxoplasmosis, rubella, cytomegalovirus, varicella) screening, a Venereal Disease Research Laboratory (VDRL) test for syphilis, and a blood test for calcium, phosphorus, glucose, and galactokinase levels can be checked.
Most inherited cataracts are autosomal dominant. Recessive and X-linked cataracts are less common. Genetic testing is a rapidly evolving field. Mutations that cause congenital cataracts have been discovered in over 100 genes. Using the latest sequencing tests, it will be possible to check all genes involved in congenital cataracts from one blood sample. This might lead to quicker and cheaper personalized treatment and counseling by the geneticist.
If cataracts are less than 3 mm in diameter or are of partial density, they may be observed or treated with dilating drops. Any dense central opacity in the lens of three or more mm in a young child is significant and requires surgery. In addition to the size of cataract, blackening of the retinoscopic reflex is the most important factor determining need for a surgery.5 In an older child, any opacity causing a decrease in quality of life should be considered for surgery. At the same time, the loss of accommodation that occurs when a child’s lens is removed should be taken into account when making a surgical decision. With increasing age, visual demands of the child increase and the assessment of whether a partial cataract is visually significant has to be constantly revisited.
Biometry is done to get keratometry measurements, preferably without a speculum. Axial length is often measured in children by A-scan ultrasound, with the immersion method being more accurate than the contact method.6,7 Often, these measurements are not possible in clinic and examination under anesthesia is required. If the child is older and cooperative, and the cataract is not very dense, then optical biometry is done.
For calculation of the IOL, third-generation theoretical formulae (eg, SRK/T, Holladay I & II, Hoffer Q I & II, and Haigis) can be used. Target refraction may be aimed for initial hypermetropia (high or low) or emmetropia. Suggested target refractions for age are given in Table 2.5 Other factors such as amblyopia, fellow eye condition or refraction, assumed compliance, and parental refractive error should also be taken into consideration when interpreting the table: one IOL power choice for every age does not work for every situation.
|
Table 2. Age at cataract surgery and residual refraction recommendations for target refraction
|
|
Age at cataract surgery
|
Residual refraction (Diopters)
|
|
<6 months
|
+6 to +10
|
|
6-12 months
|
+4 to +6
|
|
1 -3 years
|
+4
|
|
3-4
|
+3
|
|
4-6
|
+2 to +3
|
|
6-8
|
+1 to +2
|
|
>8
|
+1 to 0
|
Surgery
Who should perform the surgery
Adult cataract surgery is a major emphasis of residency training programs in ophthalmology. The skills needed to perform adult cataract surgery are also important for performing pediatric cataract surgery, but additional skills are needed for the pediatric surgery. Pediatric cataract surgery should only be performed by ophthalmic surgeons who perform them on a weekly or biweekly basis so that they can perform them with a high level of competency.8 For this reason, most large group practices assign only one surgeon in their practice to perform these surgeries. When possible, children should be referred to regional centers where large numbers of pediatric cataract surgeries are performed. After the postoperative period, in most cases these children can then be followed on a long-term basis by a local doctor and only referred back to the regional center if problems arise. Pediatric ophthalmologists interested in performing pediatric cataract surgery should pursue fellowship training at an institution where they will be trained how to perform pediatric cataract surgery. After completing their fellowship, they should take instructional courses as needed to incorporate new techniques as they arise. While adult cataract surgeons are usually skillful at performing intraocular surgery, they often have not been taught the special techniques required to successfully perform pediatric cataract surgery. If they are interested in performing pediatric cataract surgery, they should seek out opportunities to learn its best practices either by observation or by taking instructional courses.
Timing and critical period
In the 1960s, Hubel and Wiesel9 introduced the concept of a “latent period” and a “critical period” for visual development. During the latent period, visual deprivation has no lasting effect on vision in the deprived eye. After the latent period, there is a critical period during which visual deprivation results in irreversible vision loss in the deprived eye. The critical period for a child with a cataract extends to age 9-10 years.
