By Jesse L. Berry, MD; Liya Xu, PhD; A. Linn Murphree, MD; Subramanian Krishnan, PhD; Kevin Stachelek, BS; Thomas C. Lee, MD; Anders Carlsson, PhD; Peter Kuhn, PhD; Jonathan W. Kim, MD; David Cobrinik, MD, PhD; James Hicks, PhD
Retinoblastoma (Rb) is a primary intraocular cancer that develops in the eyes of young children most often under the age of 5 years.1 Despite advances in chemotherapy delivery, nearly 50% of advanced eyes worldwide (Groups D and E) have required enucleation, the surgical removal of the eye.2-7 Even with treatment, many affected eyes are legally blind due to the effects of the tumor and/or the treatment on the retina.8-11 Currently, aside from the size and location of the tumor and the presence of seeding (seeds are small foci of active tumor cells floating in the vitreous or subretinal space), there are no known prognostic features that are effective for predicting the response of retinoblastoma tumors to treatment or determining which eyes will ultimately require secondary enucleation due to multiple tumor recurrences during and after chemotherapy.12,13 Further, currently there are no targeted treatments or personalized medicine yet available for this childhood cancer, despite the fact that it has a known genetic etiology for tumorigenesis. This is because biopsy of the intraocular tumor is contraindicated due to the risk of extraocular tumor spread and metastatic disease.14-21 Thus, the only time tumor is available is after an eye is enucleated.
Research on Rb tumor tissue from enucleated (ie, surgically removed) eyes has provided a wealth of information on tumorigenesis in Rb. However, because the eyes have been enucleated, this knowledge cannot be correlated with clinical outcomes such as tumor response to therapy or tumor recurrence requiring secondary enucleation. It is well established that development of Rb is initiated by a mutation in the RB1 tumor suppressor gene, the first described tumor suppressor gene, which is located on chromosome 13q in 98% of cases; the remaining cases are thought to be initiated by MYCN amplification on chromosome 2p.22-28 RB1 mutant retinoblastoma has two genetic forms: non-heritable and heritable. The non-heritable form (60% of cases) has biallelic somatic mutations in the tumor, while the heritable form (40% of cases) has an RB1 mutation in all (germline) or most (mosaic) cells of the body and a second somatic mutation in the tumor(s).29-31 Tumor cells also harbor recurrent secondary genomic changes that may contribute to tumorigenesis. In Rb, these are often restricted to gains and losses of small regions of chromosomes (called copy number variations, CNVs).32-37 Thus, while a genetic marker of retinoblastoma, mutation analysis, other tumor markers, and gene expression profiling can be done on Rb tumor cells, none of these had been associated with clinical outcomes, because of the long-standing "golden rule" that during treatment, the eye is inviolable and the intraocular space cannot be safely accessed.38-39
This rule changed in 2012 when Francis Munier introduced a safety-enhanced procedure to inject a chemotherapeutic agent, melphalan, directly into the vitreous cavity of eyes with Rb seeding.40,41 An initial step in this procedure is a paracentesis to withdraw aqueous humor (AH) from the anterior chamber, a separate compartment of the eye from where the tumor forms (Figure 1). This is done in order to lower the intraocular pressure prior to injection and prevent seeds from refluxing to the pars plana injection site. This procedure has been deemed safe, with no cases of metastatic disease reported.42-49 In a ten-center retrospective cohort study, with an accumulated 3565 injections in 656 patients, there were no events of extraocular tumor that could be associated with prior intravitreal chemotherapy injections.50 Aside from providing a new treatment strategy for seeding, for the first time, the procedure enabled safe access to the intraocular space and specifically to the AH in Rb eyes.
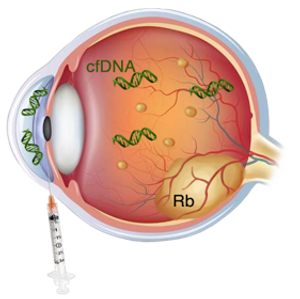
A.
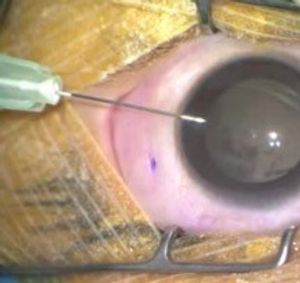
B.
