The modern classification of childhood glaucoma began with the term buphthalmos, or ox-eyed, which was used to describe the secondary effect of elevated intraocular pressure (IOP) on the elastic infantile eye.1 Buphthalmos was subdivided into simple buphthalmos due to a primary mechanism, now known as primary congenital glaucoma, and buphthalmos associated with other developmental anomalies. The classification evolved into the more complex anatomic classification of Hoskins, which divided the developmental abnormalities into trabecular meshwork, iris, and/or cornea.2 The Shaffer-Weiss classification introduced the categories of isolated congenital (infantile) glaucoma, glaucomas associated with congenital anomalies, and acquired glaucoma.3 Walton proposed further additions and modifications to the Shaffer-Weiss classification system with an exhaustive listing of all disorders known to be associated with childhood glaucoma.4
Efforts to classify childhood glaucoma have been dogged by overlapping and variably defined nomenclature, which frequently denotes an age of onset rather than the underlying mechanism for the glaucoma. The term congenital glaucoma has been used to denote primary congenital glaucoma that occurs at or shortly after birth or glaucoma of any etiology that occurs in the same time frame. Primary congenital glaucoma has been used to refer only to cases that present at or shortly after birth, with primary infantile glaucoma used for the genetically identical condition that presents 1–2 months later. The term juvenile glaucoma has been similarly confused with the more specific term juvenile open-angle glaucoma. The onset of glaucoma following congenital cataract surgery has been termed aphakic glaucoma, yet many of these children have had a primary or secondary intraocular lens placed. Developmental glaucoma has been used as a giant waste-basket term for nearly all the childhood glaucomas that are not acquired after birth. Studies of childhood glaucoma have been hampered by inconsistent classification efforts and the rarity of many of the associated disorders.
The adult definition of glaucoma has focused on the pathology of the optic nerve to make the diagnosis. Defining the onset of glaucoma in children is more difficult because the optic nerve can be difficult or impossible to evaluate properly due to corneal opacity. Furthermore, infants and young children develop ocular enlargement with the onset of glaucoma, as noted by Duke-Elder.1 The difficulties of diagnosing glaucoma in children have led some to use elevated IOP as a surrogate for the diagnosis, however, it has been demonstrated that children can develop the onset of elevated IOP without developing evidence of optic-nerve damage, termed ocular hypertension or glaucoma suspect in adult terminology.5,6 The Infant Aphakia Treatment Study has demonstrated the importance of standardized definitions of glaucoma suspect and glaucoma and the high risk of children developing ocular hypertension and glaucoma following congenital cataract surgery.5
The Childhood Glaucoma Research Network (CGRN), composed of clinicians and scientists who specialize in treating children with glaucoma, played a pivotal role in developing a new international classification system for childhood glaucoma. The following definitions of childhood glaucoma and glaucoma suspect, and a new childhood classification system represent the end result of an international collaboration on childhood glaucoma for the World Glaucoma Association (WGA) meeting in July 2013 in Vancouver, British Columbia.7 The definition of childhood glaucoma is IOP-related damage to the eye, rather than being based solely on optic-nerve criteria. Occasionally, this definition can be met only after multiple evaluations when progressive changes are established. The classification covers glaucoma occurring at any point in childhood, which has various international definitions. For each patient, the disease classification can evolve with ongoing systemic evaluation as secondary causes are uncovered, and a child with bilateral glaucoma can carry a different diagnosis for each eye. An algorithm for classifying a patient with childhood glaucoma using the new system is presented in Figure 1.