Unilateral
The optimal age for performing cataract surgery in a child with a unilateral congenital cataract is generally agreed to be 6 weeks of age. Birch and Stager10 evaluated the relationship between the age at cataract surgery and visual outcomes in newborns with a dense unilateral congenital cataract. The model that best fit their data was bilinear, with no differences in the visual outcomes if the surgery was performed between birth and age 6 weeks. However, after age 6 weeks, there was a linear decline in visual outcomes related to the age at cataract surgery. Their model would suggest that there is a 6-week latent period for dense unilateral cataracts in humans. More recently, Hartmann et al11 found that the age at cataract surgery was only weakly associated with visual acuity. While the median visual acuity was better among patients who had cataract surgery between ages 4 and 6 weeks, the association between age at cataract surgery and the visual outcome was less robust than the data reported by Birch and Stager.
Bilateral
It is generally agreed that bilateral congenital cataracts should be removed by 8 weeks of age to achieve the best visual outcomes. Lambert and coworkers12 noted that delaying cataract surgery to 10 weeks of age or later increased the likelihood of a 20/100 or worse visual outcome. Birch and coworkers13 reported a bilinear relationship between the age of surgery and the visual outcome in infants with dense bilateral congenital cataracts. Between birth and 14 weeks of age they noted progressively worse visual outcomes the older a child was at the time of cataract surgery. However, after age 14 weeks until 31 weeks, the visual outcome was independent of the child’s age at the time of cataract surgery. Since it is unclear if there is a latent period in children with dense bilateral congenital cataracts, the timing of cataract surgery in these children is often determined by other comorbidities and the increased risk of glaucoma associated with very early cataract surgery.
Threshold/indication for surgery
Determining the need for surgery in preverbal children
Dense cataracts that block the red reflex before the pupils are dilated and are associated with abnormal visual behavior should be removed during infancy. Other signs suggestive of visually significant cataracts are strabismus in a child with a unilateral cataract or nystagmus in a child with bilateral cataracts. Incomplete cataracts do not always require cataract surgery. If the child has incomplete cataracts and normal visual behavior and the fundi can be clearly viewed with an ophthalmoscope, cataract surgery should be deferred. Generally, posterior lenticular opacities are more visually significant than anterior lens opacities. If the incomplete cataract(s) is unilateral or asymmetrical, part-time patching therapy of the normal/better eye may be beneficial to improve or maintain vision in the most affected eye.
Visual acuity chart threshold for surgery
Generally, cataract surgery should not be performed on children with bilateral cataracts who have best corrected visual acuity of 20/40 or better. However, the visual threshold for performing cataract surgery should be tailored to the needs of the child. For instance, if a child has visual acuity worse than 20/40, but is doing well in school and does not have any visual behavioral problems, cataract surgery can be deferred until later. Visual behavior is less helpful in assessing the need for cataract surgery in children with a unilateral cataract. Generally, if best corrected visual acuity cannot be improved to 20/50 or better with amblyopia therapy, cataract surgery should be considered.
Visual dysfunction weighed against post-op loss of accommodation
The improvement in visual acuity associated with cataract surgery must be weighed against the loss of accommodation associated with removing the crystalline lens. While multifocal or accommodative IOLs are available for adults and may mitigate, somewhat, the loss of accommodation associated with cataract surgery, they are infrequently implanted in growing children because of the refractive changes that occur as an immature eye grows. Parents should be told that while their child may see more clearly after undergoing cataract surgery, the child will have to wear bifocals in order to optimize distance and near vision.
Informed consent/parenting counseling
The risks and benefits of cataract surgery should be clearly outlined to parents. It is often helpful to show them models of the eye or illustrations to help them understand what a cataract is and how cataract surgery will be performed. The importance of amblyopia therapy and optical correction following cataract surgery should be discussed in detail. The pros and cons of implanting an IOL or creating a posterior capsulotomy should be discussed with parents. It should also be explained that the US Food and Drug Administration (FDA) has not approved the implantation of IOLs in children, and their use in children is off-label.
Immediate sequential bilateral cataract surgery for children
The option of performing immediate sequential bilateral cataract surgery should be discussed with the parents of infants, particularly if there are comorbidities that increase the risk of general anesthesia. They should be informed of the risks and benefits associated with immediate sequential bilateral cataract surgery, including the benefit of administering only one general anesthetic, but the increased risk of bilateral endophthalmitis.14 It should also be explained that precautions will be taken to reduce the risk of endophthalmitis, including using different trays of instruments for each eye, disposable cannulas, re-draping between eyes, and using different lots of irrigating solution and medications for each eye.