Figure 1. AH extraction (A) cartoon rendering and (B) clinical image
Access to the AH of eyes with active Rb has enabled the first-ever genetic analyses of Rb tumors in situ (ie, without enucleation). This is the first time that retinoblastoma tumor DNA has been available from eyes before they were enucleated. Our initial genetic analysis of AH DNA revealed the presence of tumor-derived cell-free DNA (cfDNA) with CNVs that correspond to chromosomal changes in the tumor. This suggests that the AH has the potential to be used to characterize tumors in situ and possibly to prognosticate therapeutic response and, in the future, may even be used to guide therapy.51
This is the current clinical situation in the care of retinoblastoma patients: two children present with exactly the same demographics and clinical features—including the same age, laterality, same tumor features—and the treating ocular oncologist will not know whether this is an eye that will respond to chemotherapy treatment with more than 50% certainty.5,7,12 Finally, analysis of the AH may finally provide information to help decide which eyes are likely to be salvaged and which are higher risk and less likely to respond to therapy. For example, it is known that primary amplification of MYCN portends a worse clinical prognosis, and it has been recommended that these eyes should be enucleated primarily and should not undergo any attempted salvage therapy.22,32 While this claim is supported by the pathological features seen post enucleation in these eyes, there is currently no way to assay for MYCN amplification before enucleation, because tumor DNA is needed to test for this amplification. Thus, evaluation of the tumor-derived DNA present in the AH of retinoblastoma eyes may finally lead to a better understanding of how certain chromosomal variations (CNVs), focal MYCN amplifications, and mutations in the RB1 tumor suppressor gene lend themselves to secondary changes that promote tumorigenesis in retinoblastoma. For the first time, we have the ability to evaluate the use of AH as a surrogate tumor biopsy for retinoblastoma.
Preliminary studies to validate the AH as a surrogate biopsy
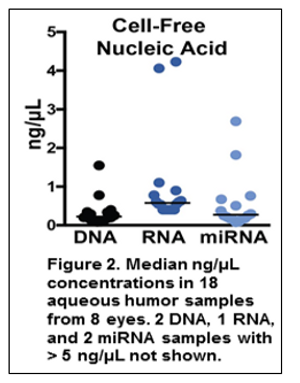
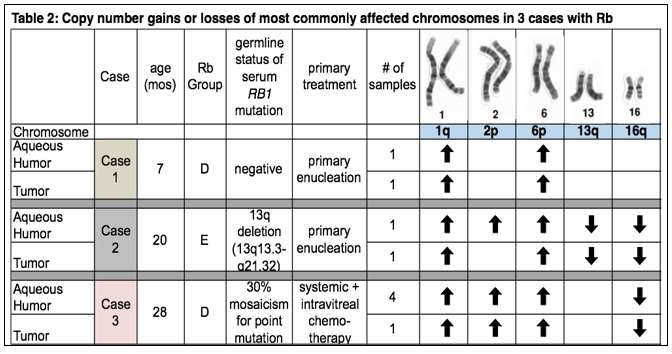
A study was done to determine that there were measurable levels of nucleic acids in the small volume (0.1 ml) of AH that can be extracted from an Rb eye and that the DNA in the AH was derived from the tumor by using CNV profiling. We analyzed 18 AH samples from 8 patients with DNA, RNA, and miRNA. Patient demographics are shown in Table 1. Nucleic concentrations were assayed using Qubit® High Sensitivity Assay Kits (Thermo Fisher). All samples of 0.1 ml of AH had DNA, RNA, and miRNA concentrations above 0.1 ng/µL (at least 10x the lower limit of detection) (Figure 2). After quantifying nucleic acid concentrations in the AH, we determined whether the DNA was tumor-derived using chromosomal CNV profiling of the AH from two primarily enucleated eyes and sequential AH cell-free DNA (cfDNA) obtained from an eye undergoing intravitreal melphalan injections for treatment of tumor seeding. Circulating cfDNA was isolated and sequencing library protocols were optimized to retain cfDNA from the AH samples from Rb patients. Shallow whole genome sequencing was performed on the Illumina® platform, followed by genome-wide copy number variation profiling. Shallow whole genome sequencing of AH cfDNA from the three patients revealed CNV profiles that would not be associated with normal cellular DNA. These include gain of 1q, 2p, 6p and loss of 13q and 16q consistent with known highly recurrent CNVs in Rb tumors.32,34,36,37,52-55 Furthermore, these recurrent CNVs, as well as other gains and losses, were observed in both the tumor and the AH (Table 2). This is described in more detail below.
Table 1: Demographics in a study analyzing 18 AH samples from 8 retinoblastoma patients
|
Case
|
Sex
|
Age Diag. (mos)
|
Laterality
|
Rb Grp
|
Serum RB1 test
|
Initial Tx
|
Time of AH Sample
|
|
1
|
M
|
7
|
unilat
|
D2
|
negative
|
ENUC
|
ENUC
|
|
2
|
M
|
20
|
unilat
|
E
|
13q-
|
ENUC
|
ENUC
|
|
3
|
F
|
28
|
bilat
|
D2
|
30% mosaic c. 1075A>T
|
CEV
|
IVM
|
|
4
|
M
|
26
|
unilat
|
D1
|
negative
|
CEV
|
IVM
|
|
5
|
M
|
5
|
unilat
|
D1
|
negative
|
CEV
|
IVM
|
|
6
|
M
|
0
|
bilat
|
B
|
g.76932_76952del21
|
CEV
|
IVM
|
|
7
|
F
|
10
|
unilat
|
D2
|
negative
|
CEV
|
IVM
|
|
8
|
F
|
59
|
unilat
|
D1
|
negative
|
CEV
|
IVM
|
|
ENUC=enucleation (surgical removal of eye); CEV=carboplatin, etoposide, vincristine chemotherapy; IVM=intravitreal injection of melphalan
|
Methods
Surgical Procedure: A paracentesis with the extraction of 0.1 µl of AH is done with a 32-gauge needle via the clear cornea pre-intravitreal injection or post enucleation as a treatment for primary or recurrent Rb.40,41 (Figure 1) The AH is then stored at -80C in tuberculin syringes.
