Embryology
The cranial nerve nuclei have developed by 4 weeks of gestation. All of the cranial motor nuclei arise from the midbrain region. The hindbrain contains rhombomeres, which are a transiently divided segment of the developing neural tube, within the hindbrain region (a neuromere) in the area that will eventually become the rhombencephalon. The rhombomeres appear as a series of slightly constricted swellings in the neural tube, caudal to the cephalic flexure. Rhombomeres determine the pattern of the following maturation of the rhombencephalon into its final parts. The final parts are defined as the pons, cerebellum and medulla. There are eight divisions in the hindbrain for nuclei of cranial nerves IV thru VII and IX thru XII. The 3rd nerve nuclei develop outside of the hindbrain. The sensory ganglia arise dorsal to the pharyngeal arches, and the parasympathetic ganglia arise from neural crest cells. (Figure 1)
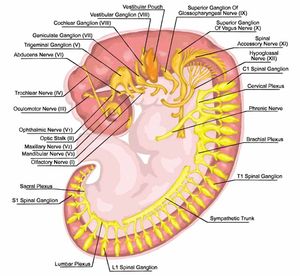
Figure 1. Formation of the branchial or pharyngeal arches and the nervous system of the human embryo - embryonic development of vertebrates evolutionary origin, embryonic components and systems (Image by Stilhi/shutterstock.com)
Anatomy
The oculomotor nerve contains the somatic motor fibers for the medial, inferior, superior, inferior oblique muscles and the levator complex (Figure 2). It also contains the parasympathetic fibers for the iris and ciliary body. Finally, sympathetic and trigeminal fibers travel with the 3rd nerve. The oculomotor nucleus is located from the posterior commissure to the trochlear nerve in the periaqueductal mesencephalon. The complex motor organization connects to other cranial nerve nuclei, the medial longitudinal fasciculus and the paramedian pontine reticular formation. The motor nuclei are organized with the superior rectus nuclei connected to the contralateral superior rectus. The medial rectus, inferior rectus and inferior oblique nuclei connect to the ipsilateral muscle. The motor nuclei are distributed along the nucleus with three subgroups for the medial rectus. The levator palpebrae nuclei supplies both ipsilateral and contralateral function from a midline subnucleus.
From the nuclei, the neural fiber fascicles within the brainstem begin dividing into the superior and inferior divisions. The mesencephalon blood supply is by the basilar artery at the origin or the superior cerebellar and posterior cerebral arteries. The trunk of the 3rd nerve passes between these as it leaves the brainstem, a location for possible compression.
The oculomotor nerve is mixed with somatic and parasympathetic fibers from the 3rd nerve nuclei along with fibers from the sympathetic chain and trigeminal nerve trunk in the cavernous sinus and orbit. Larger somatic fibers supply the muscles and finer parasympathetic fibers innervate the iris and ciliary body. When entering the cavernous sinus, the 3rd nerve courses along the lateral wall with the trochlear nerve and a portion of the trigeminal nerve. The blood supply in the cavernous sinus is from branches of the meningo-hypophyseal trunk and the ophthalmic artery.
The 3rd nerve passes through the superior orbital fissure and annulus of Zinn into the orbit and divides into distinct superior and inferior trunks. The superior branch divides to the superior rectus and levator muscle. The nerve fibers for the levator may pass through the lateral portion of the superior rectus. The inferior branch divides to innervate the inferior rectus and inferior oblique. Again, the inferior oblique fibers may pass through the inferior rectus muscle first. The parasympathetic fibers in the superficial superior portion of the inferior branch of the 3rd nerve travel to the ciliary ganglion.
The pupillary sphincter and ciliary muscles are controlled by the parasympathetic output from the Edinger-Westphal nucleus, part of the 3rd nerve nuclei. This nucleus contributes to both ipsilateral and contralateral fibers. The pupillary fibers synapse in the orbital ciliary ganglion, then pass through to the iris sphincter. These fibers start in the superior portion of the 3rd nerve and rotate medially and inferiorly in the cavernous sinus. Finally, they branch off with the inferior branch of the 3rd nerve. The fibers are in the nerve periphery.
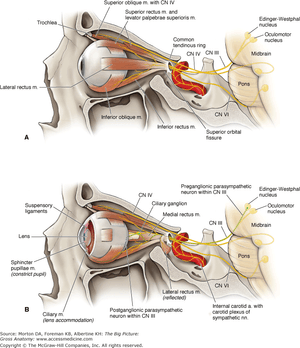
Figure 2. The course of the oculomotor (third cranial) nerve. (Morton DA, Foreman KB, Albertine KH: The Big Picture: Gross Anatomy: www.accessmedicine.com. Copyright ©The McGraw-Hill Companies, Inc. All rights reserved.)