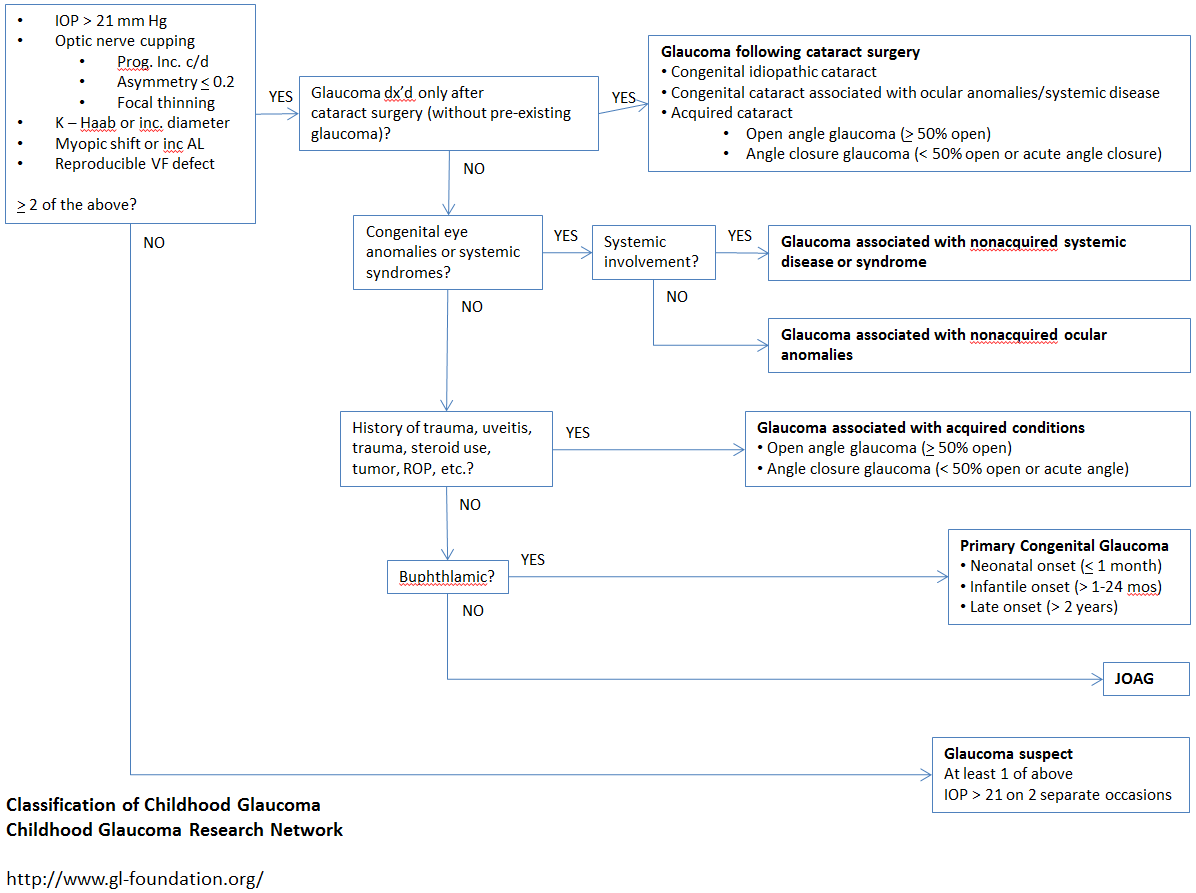
Figure 1. Childhood Glaucoma Research Network/World Glaucoma Association algorithm for the classification of childhood glaucoma.
Definitions7
- Childhood age is based on national criteria:
- US: younger than 18 years
- EU, UK, UNICEF: 16 years or younger
- Glaucoma: IOP-related damage to the eye; at least 2 criteria required for diagnosis:
- IOP > 21 mm Hg; investigator discretion if exam under anesthesia data alone due to variable effects of anesthesia on all methods of IOP assessment
- Optic disc cupping: progressive increase in cup-disc ratio, cup-disc asymmetry of ≥ 0.2 when the optic discs are similar size, or focal rim thinning
- Corneal findings: Haab striae or diameter ≥ 11 mm in newborn, > 12 mm in child < 1 year, or > 13 mm any age
- Progressive myopia, myopic shift, or an increase in ocular dimensions out of keeping with normal growth
- Reproducible visual-field defect consistent with glaucomatous optic neuropathy with no other observable reason for defect
- Glaucoma suspect: no IOP-related damage and at least 1 of the following criteria:
- IOP > 21 mm Hg on 2 separate occasions
- Suspicious optic disc appearance for glaucoma, i.e., increased cup-disc ratio for size of optic disc
- Suspicious visual field for glaucoma
- Increased corneal diameter or axial length in setting of normal IOP
International Classification (CGRN/WGA) of Childhood Glaucoma7
Primary childhood glaucoma:
- Primary congenital glaucoma (PCG) (Figure 2)
- Isolated angle anomalies, +/-mild congenital iris anomalies
- Meets glaucoma definition, usually with ocular enlargement
- Subcategories based on age at onset:
- Neonatal or newborn: 0–1 month
- Infantile: > 1–24 months
- Late-onset or late-recognized: > 2 years
- Spontaneously arrested cases with normal IOP, but typical signs of PCG, can be classified as PCG.