Anesthesia management considerations
General anesthesia is required to perform pediatric cataract surgery. The anesthetic agents should be administered only under the direct supervision of an anesthesiologist with special experience or special training in pediatric anesthesia. Very young children, especially when born prematurely, will often need to be hospitalized overnight after cataract surgery because of their increased risk of experiencing apnea after undergoing general anesthesia. Cataract surgery can be performed as an outpatient procedure in older children.
Operative techniques
Preoperative preparation is typically done using povidone-iodine. The use of intracameral antibiotics in either the irrigating solution or injected postoperatively has been extensively tested in adults, and while not widely practiced among pediatric cataract surgeons, trends forecast more acceptance in the coming years.
Surgical incisions are usually done anteriorly through clear cornea or using a scleral tunnel. If no IOL is to be placed, a minority of surgeons will opt for a posterior pars plana/plicata approach. Continuous curvilinear capsulorhexis with or without capsular staining is the gold standard capsulotomy, but vitrectorhexis also works well and is commonly used in the first few years of age when the capsule is very elastic. The anterior chamber is maintained with either a separate non-held infusion cannula (an anterior chamber maintainer) or with matched hand-held bimanual irrigation and aspiration handpieces. Pupil dilation is enhanced with non-preserved epinephrine or phenylephrine/ketorolac (recently FDA approved for adults) added to the infusion bottle.
The lens contents are aspirated completely (Figure 11). Phacoemulsification ultrasound energy is never needed with pediatric cataracts. Hydrodissection is not necessary, but can be used at the surgeon’s discretion. However, the large number of pediatric lens opacities associated with posterior capsule pathology must be noted. Hydrodissection is contraindicated in posterior polar cataracts.
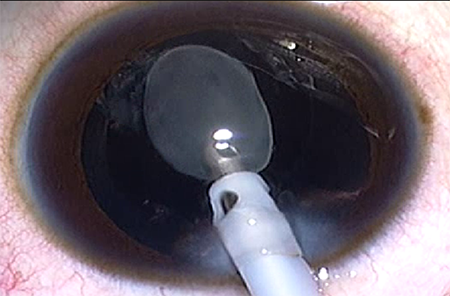
Figure 11. An irrigation/aspiration handpiece removing a lamellar cataract (Courtesy of Faruk H Orge).
A posterior chamber IOL inserted into the capsular bag is always preferred, but ciliary sulcus placement of a foldable acrylic or single-piece rigid IOL can be done. In cases of no capsular support, posterior chamber IOLs can be sewn in place; however, placement of iris (claw) fixated lenses is becoming more popular.
In children too young to tolerate a YAG laser posterior capsulotomy in the office, a primary posterior capsulotomy at the time of initial cataract surgery is recommended. This can be done either before or after an IOL is placed and can be done anteriorly through the corneal tunnel or posteriorly through the pars plana. All but the smallest watertight incisions should be closed in children, usually with a synthetic absorbable 10-0 suture.
Postoperative medications
Antibiotics
After pediatric cataract surgery, either moxifloxacin or tobramycin, the two most widely used antibiotic eye drops, can be used. The eye drops are instilled four times per day for a week. There is no need to prescribe systemic antibiotics.
Steroids
Prednisolone eye drops are the mainstay of treatment to control severe inflammation, which is generally inevitable. In some cases of very severe postoperative inflammation, steroid eye drops must be instilled as frequently as on an hourly basis. Otherwise, the routine dosage range is 4-8 times per day. Some surgeons advocate supplementing the topical steroid with oral prednisolone dosed at 1 mg/kg/day for the first week to help reduce inflammation.
Cycloplegics and mydriatics
Homatropine or atropine eye drops are sometimes used postoperatively as cycloplegics. The possible side effects of atropine must be discussed with the patient’s parents.
Follow-up
Pediatric cataract cases are normally examined on the first postoperative day. The next follow-up depends on the amount of inflammation but is most often at 1 week after surgery. Once both eyes are operated on, periodic examinations are required to determine refraction, IOP, and retinal evaluation. Glasses or contact lenses are prescribed as early as possible, preferably within the first week for aphakic correction and within 4 weeks for residual refractive error in pseudophakic children.