CfDNA Isolation and Purification: AH plasma is separated by centrifugation followed by extraction of cfDNA with the QIAamp circulating nucleic acid kit (Qiagen). DNA is extracted by QIAamp MinElute Virus Spin Kit (Qiagen) to recycle all nucleic acid from AH.
Library Construction: DNA libraries are constructed with QIAseq Ultralow Input Library Kit (Qiagen). Each library is constructed with a sample barcode (New England Biolabs, Ipswich, MA) to permit pooling of multiple samples on a single Illumina® HiSeq lane.
Next Generation Sequencing (NGS): DNA libraries are sequenced on the Illumina® HiSeq platform for single-end 50 base pair (bp) protocol.
Bioinformatics Analysis: Briefly, this involves three steps: first, deconvoluting the sequence reads; second, mapping the reads to the human genome (hg19, Genome Reference Consortium GRCh37, UCSC Genome Browser database https://genome.ucsc.edu/), and removing PCR duplicates; and third, normalizing for guanine-cytosine (GC) content and estimating copy number using the CBS segmentation algorithm. The CNV profiles are based on 5,000 variable length genome bins, averaging a length of 150 kilo-base pairs each, and are calculated as a ratio to normal.65
Copy Number Variation Analysis: Hierarchical clustering is performed using the heatmap2 function in the gplots package in R (https://cran.r-project.org/web/packages/gplots/index.html). Using Ward’s method66-68 for the Euclidean distance metric and median-centered data, the heatmap is created and colored red for chromosomal gains and blue for losses. Thresholds of 0.8 and 1.25 relative to the median are used to define deletions and amplifications, respectively.
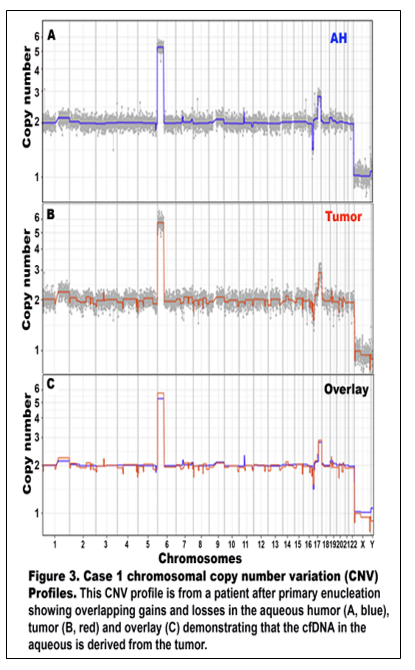
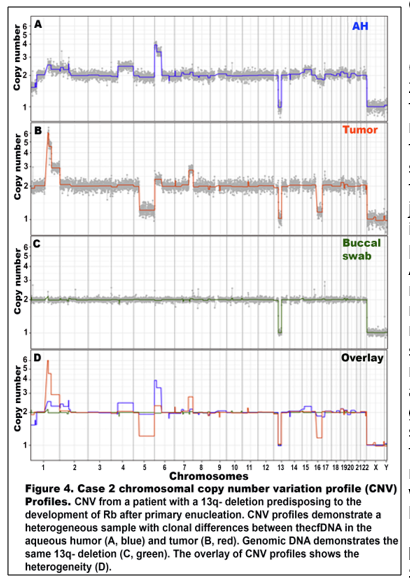
Figures 3 and 4 show CNV profiles in the AH and tumor of the two patients after primary nucleation. Interestingly, Case 2 has a germline 13q deletion causing pre-disposition to develop Rb. In this case, not all CNVs were present in either the AH or the tumor. This suggests multiclonality of the tumors with a heterogeneous mix of tumor-derived cfDNA from the tumor clones represented in the AH.