Oculomotor Anomalies
Congenital
Congenital 3rd nerve palsies may be developmental or acquired. The rare congenital 3rd nerve palsy usually involves ptosis, an ophthalmoplegia of some degree, and pupillary mydriasis. It is often an isolated finding but may be associated with aberrant regeneration, other cranial nerve palsies, other central nervous system anomalies, or developmental delay. Frequent pupil involvement does not indicate a compressive lesion, as in adults.
Amblyopia is frequently seen in 3rd nerve palsy in the affected eye (although exceptions are seen) requiring early and aggressive treatment.
Several congenital syndromes have been described. "Congenital adduction palsy with synergistic divergence" is a unilateral medial rectus paresis with bilateral abduction upon looking into the field of action of the affected medial rectus. The lateral rectus on the affected side is innervated by the 3rd nucleus. The "vertical retraction syndrome" has a limitation of supra- and infraduction with co-contraction and retraction upon vertical gaze, most prominently with infraduction. Anomalous innervation is the cause. "Cyclic oculomotor paresis" is cycles of paresis with simultaneous lid elevation, adduction, miosis, and accommodation every two minutes. The spasm lasts only 10 to 30 seconds. This is very rare with an unknown cause.1
Acquired
Acquired pediatric 3rd nerve palsies in the pediatric age group are associated with traumatic deliveries, especially with forceps use. Most will resolve with variable amounts of residual paresis and are associated with aberrant regeneration. An acquired, slowly progressive third nerve palsy with a meningioma or schwannoma may be associated with aberrant regeneration also. Decreased corneal sensitivity in neurofibromatosis type 2 indicates 5th nerve involvement with the 3rd nerve palsy.2
Adult acquired 3rd nerve palsy is bilateral 11% of the time, a complete palsy in 33%, and has no other associated neurologic sign in 36%.3 The list of causes of a 3rd nerve palsy is extensive: vascular, traumatic, infectious, inflammatory, compressive, migraines, and neurodegenerative disorders. A propensity to migraines has been shown to be a risk factor for development of a 3rd nerve palsy in a large national Taiwan cohort with a hazard ratio of 2.67.4 The presentation is determined by the location of the lesion (Table 1). The locations of the lesions were noted to be subarachnoid (32%), cavernous sinus (23%), brainstem (14%) and non-localized (18%).2 Brainstem lesions may present with isolated effect on portions of the nuclei, but often several sub-nuclei are affected with associated neurologic signs. Bilateral ptosis is often associated with an ophthalmoplegia, given the levator nuclei midline location. A lesion on one side of the nucleus may cause an ipsilateral effect on the inferior rectus and inferior oblique but the contralateral superior rectus. Nuclear lesions are generally ischemic, but neuronal degeneration such as amyotrophic lateral sclerosis could be seen as a cause as well.
Fascicular lesions usually present with a total ophthalmoplegia and pupil involvement. Lesions may be ischemic, infiltrative, degenerative, compressive. Several syndromes have been described with fascicular lesions. Nothnagel syndrome has cerebellar ataxia associated with a unilateral 3rd nerve palsy. Benedikt syndrome has associated involuntary movements such as hemichorea, hemiballismus, and tremors. Weber syndrome demonstrates a contralateral hemiplegia.
As the 3rd nerve passes from the mesencephalon to the cavernous sinus, the nerve passes between the superior cerebellar and the posterior cerebral arteries in the subarachnoid space. Isolated unilateral pupillary dilation may occur from an aneurysm of these vessels at the junction of the internal carotid and posterior communicating arteries or with a basilar artery aneurysm. The pupillary fibers are in the periphery of the nerve and subject to an external compressive force. Lesions presenting with isolated pupillary involvement do evolve to a partial or complete ophthalmoplegia. Isolated pupillary dilation is a rare occurrence usually due to a ciliary ganglion lesion or pharmacologic dilation. Infiltrative lesions in the subarachnoid space usually have an associated ophthalmoplegia. As the 3rd nerve enters the cavernous sinus, it is fixed and subject to stretching and damage from compressive lesions such as tumors, aneurysms, or uncal herniation.
Cavernous sinus and superior orbital fissure lesions have similar presentations. The oculomotor, abducens, and trochlear nerves are classically involved with the ophthalmic division of the trigeminal nerve. With a lesion in the cavernous sinus, the maxillary division of the trigeminal nerve may also be involved. Due to the trigeminal nerve involvement, a painful ophthalmoplegia is common. This may be associated with proptosis, lid edema, and chemosis. Parasympathetic or sympathetic innervation may be compromised. The pupil may be dilated, mid-dilated, or small but poorly reactive.