Frequency
Typical follow-up frequency is as follows: postoperative day 1, week 1, month 1, month 3, every 3 months for 2 years, and thereafter every 6 months for 3 years.
Evaluation
It is crucial to check visual acuity, ocular alignment, IOP, refraction, and clarity of the visual axis at every visit. Should there be any complication detected in any of the follow-up visits, it should be tackled promptly.
Optical rehabilitation after cataract surgery
Since uncorrected refractive error in the early years can lead to amblyopia, attention to appropriate refractive correction after cataract surgery is crucial in order to obtain good final visual acuity. For infants and toddlers, refractive correction should result in good near vision (myopic refraction of approximately -2 diopters). However, correction for distance vision and a bifocal correction for near viewing should be offered after the age of 2 or 3 years, or by pre-kindergarten. Children who use a contact lens may also benefit from a spectacle overcorrection after age 2 or 3 years.
Spectacles
For children who have IOL implantation, some residual refractive error is typical and spectacle correction may be needed for distance and/or near viewing. Additionally, when IOL implantation occurs at an early age, the growing eye will experience a myopic shift, so that changing refraction is expected with residual hyperopia in the early years but some degree of myopia expected later. Correction of aphakia with spectacles may be preferred for infants and young children in whom IOL implantation is not possible or is purposely delayed. Aphakic spectacles are generally well tolerated, particularly by children who are bilaterally aphakic. Unilateral aphakia can also be corrected with spectacles, though this is less desirable because of marked image-size disparity (aniseikonia) and potential disruption of binocular vision, if present.
Contact lenses
Contact lens correction of aphakia is often planned for very young infants after lensectomy, typically with either a silicone elastomer lens (extended wear) or rigid gas permeable lens (daily wear). One advantage of contact lens wear is easy adjustment in power for the rapidly changing refractions encountered in young children. Contact lens correction of residual refractive error is also possible after IOL implantation, and is sometimes requested by adolescent patients.
Postoperative Complications and Sequelae
Postoperative complications after pediatric cataract surgery are inversely proportional to the age at the time of surgery. Associated ocular anomalies, surgical technique, and follow-up duration are some of the other important variables influencing the prevalence and severity of the postoperative complications after cataract surgery in children.
Visual axis opacification
If the posterior capsule is left intact at the time of cataract surgery in children, posterior capsule opacification (PCO) is inevitable. The younger the child, the more acute will be the opacity. After primary posterior capsulectomy and vitrectomy, visual axis opacification (VAO) is rare in older children; however, despite posterior capsulectomy and vitrectomy, VAO is commonly observed in infants. VAO in infants receiving posterior capsulectomy and vitrectomy typically requires surgical removal from 3 months to 1 year after the original surgery, while PCO in older children who had an intact posterior capsule typically requires Nd:YAG laser or surgical removal of the PCO 2 years or more after cataract surgery.15
Glaucoma
Secondary glaucoma is the most sight-threatening complication of pediatric cataract surgery. Younger age at the time of surgery is the most commonly reported risk factor. Open-angle glaucoma can develop months to many years after the surgery, and children must be followed for this regularly for their entire life.
Inflammatory complications
Due to increased tissue reactivity, inflammatory complications (eg, anterior chamber cell and flare, cell deposits on the IOL optic, posterior synechiae, etc.) are more frequently observed in children. Toxic anterior segment syndrome (TASS) is a rare inflammatory condition usually observed during the early postoperative period.
Contact lens related
Bacterial keratitis, corneal opacity due to tight contact lenses, and corneal vascularization are the most common contact lens-related complications.
IOL malposition
Excessive capsular fibrosis and asymmetric IOL fixation are the most common causes leading to malposition of an IOL. It can also occur because of traumatic zonular loss and/or inadequate capsular support. The IOL may have to be repositioned or explanted in some cases when there is significant decentration/dislocation.