- Juvenile open-angle glaucoma (JOAG) (Figure 3)
- No ocular enlargement
- No congenital ocular anomalies or syndromes
- Open angle, normal appearance
- Meets glaucoma definition
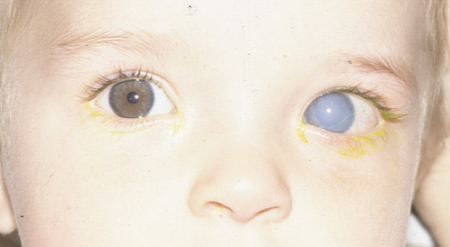
Figure 2. Unilateral primary congenital glaucoma demonstrating corneal hydrops OS. Beck AD, Lynch MG, Focal Points: Pediatric Glaucoma, American Academy of Ophthalmology, June 1997.
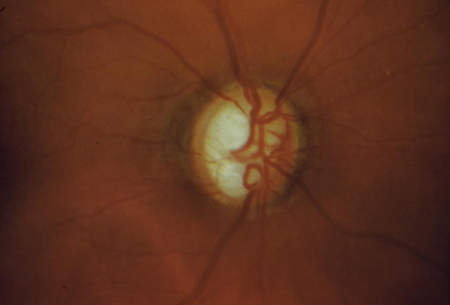
Figure 3. Anterior segment exam of child with juvenile open-angle glaucoma is normal. Diagnosis made by documentation of glaucomatous optic-nerve changes combined with elevated IOP.
Secondary childhood glaucoma:
- Glaucoma associated with nonacquired ocular anomalies (Figure 4)
- Includes conditions of predominantly ocular anomalies present at birth that might or might not be associated with systemic signs
- Meets glaucoma definition
- List common ocular anomalies (Table 1)
- Glaucoma associated with nonacquired systemic anomalies (Figure 5)
- Includes conditions predominantly of systemic disease present at birth that might be associated with ocular signs
- Meets glaucoma definition
- List common systemic syndrome or disease (Table 2)
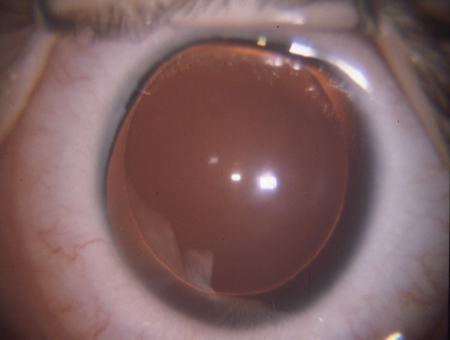
Figure 4. Glaucoma is frequently associated with aniridia, a nonacquired ocular condition.
Table 1. Glaucoma associated with nonacquired ocular anomalies.7
|
Conditions with predominantly ocular anomalies present at birth that might or might not be associated with systemic signs:
- Axenfeld Rieger anomaly (syndrome if systemic associations)
- Peters anomaly (Syndrome if systemic associations)
- Ectropion uveae
- Congenital iris hypoplasia
- Aniridia
- Persistent fetal vasculature/PFV (if glaucoma present before cataract surgery)
- Oculodermal melanocytosis (nevus of Ota)
- Posterior polymorphous dystrophy
- Microphthalmos
- Microcornea
- Ectopia lentis
- Simple ectopia lentis (no systemic associations, possible Type 1 FBN mutation)
- Ectopia lentis et pupillae
|
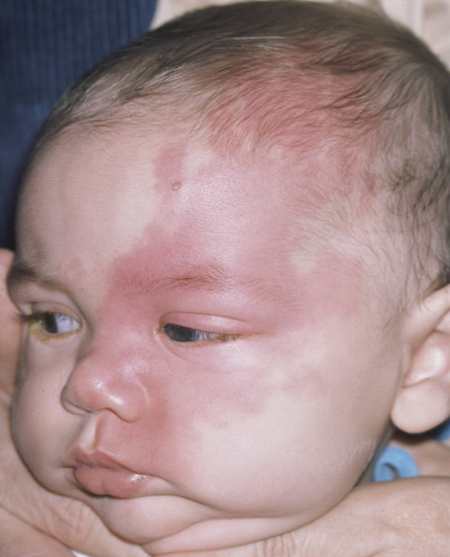
Figure 5. The 2 essential features of Sturge Weber syndrome are a facial cutaneous angioma (port-wine nevus) and a leptomeningeal angioma. Unilateral childhood glaucoma is commonly associated with this nonacquired systemic syndrome.