A fixed dilated pupil and 3rd nerve palsy with other neurologic deficits is associated with a ventral midbrain lesion. An isolated 3rd nerve palsy with a dilated pupil can be associated with a lesion from the interpeduncular fossa to the ciliary ganglion. These include basal aneurysm, supratentorial lesions, granulomatous meningitis, parasellar tumor, and parasellar inflammation. Pupil-sparing 3rd nerve lesions are seen with ischemic lesions such as diabetes, hypertension, or arteriosclerotic disease. Rare reports of pupil involvement have been seen with diabetic 3rd nerve palsy. (Table 2)
|
Table 1. Etiology of Pediatric Third Nerve Palsy
|
|
Etiology
|
Ng5
|
Biglan6
|
Keane (adult)
|
|
Congenital
|
33%
|
38%
|
|
|
Trauma
|
28%
|
32%
|
26%
|
|
Tumor
|
22%
|
11%
|
12%
|
|
Vascular
|
11%
|
11%
|
18%
|
|
Infection
|
6%
|
|
5%
|
|
Diabetes
|
|
|
11%
|
|
Demyelinating
|
|
|
5%
|
|
Surgical
|
|
|
10%
|
|
Table 2. Common Third Nerve Palsy Characteristics
|
|
Condition
|
Location
|
Pupil
|
Pain
|
Other
|
|
Congenital
|
|
+/-
|
No
|
Aberrant regeneration
|
|
Aneurysm
|
Brainstem
Cavernous Sinus
|
+
|
+(94%)
|
|
|
Diabetes, HTN
|
|
NO
|
+/- (69%)
|
Often resolves spontaneously
|
|
|
Brainstem
Fascicular
|
+
|
NO
|
Bilat Ptosis, unilateral III; Contralateral superior rectus
|
|
|
Cavernous Sinus
Superior fissure
|
+/-
|
+ Often with 5th nerve
|
Multiple cranial nerves
|
Management
In the pediatric patient, 3rd nerve palsy usually is congenital, traumatic (surgical), or caused by a tumor or aneurysm. Ophthalmoplegic migraine may occur with a family history of migraines. Although any cause is possible, an ischemic cause is rare in the pediatric population. In the adult, ischemic causes are the most common if the condition is pupil sparing. Medical evaluation and imaging is required to determine the cause. An aneurysm is a surgical emergency. Of interest, treating the aneurysm with a coil or surgical clipping may result in complete or partial third nerve recovery in over 80% of patients.7,8
An ophthalmic assessment can determine the extent of the motility limitation, ptosis, and pupillary status. Restrictive strabismus can mimic a partial 3rd nerve palsy and require assessment of forced ductions and generations. With a total 3rd nerve palsy, the 4th nerve may be difficult to assess. The examiner needs to have the patient attempt infraduction and adduction to look for intorsion. The slit lamp may assist in looking for intorsion using a conjunctival or iris vessel as a marker.
A divisional branch of the 3rd nerve may be involved with an aneurysm or orbital disease. The superior branch is most commonly involved in adults with a picture of an acquired monocular elevation deficit with ptosis. With a complex congenital ptosis, this should be considered in children. An inferior branch palsy is most likely associated with orbital pathology. Aberrant regeneration or innervation is commonly seen with congenital 3rd and traumatic 3rd nerve palsy. Ischemic causes generally do not demonstrate aberrant regeneration. Anomalous eye movements can occur with adduction, elevation, or depression. Reported anomalies include pupil, lid, and gaze dyskinesis. The regeneration is random.
Once the systemic evaluation is addressed and the patient is stable, it is recommended to wait at least 6 months for resolution. The patient needs to be examined periodically by then, and if some improvement is seen, further recovery could be seen up to 1 year post incident. With a complete ptosis, diplopia is not an issue but may require occlusion if the ptosis is partial. In children, amblyopia needs to be addressed with patching and lid elevation with tape or a glasses crutch. Light sensitivity with a dilated pupil may be controlled with pilocarpine drops. Botulinum toxin has been used in the lateral rectus with limited usefulness in the short term.