Endophthalmitis
The incidence of postoperative endophthalmitis in children is similar to that reported in adult surgery. Common organisms are Staphylococcus aureus, Staphylococcus epidermidis, and Streptococcus viridans. Recent studies in adults have reported a marked decrease in endophthalmitis when intracameral antibiotics are used. In the US, the absence of an ophthalmic preparation specific for use as an intracameral injection has slowed adoption of intracameral antibiotics for fear of toxicity from dilution errors during medication preparation. Studies in adults have used cefuroxime, vancomycin, and undiluted moxifloxacin.16, 17, 18, 19, 20
Retinal detachment
The incidence of retinal detachment (RD) following pediatric cataract surgery appears to have decreased markedly as surgical techniques have advanced. However, because RD may develop many years after surgery, a retinal examination is recommended after cataract surgery at least yearly. This is especially important for those eyes at higher risk for RD by virtue of a long axial length for age, persistent fetal vasculature, traumatic cataract, ectopia lentis, Stickler syndrome, repeated surgeries, etc.
Myopic shift
A tendency toward axial elongation and a myopic shift of refraction is well known. This is more concerning if the child receives an IOL. The younger the child at the time of implantation, the higher the myopic shift. High myopia in pseudophakic eyes can be treated using spectacles or contact lens. Alternatively, IOL exchange, piggyback IOL implantation, or corneal refractive surgery may be required.
Other complications
Corneal edema, corneal decompensation, iris prolapse, heterochromia iridis, suture-related complications, a postoperative IOP spike, astigmatism, ptosis, or phthisis bulbi are other complications reported after pediatric cataract surgery.
Strabismus
Strabismus can coincide with congenital cataract and is more commonly seen in unilateral cases but not rare in bilateral cataract cases, especially when nystagmus is present. Esotropia is the most common form of strabismus in congenital cataract, although cyclovertical strabismus may also contribute to the clinical picture. In a minority of patients, exotropia of the involved eye is the presenting sign of congenital cataract.
Management of co-existing amblyopia
Deprivation amblyopia is very common in children with unilateral cataract, especially when the opacity is congenital or infantile. Also, children with bilateral cataracts can develop unilateral or bilateral deprivation amblyopia when the cataracts are asymmetric, when they are removed too late, or when the aphakia is not properly corrected. Sensory nystagmus will further limit visual outcome. The management of the amblyopia should start as soon as possible, since compliance in small infants is better than in 2- to 3-year-old children. Patching of the sound eye is the mainstay of treatment. However, atropine penalization can be an alternative if the amblyopic eye can take over fixation. This is quite rare because the aphakic or pseudophakic eye has lost accommodation and for that reason is always at a disadvantage to the sound eye, which can accommodate up to 10 diopters depending on the child’s age. In bilateral aphakic eyes with contact lenses, the contact lens of the dominant eye can be removed a few hours or several days per week as a penalization strategy. The younger the child, the better the effect of amblyopia treatment per hour of occlusion.
Low vision rehabilitation and quality of life measures
In cases when the treatment of the congenital cataract is less successful, low vision rehabilitation has an important role in how the patient can cope with the limited visual capacities in education and daily life. In most countries, visual rehabilitation and education for visually impaired and blind patients are organized either by the government, various nongovernmental organizations, or private foundations. The motto should be: Use the remaining visual function with all other senses to achieve the optimum quality of life.
Future directions
Early detection will allow more timely treatment of pediatric cataract in the future. Vision screening programs and improved education of primary health care workers and the public will help with this evolution. Surgical techniques continue to improve and will allow childhood cataract removal with less and less surgical trauma. Planning for IOL implantation will become easier as our knowledge of myopic shift and axial globe growth evolve. Ultimately, future IOL technological advances will be aimed at restoration or preservation of youthful accommodation and the ability to easily compensate for the inevitable myopic shift. Intracameral medications specifically for ophthalmic use are being developed and these will improve outcomes for children as they decrease the reliance we now have on the ability of parents to administer topical medications after surgery.
References
- Gilbert C. Worldwide causes of blindness in children. In: Wilson ME, Saunders RA, Trivedi RH, eds. Pediatric Ophthalmology: Current Thought and a Practical Guide. Heidelberg, Germany: Springer; 2009: 47-60.
- Haargaard B, Wohlfahrt J, Fledelius HC, Rosenberg T, Melbye M. Incidence and cumulative risk of childhood cataract in a cohort of 2.6 million Danish children. Invest Ophthalmol Vis Sci. 2004;45(5):1316-1320.