Figure 5 displays CNV profiles of sequential AH biopsies for a patient undergoing intravitreal melphalan injection. The profiles were consistent over time, albeit with minor changes in 4q and 7q gains in a subset of samples. Figure 6 shows that CNV profiles summed from the 6 AH samples were similar to the aggregated profiles of 71 Rb tumors.35 This further indicates that recurrent chromosomal changes can be seen in the AH cfDNA.51
Innovation: This study was the first to evaluate AH from Rb eyes undergoing salvage therapy with intravitreal injection of melphalan, the first to demonstrate that there are quantifiable levels of nucleic acids in the AH of treated and untreated Rb eyes, and the first to demonstrate that tumor-derived cell-free DNA is present in the AH of eyes with Rb. These results suggest that AH can serve as a "surrogate tumor biopsy" when tumor tissue is not directly available, as is always the case in eyes with retinoblastoma that are undergoing salvage treatment. While we have confirmed the presence of cell-free tumor-derived DNA in advanced retinoblastoma eyes, we believe that the AH holds even more potential to serve as a surrogate biopsy. We hypothesize that similar to breast cancer,56-58 leukemia,59,60 lung cancer,61 and prostate cancer,62,63 specific genetic or genomic changes that can be assayed from the AH will be informative regarding therapeutic response and/or prognostication for patients with retinoblastoma.
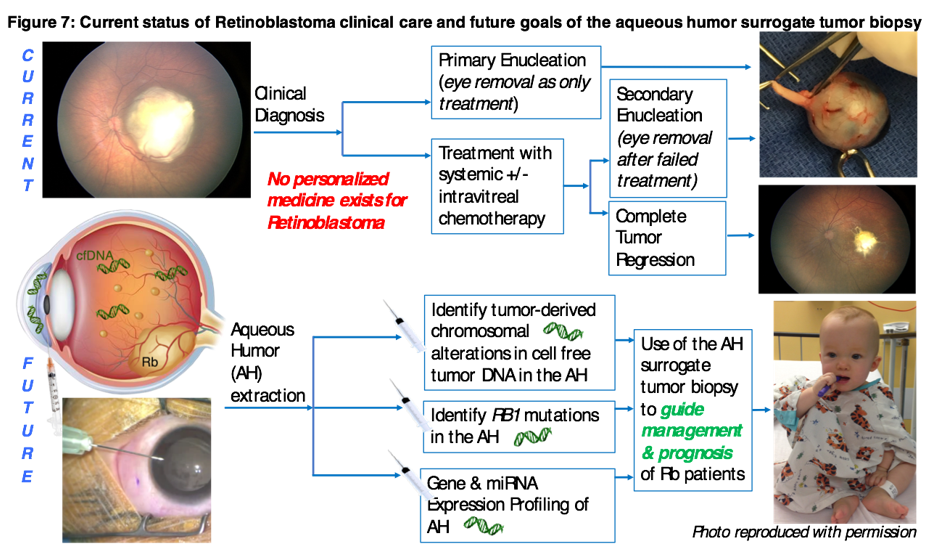
Direct tumor biopsy is not possible for retinoblastoma, due to risk of extraocular tumor spread, which could cause mortality to the child. For years, this limited the ability to assay genetic information from the tumor before the eye was enucleated. This prevented researchers and ocular oncologists from developing any true diagnostic or prognostic assays for retinoblastoma based on the tumor profile, and currently any form of personalized medicine for the care of Rb patients is lacking. While the AH has shown tremendous potential as a surrogate to tumor biopsy, significant further research is required to establish this as a diagnostic and prognostic test or one that can guide personalized, precision medicine for these children. For example, future research must focus on developing and optimizing the use of the AH in three significant ways: identifying chromosomal profiles with recurrent CNVs, isolating RB1 mutations, and evaluating expression profiles from the AH. However, the critical piece is the ability to correlate these findings with clinical tumor outcomes, including response to therapy or tumor recurrence after therapy (Figure 7).
Most critically, we must determine whether there are corroborative patterns of CNV profiles, expression profiles, or particular mutations that portend response to therapy for retinoblastoma patients. Because the AH can safely be extracted from eyes undergoing active therapy, this will, for the first time, enable the correlation of genomic information in real time with the treatment regimen rather than after enucleation (when tumor tissue is finally available). Ultimately, this information will provide insights into tumor cell heterogeneity,34 retinoblastoma progression states,37 candidate genes for tumorigenesis36 and treatment responses.7,64 It holds the potential to unlock a new era of precision medicine that will transform the way we detect Rb, guide the management and prognosis for this childhood cancer, and lead to alternative treatment regimens that offer a personalized approach to medicine for these children with a greater chance of saving the eye.
Please see the interview with Dr. Berry at https://www.aao.org/interview/aqueous-humor-biopsy-retinoblastoma.
References
- Harbour JW, editor. Retinoblastoma: pathogenesis and diagnosis. Philadelphia: B.C. Decker; 2001.
- Scelfo C, Francis JH, Khetan V, et al. An international survey of classification and treatment choices for group D retinoblastoma. Int J Ophthalmol. 2017;10(6):961-967. doi: 10.18240/ijo.2017.06.20. PubMed PMID: 28730089; PMCID: PMC5515152.
- Fabian ID, Stacey AW, Johnson KP, et al. Primary intravenous chemotherapy for group D retinoblastoma: a 13-year retrospective analysis. Br J Ophthalmol. 2017;101(1):82-88. Epub 2016/12/15. doi: 10.1136/bjophthalmol-2016-309710. PubMed PMID: 27965263.