Surgical Management
Surgical correction is a challenge. Aberrant regeneration can particularly complicate the treatment plan. Counselling the patient as to expectations is critical. Especially in complete palsies, the problem cannot be fixed in its entirety and residual diplopia is common. The approach will depend upon the residual deficit and if partial or complete. The patient may present with vertical, horizontal, or torsional components with a varying degree of ptosis. Partial horizontal deficits may respond to simple horizontal rectus surgery on the involved eye or on the contralateral eye. Subsequent exotropic shifts have been addressed with a lateral rectus disinsertion with periosteal fixation.1 In another approach Gokyigit2 achieved good primary alignment in 5 of 10 patients with a medial transposition of a split lateral rectus muscle under the vertical muscles. For complete 3rd nerve palsy, in 1933 Peter described fracturing the trochlea and shortening and transposing the superior oblique.3 Scott4 described disinserting and shortening the superior oblique tendon without trochlear disinsertion and suturing it to the medial rectus. This eliminates the superior oblique adduction effect but patients have developed vertical deviations with residual exotropia. Less residual vertical and horizontal deviation was reported with superior oblique surgery in combination with a lateral rectus and medial rectus surgery.5
Another approach has been to disinsert the lateral rectus in combination with a large medial rectus resection or a globe anchoring procedure. The lateral rectus is disinserted and attached to the periosteum of the lateral orbital wall. The medial rectus resection is large (8 to 12 mm) with or without an adjustable suture. Another option is use of a permanent anchor material (periosteal flap,6 silicone band,7 superior oblique tendon, fascial lata,8 5-0 polyester,9 etc.) to tether the medial rectus insertion to the retrocanalicular or posterior medial orbital wall. Use of a titanium T-plate has also been described to anchor material more securely to the orbital wall.10
Surgical correction may give a satisfactory cosmetic outcome, but persistent diplopia is still a significant issue. The field of single binocular function tends to be limited. An “occluder” contact lens or prism has been of value postoperatively.
References
- Porter JD, Baker RS. Anatomy and Embryology of the Ocular Motor System. Chapter 25, In Miller NR, Newman NJ (ed): Clinical Neuro-Ophthalmology: Volume One. Philadelphia PA: Williams and Wilkins 1982.
- Lyons CJ, Godoy F, Al Qahtani E. Cranial Nerve Palsies in Childhood. Eye 2015; 29(2):246-251.
- Keane JR. Third Nerve Palsy: Analysis of 1400 personally-examined patients. Can J Neurol Sci. 2010; 37(5):662-670.
- Yang CP, Chen YT, Fuh JL, Wang SJ. Migraine and Risk of Ocular Motor Cranial Nerve Palsies: A Nationwide Cohort Study. Ophthalmol. 2016; 123:191-197.
- Nt Y, Lyons CJ. Oculomotor Nerve Palsy in Childhood. Can J Ophthalmol. 2005;40(5):645-653.
- Schumacher-Ferro LA, Yoo KW, Solari FM, Biglan AW. Third Cranial Nerve Palsy in Children. Am J Ophthalmol. 1999;128(2):216-221.
- Gaberel T. Borha A, di Plama C, Emery E. Clipping versus Coiling in the Management of posterior Communicating Artery Aneurysm with Third Nerve Palsy: a Systematic Review and Meta-Analysis. World Neurosurg. 2015 Sep 25 pii:S1878-8750.
- Englehardt J, Berge J, Curr E, Parchet. Oculomotor Nerve Palsy Induced by an Internal Carotid Aneurysm: Prognostic Factors for Recovery. Acta Neurochir. 2015;157(7):1103-1111.
- Morad Y, Kowal L, Scott AB. Lateral Rectus Muscle Disinsertion and Reattachment to the Lateral Orbital Wall. Br J Ophthalmol. 2005; 89:983-985.
- Gokigit B, Akar S, Santan B, et al. Medial Transposition of a Split Lateral Rectus Muscle for Complete Oculomotor Nerve Palsy. J AAPOS. 2013; 17:402-410.
- Peter LC. The Use of the Superior Oblique as an Internal Rotator in Third Nerve Paralysis. Trans Amer Ophthalmol Society. 1933; 31:232-237.
- Scott AB. Transposition of the Superior Oblique. Am Orthoptic J 1977; 27:11-14.
- Eraslan M, Cerman E, Omal S, Ogut MS. Superior Oblique Anterior Transposition with Horizontal Recti Recession-Resection for Total Third-Nerve Palsy. J Ophthalmol. 2015; 2015:1-8. 780139 E pub.
- Goldberg RA, Rosenbaum AL, Tong JT. Use of Apically Based Periosteal Flaps as Globe Tethers in Severe Paretic Strabismus. Arch Ophthalmol. 2000; 37: 431-437.
- Bicas HE. A Surgically Implanted Elastic Band to Restore Paralyzed Ocular Rotations. J Pediatr Ophthalmol Strabismus. 1991; 28:10-13.
- Salazar-Leon JA, Ramirez-Ortiz MA, Salas-Vargas. The Surgical Correction of Paralytic Strabismus using Fascia Lata. J Pediatr Ophthalmol Strabismus. 1998; 35:27-32.
- Yonghong J, Kanxing Z, Wei L, et al. Surgical Management of Large Angle Incomitant Strabismus with Oculomotor Nerve Palsy. J AAPOS. 2008; 12:49-53.
- Tse DT, Shriver EM, Krantz KB, et al. The Use of Titanium T-plate as a platform for globe alignment in severe paralytic and restrictive strabismus. Am J Ophthalmol. 2010; 150:404-411.