- Xu LT, Traboulsi EI. Genetics of congenital cataracts. In: Wilson ME, Trivedi RH, editors. Pediatric Cataract Surgery: Techniques, Complications and Management. Philadelphia: Lippincott Williams & Wilkins; 2014: 1-8.
- Gillespie RL, O'Sullivan J, Ashworth J, Bhaskar S, Williams S, Biswas S, et al. Personalized diagnosis and management of congenital cataract by next-generation sequencing. Ophthalmology. 2014;121(11):2124-2137 e1-2.
- Serafino M, Trivedi RH, Levin AV, Wilson ME, Nucci P, Lambert SR, et al. Use of the Delphi process in paediatric cataract management. Br J Ophthalmol. 2015. doi: 10.1136/bjophthalmol-2015-307287. [Epub ahead of print].
- Trivedi RH, Wilson ME. Prediction error after pediatric cataract surgery with intraocular lens implantation: Contact versus immersion A-scan biometry. J Cataract Refract Surg. 2011;37(3):501-505.
- Trivedi RH, Wilson ME. Axial length measurements by contact and immersion techniques in pediatric eyes with cataract. Ophthalmology. 2011; 118(3):498-502.
- Bell CM, Hatch WV, Cernat G, Urbach DR. Surgeon volumes and selected patient outcomes in cataract surgery: a population-based analysis. Ophthalmology. 2007; 114(3):405-410.
- Hubel DH, Wiesel TN. The period of susceptibility to the physiological effects of unilateral eye closure in kittens. J Physiol. 1970; 206(2):419-436.
- Birch EE, Stager DR. The critical period for surgical treatment of dense congenital unilateral cataract. Invest Ophthalmol Vis Sci. 1996; 37(8):1532-1538.
- Hartmann EE, Lynn MJ, Lambert SR, Infant Aphakia Treatment Study Group. Baseline characteristics of the infant aphakia treatment study population: predicting recognition acuity at 4.5 years of age. Invest Ophthalmol Vis Sci. 2014; 56(1):388-395.
- Lambert SR, Lynn MJ, Reeves R, Plager DA, Buckley EG, Wilson ME. Is there a latent period for the surgical treatment of children with dense bilateral congenital cataracts? J AAPOS. 2006;10(1):30-36.
- Birch EE, Cheng C, Stager DR Jr, Weakley DR Jr, Stager DR Sr. The critical period for surgical treatment of dense congenital bilateral cataracts. J AAPOS. 2008; 13:67-71.
- Dave H, Phoenix V, Becker ER, Lambert SR. Simultaneous vs sequential bilateral cataract surgery for infants with congenital cataracts: Visual outcomes, adverse events, and economic costs. Arch Ophthalmol. 2010; 128(8):1050-1054.
- Wilson ME, Jr., Trivedi RH, Buckley EG, Granet DB, Lambert SR, Plager DA, et al. ASCRS white paper. Hydrophobic acrylic intraocular lenses in children. J Cataract Refract Surg. 2007; 33(11):1966-1973.
- Braga-Mele R, Chang DF, Henderson BA, Mamalis N, Talley-Rostov A, Vasavada A. ASCRS Clinical Cataract Committee. Intracameral antibiotics: Safety, efficacy, and preparation. J Cataract Refract Surg. 2014; 40(12):2134-2142.
- Tan CS, Goh AG, Ngo WK, Lim LW, Fam HB. Safety of intracameral antibiotic use after cataract surgery. J Cataract Refract Surg. 2014; 40(11):1940-1941.
- Shorstein NH, Winthrop KL, Herrinton LJ. Decreased postoperative endophthalmitis rate after institution of intracameral antibiotics in a Northern California eye department. J Cataract Refract Surg. 2013; 39(1):8-14.
- Espiritu CR, Caparas VL, Bolinao JG. Safety of prophylactic intracameral moxifloxacin 0.5% ophthalmic solution in cataract surgery patients. J Cataract Refract Surg. 2007; 33(1):63-68.
- Beselga D, Campos A, Castro M, Fernandes C, Carvalheira F, Campos S, Mendes S, Neves A, Campos J, Violante L, Sousa JC. Postcataract surgery endophthalmitis after introduction of the ESCRS protocol: a 5-year study. Eur J Ophthalmol. 2014; 24(4):516-519.