- Fabian ID, Stacey AW, Johnson KC, et al. Primary enucleation for group D retinoblastoma in the era of systemic and targeted chemotherapy: the price of retaining an eye. Br J Ophthalmol. 2017. Epub 2017/07/01. doi: 10.1136/bjophthalmol-2017-310624. PubMed PMID: 28659391.
- Shields CL, Ramasubramanian A, Thangappan A, et al. Chemoreduction for group E retinoblastoma: comparison of chemoreduction alone versus chemoreduction plus low-dose external radiotherapy in 76 eyes. Ophthalmology. 2009;116(3):544-551 e1. Epub 2009/01/23. doi: 10.1016/j.ophtha.2008.10.014. PubMed PMID: 19157557.
- Abramson DH, Daniels AB, Marr BP, et al. Intra-Arterial Chemotherapy (Ophthalmic Artery Chemosurgery) for Group D Retinoblastoma. PLoS One. 2016;11(1):e0146582. Epub 2016/01/13. doi: 10.1371/journal.pone.0146582. PubMed PMID: 26756643; PMCID: PMC4710506.
- Berry JL, Jubran R, Kim JW, et al. Long-term outcomes of Group D eyes in bilateral retinoblastoma patients treated with chemoreduction and low-dose IMRT salvage. Pediatr Blood Cancer. 2013;60(4):688-693. Epub 2012/09/22. doi: 10.1002/pbc.24303. PubMed PMID: 22997170.
- Narang S, Mashayekhi A, Rudich D, Shields CL. Predictors of long-term visual outcome after chemoreduction for management of intraocular retinoblastoma. Clin Exp Ophthalmol. 2012;40(7):736-742. Epub 2012/02/04. doi: 10.1111/j.1442-9071.2012.02757.x. PubMed PMID: 22300311.
- Berry JL, Jubran R, Wong K, Lee TC, Murphree AL, Kim JW. Factors predictive of long-term visual outcomes of Group D eyes treated with chemoreduction and low-dose IMRT salvage: the Children's Hospital Los Angeles experience. Br J Ophthalmol. 2014;98(8):1061-1065. doi: 10.1136/bjophthalmol-2013-304411. PubMed PMID: 24671926.
- Tsimpida M, Thompson DA, Liasis A, et al. Visual outcomes following intraophthalmic artery melphalan for patients with refractory retinoblastoma and age appropriate vision. Br J Ophthalmol. 2013;97(11):1464-1470. Epub 2013/09/17. doi: 10.1136/bjophthalmol-2013-303694. PubMed PMID: 24037611.
- Fabian ID, Naeem Z, Stacey AW, et al. Long-term Visual Acuity, Strabismus, and Nystagmus Outcomes Following Multimodality Treatment in Group D Retinoblastoma Eyes. Am J Ophthalmol. 2017;179:137-144. Epub 2017/05/16. doi: 10.1016/j.ajo.2017.05.003. PubMed PMID: 28501391.
- Shields CL, Mashayekhi A, Au AK, et al. The International Classification of Retinoblastoma predicts chemoreduction success. Ophthalmology. 2006;113(12):2276-2280. doi: 10.1016/j.ophtha.2006.06.018. PubMed PMID: 16996605.
- Kaliki S, Shields CL, Rojanaporn D, et al. High-risk retinoblastoma based on international classification of retinoblastoma: analysis of 519 enucleated eyes. Ophthalmology. 2013;120(5):997-1003. doi: 10.1016/j.ophtha.2012.10.044. PubMed PMID: 23399379.
- Karcioglu ZA. Fine needle aspiration biopsy (FNAB) for retinoblastoma. Retina. 2002;22(6):707-710. PubMed PMID: 12476095.
- Karcioglu ZA, Gordon RA, Karcioglu GL. Tumor seeding in ocular fine needle aspiration biopsy. Ophthalmology. 1985;92(12):1763-1767. PubMed PMID: 4088631.
- Andrew JM, Smith DR. Unsuspected retinoblastoma. Am J Ophthalmol. 1965;60(3):536-540. Epub 1965/09/01. PubMed PMID: 5294147.
- Spaulding AG, Naumann G. Unsuspected retinoblastoma. Enucleation in an 11-year-old girl following injury and panophthalmitis. Arch Ophthalmol. 1966;76(4):575-577. Epub 1966/10/01. PubMed PMID: 5928145.
- Shields CL, Honavar S, Shields JA, Demirci H, Meadows AT. Vitrectomy in eyes with unsuspected retinoblastoma. Ophthalmology. 2000;107(12):2250-2255. Epub 2000/11/30. PubMed PMID: 11097606.
- Murthy R, Honavar SG, Vemuganti GK, Naik MN, Reddy VP. Systemic metastasis following hyphema drainage in an unsuspected retinoblastoma. J Pediatr Ophthalmol Strabismus. 2007;44(2):120-123. Epub 2007/04/07. PubMed PMID: 17410964.
- Biswas J, Das D, Vaijayanthi P, Khetan V, Kumar SK. Clinicopathological Study of a Case of Unsuspected Retinoblastoma. J Pediatr Ophthalmol Strabismus. 2009. Epub 2009/10/02. doi: 10.3928/01913913-20090918-04. PubMed PMID: 19791715.
- Chawla B, Hada M, Seth R, et al. Trabeculectomy in eyes with unsuspected retinoblastoma. Ophthalmic Genet. 2016;37(4):437-440. Epub 2016/11/03. doi: 10.3109/13816810.2015.1126610. PubMed PMID: 26966836.
- Rushlow DE, Mol BM, Kennett JY, et al. Characterisation of retinoblastomas without RB1 mutations: genomic, gene expression, and clinical studies. Lancet Oncol. 2013;14(4):327-334. Epub 2013/03/19. doi: 10.1016/S1470-2045(13)70045-7. PubMed PMID: 23498719.
- Li WB, Buckley J; Sanchez-Lara, PA, et al. A Rapid and Sensitive Next Generation Sequencing Method to Detect RB1 Mutations Improves Care for Retinoblastoma Patients and Their Families. J Mol Diagn. 2016. Epub In Press.
- Murphree AL, Benedict WF. Retinoblastoma: clues to human oncogenesis. Science. 1984;223(4640):1028-1033. PubMed PMID: 6320372.
- Sparkes RS, Murphree AL, Lingua RW, et al. Gene for hereditary retinoblastoma assigned to human chromosome 13 by linkage to esterase D. Science. 1983;219(4587):971-973. PubMed PMID: 6823558.
- Benedict WF, Murphree AL, Banerjee A, Spina CA, Sparkes MC, Sparkes RS. Patient with 13 chromosome deletion: evidence that the retinoblastoma gene is a recessive cancer gene. Science. 1983;219(4587):973-975. PubMed PMID: 6336308.
- Fung YK, Murphree AL, T'Ang A, Qian J, Hinrichs SH, Benedict WF. Structural evidence for the authenticity of the human retinoblastoma gene. Science. 1987;236(4809):1657-1661. PubMed PMID: 2885916.
- Benedict WF, Fung YK, Murphree AL. The gene responsible for the development of retinoblastoma and osteosarcoma. Cancer. 1988;62(8 Suppl):1691-1694. PubMed PMID: 3167788.
- Dimaras H, Corson TW, Cobrinik D, et al. Retinoblastoma. Nat Rev Dis Primers. 2015;In Press.
- Knudson AG Jr, Hethcote HW, Brown BW. Mutation and childhood cancer: a probabilistic model for the incidence of retinoblastoma. Proc Natl Acad Sci U S A. 1975;72(12):5116-5120. PubMed PMID: 1061095; PMCID: PMC388887.
- Knudson A. Retinoblastoma: teacher of cancer biology and medicine. PLoS Med. 2005;2(10):e349. doi: 10.1371/journal.pmed.0020349. PubMed PMID: 16231982; PMCID: PMC1261516.
- Theriault BL, Dimaras H, Gallie BL, Corson TW. The genomic landscape of retinoblastoma: a review. Clin Exp Ophthalmol. 2014;42(1):33-52. doi: 10.1111/ceo.12132. PubMed PMID: 24433356; PMCID: PMC3896868.
- Villegas VM, Gold AS, Wildner A, Ehlies F, Murray TG. Genomic landscape of retinoblastoma. Clin Exp Ophthalmol. 2014;42(1):2-3. doi: 10.1111/ceo.12277. PubMed PMID: 24433353.
- Kooi IE, Mol BM, Moll AC, et al. Loss of photoreceptorness and gain of genomic alterations in retinoblastoma reveal tumor progression. EBioMedicine. 2015;2(7):660-670. doi: 10.1016/j.ebiom.2015.06.022. PubMed PMID: 26288838; PMCID: PMC4534696.
- Kooi IE, Mol BM, Massink MP, et al. Somatic genomic alterations in retinoblastoma beyond RB1 are rare and limited to copy number changes. Sci Rep. 2016;6:25264. doi: 10.1038/srep25264. PubMed PMID: 27126562; PMCID: PMC4850475.
- Kooi IE, Mol BM, Massink MP, et al. A Meta-Analysis of Retinoblastoma Copy Numbers Refines the List of Possible Driver Genes Involved in Tumor Progression. PLoS One. 2016;11(4):e0153323. doi: 10.1371/journal.pone.0153323. PubMed PMID: 27115612; PMCID: PMC4846005.
- Cobrinik D. Retinoblastoma Progression. EBioMedicine. 2015;2(7):623-624. doi: 10.1016/j.ebiom.2015.07.023. PubMed PMID: 26288828; PMCID: PMC4534706.
- Dyer MA, Bremner R. The search for the retinoblastoma cell of origin. Nat Rev Cancer. 2005;5(2):91-101. Epub 2005/02/03. doi: 10.1038/nrc1545. PubMed PMID: 15685194.
- Mendoza PR, Geisert EE, Ziesel AC, et al. Gene Expression Profiling of Retinoblastoma and Retinocytoma. Investigative Ophthalmology & Visual Science 2015;56:3444.
- Munier FL, Soliman S, Moulin AP, Gaillard MC, Balmer A, Beck-Popovic M. Profiling safety of intravitreal injections for retinoblastoma using an anti-reflux procedure and sterilisation of the needle track. Br J Ophthalmol. 2012;96(8):1084-1087. doi: 10.1136/bjophthalmol-2011-301016. PubMed PMID: 22368262.
- Munier FL, Gaillard MC, Balmer A, et al. Intravitreal chemotherapy for vitreous disease in retinoblastoma revisited: from prohibition to conditional indications. Br J Ophthalmol. 2012;96(8):1078-1083. Epub 2012/06/15. doi: 10.1136/bjophthalmol-2011-301450. PubMed PMID: 22694968.
- Ghassemi F, Shields CL. Intravitreal melphalan for refractory or recurrent vitreous seeding from retinoblastoma. Arch Ophthalmol. 2012;130(10):1268-1271. doi: 10.1001/archophthalmol.2012.1983. PubMed PMID: 23044940.
- Ghassemi F, Shields CL, Ghadimi H, Khodabandeh A, Roohipoor R. Combined intravitreal melphalan and topotecan for refractory or recurrent vitreous seeding from retinoblastoma. JAMA Ophthalmol. 2014;132(8):936-941. doi: 10.1001/jamaophthalmol.2014.414. PubMed PMID: 24789622.
- Shields CL, Manjandavida FP, Arepalli S, Kaliki S, Lally SE, Shields JA. Intravitreal melphalan for persistent or recurrent retinoblastoma vitreous seeds: preliminary results. JAMA Ophthalmol. 2014;132(3):319-325. Epub 2014/01/11. doi: 10.1001/jamaophthalmol.2013.7666. PubMed PMID: 24407202.
- Tuncer S, Balci Ö, Tanyildiz B, Kebudi R, Shields CL. Intravitreal Lower-Dose (20 microg) Melphalan for Persistent or Recurrent Retinoblastoma Vitreous Seeds. Ophthalmic Surg Lasers Imaging Retina. 2015;46(9):942-948. doi: 10.3928/23258160-20151008-07. PubMed PMID: 26469234.
- Lawson BM, Saktanasate J, Say EA, Shields CL. Intravitreal chemotherapy provides control for massive vitreous seeding from retinoblastoma. J Pediatr Ophthalmol Strabismus. 2014;51 Online:e92-4. doi: 10.3928/01913913-20141203-05. PubMed PMID: 25490242.
- Manjandavida FP, Shields CL. The role of intravitreal chemotherapy for retinoblastoma. Indian J Ophthalmol. 2015;63(2):141-145. doi: 10.4103/0301-4738.154390. PubMed PMID: 25827545; PMCID: PMC4399123.
- Suzuki S, Aihara Y, Fujiwara M, Sano S, Kaneko A. Intravitreal injection of melphalan for intraocular retinoblastoma. Jpn J Ophthalmol. 2015;59(3):164-172. doi: 10.1007/s10384-015-0378-0. PubMed PMID: 25808017.
- Smith SJ, Smith BD. Evaluating the risk of extraocular tumour spread following intravitreal injection therapy for retinoblastoma: a systematic review. Br J Ophthalmol. 2013;97(10):1231-6. doi: 10.1136/bjophthalmol-2013-303188. PubMed PMID: 23740960.
- Francis JH, Abramson DH, Ji XD, et al. Risk of Extraocular Extension in Retinoblastoma Eyes Receiving Intravitreous Chemotherapy. JAMA Ophthalmol. 2017 Nov 2. doi: 10.1001/jamaophthalmol.2017.4600. [Epub ahead of print].
- Berry JL, Xu L, Murphree A, et al. Potential of Aqueous Humor as a Surrogate Biopsy for Retinoblastoma. JAMA Ophthalmol. 2017; 135:1221-1230. Epub November 1, 2017. doi: 10.1001/jamaophthalmol.2017.4097.
- Kooi IE, van Mil SE, MacPherson D, et al. Genomic landscape of retinoblastoma in Rb-/- p130-/- mice resembles human retinoblastoma. Genes Chromosomes Cancer. 2016. doi: 10.1002/gcc.22429. PubMed PMID: 27750399.
- Paderova J, Orlic-Milacic M, Yoshimoto M, da Cunha Santos G, Gallie B, Squire JA. Novel 6p rearrangements and recurrent translocation breakpoints in retinoblastoma cell lines identified by spectral karyotyping and mBAND analyses. Cancer Genet Cytogenet. 2007;179(2):102-111. doi: 10.1016/j.cancergencyto.2007.08.014. PubMed PMID: 18036396.
- Grasemann C, Gratias S, Stephan H, et al. Gains and overexpression identify DEK and E2F3 as targets of chromosome 6p gains in retinoblastoma. Oncogene. 2005;24(42):6441-6449. doi: 10.1038/sj.onc.1208792. PubMed PMID: 16007192.
- Cano J, Oliveros O, Yunis E. Phenotype variants, malignancy, and additional copies of 6p in retinoblastoma. Cancer Genet Cytogenet. 1994;76(2):112-115. PubMed PMID: 7923058.
- Pongor L, Kormos M, Hatzis C, Pusztai L, Szabó A, Györffy B. A genome-wide approach to link genotype to clinical outcome by utilizing next generation sequencing and gene chip data of 6,697 breast cancer patients. Genome Med. 2015;7:104. doi: 10.1186/s13073-015-0228-1. PubMed PMID: 26474971; PMCID: PMC4609150.
- Hicks J, Krasnitz A, Lakshmi B, et al. Novel patterns of genome rearrangement and their association with survival in breast cancer. Genome Res. 2006;16(12):1465-1479. Epub 2006/12/05. doi: 10.1101/gr.5460106. PubMed PMID: 17142309; PMCID: PMC1665631.
- Navin N, Krasnitz A, Rodgers L, Cook K, Meth J, Kendall J, Riggs M, Eberling Y, Troge J, Grubor V, Levy D, Lundin P, Maner S, Zetterberg A, Hicks J, Wigler M. Inferring tumor progression from genomic heterogeneity. Genome Res. 2010;20(1):68-80. Epub 2009/11/12. doi: 10.1101/gr.099622.109. PubMed PMID: 19903760; PMCID: PMC2798832.
- Rigolin GM, Saccenti E, Bassi C, et al. Extensive next-generation sequencing analysis in chronic lymphocytic leukemia at diagnosis: clinical and biological correlations. J Hematol Oncol. 2016;9(1):88. doi: 10.1186/s13045-016-0320-z. PubMed PMID: 27633522; PMCID: PMC5025606.
- Kriangkum J, Motz SN, Mack T, et al. Single-Cell Analysis and Next-Generation Immuno-Sequencing Show That Multiple Clones Persist in Patients with Chronic Lymphocytic Leukemia. PLoS One. 2015;10(9):e0137232. doi: 10.1371/journal.pone.0137232. PubMed PMID: 26353109; PMCID: PMC4564241.
- DiBardino DM, Saqi A, Elvin JA, et al. Yield and Clinical Utility of Next-Generation Sequencing in Selected Patients With Lung Adenocarcinoma. Clin Lung Cancer. 2016;17(6):517-522 e3. doi: 10.1016/j.cllc.2016.05.017. PubMed PMID: 27378171.
- Spans L, Clinckemalie L, Helsen C, et al. The genomic landscape of prostate cancer. Int J Mol Sci. 2013;14(6):10822-10851. doi: 10.3390/ijms140610822. PubMed PMID: 23708091; PMCID: PMC3709705.
- Dago AE, Stepansky A, Carlsson A, et al. Rapid phenotypic and genomic change in response to therapeutic pressure in prostate cancer inferred by high content analysis of single circulating tumor cells. PLoS One. 2014;9(8):e101777. Epub 2014/08/02. doi: 10.1371/journal.pone.0101777. PubMed PMID: 25084170; PMCID: PMC4118839.
- Berry JL, Shah S, Bechtold M, Zolfaghari E, Jubran R, Kim JW. Long-term outcomes of Group D retinoblastoma eyes during the intravitreal melphalan era. Pediatr Blood Cancer. 2017. doi: 10.1002/pbc.26696. PubMed PMID: 28646513.
- Baslan T, Kendall J, Ward B, et al. Optimizing sparse sequencing of single cells for highly multiplex copy number profiling. Genome Res. 2015;25(5):714-724. Epub 2015/04/11. doi: 10.1101/gr.188060.114. PubMed PMID: 25858951; PMCID: PMC4417119.
- Strauss T, von Maltitz MJ. Generalising Ward's Method for Use with Manhattan Distances. PLoS One. 2017;12(1):e0168288. doi: 10.1371/journal.pone.0168288. PubMed PMID: 28085891; PMCID: PMC5235383.
- Varin T, Bureau R, Mueller C, Willett P. Clustering files of chemical structures using the Szekely-Rizzo generalization of Ward's method. J Mol Graph Model. 2009;28(2):187-195. doi: 10.1016/j.jmgm.2009.06.006. PubMed PMID: 19640752.
- Ward J. Hierarchical Grouping to Optimize an Objective Function. Journal of the American Statistical Association. 1963;58:236-244.