Table 2. Glaucoma associated with nonacquired systemic disease or syndrome.7
|
Conditions with predominantly known syndromes, systemic anomalies or systemic disease present at birth that might be associated with ocular signs
- Chromosomal disorders such as Trisomy 21 (Down syndrome)
- Connective tissue disorders
- Marfan syndrome
- Weill-Marchesani syndrome
- Stickler syndrome
- Metabolic disorders
- Homocystinuria
- Lowe syndrome
- Mucopolysaccharidoses
- Phacomatoses
- Neurofibromatosis (NF-1, NF-2)
- Sturge-Weber syndrome
- Klippel-Trenaunay-Weber syndrome
- Rubinstein-Taybi
- Congenital rubella
|
Glaucoma associated with acquired condition (Figure 6)
- Meets glaucoma definition after acquired condition is recognized; an acquired condition is one that is not inherited or present at birth.
- Glaucoma developing after cataract surgery is excluded from this category to highlight its frequency and differences from other conditions in the acquired condition category.
- List common acquired conditions (Table 3)
- Based on gonioscopy results:
- Open-angle glaucoma: >/= 50% open or
- Angle-closure glaucoma: < 50% open or acute angle closure
- Glaucoma following cataract surgery (Figure 7)
- Meets glaucoma definition only after cataract surgery is performed and is subdivided into 3 categories:
- Congenital idiopathic cataract
- Congenital cataract associated with ocular anomalies/systemic disease, no previous glaucoma
- Acquired cataract, no previous glaucoma
- Based on gonioscopy results:
- Open-angle glaucoma: >/=50% open or
- Angle-closure glaucoma: <50% open or acute-angle closure
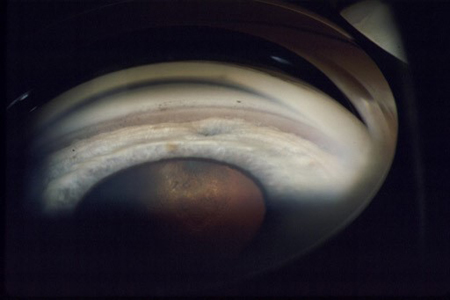
Figure 6. Trauma is an acquired condition that can lead to childhood glaucoma. The amount of angle recession correlates with the risk of developing glaucoma, as documented by a broadening of the ciliary body band and a deepening of the angle recess.
Table 3. Glaucoma associated with acquired condition7
- Uveitis
- Trauma (hyphema, angle recession, ectopia lentis)
- Steroid induced
- Tumors (benign/malignant, ocular/orbital)
- Retinopathy of Prematurity
- Prior ocular surgery other than cataract surgery
|
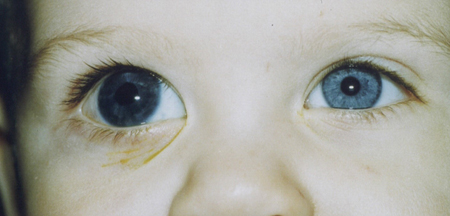
Figure 7. Glaucoma following cataract surgery OD demonstrating that ocular enlargement can occur with any glaucoma that presents in early childhood. Beck AD. Diagnosis and management of pediatric glaucoma. Lambert S, ed. Ophthalmology Clinics of North America: Pediatric Ophthalmology. Saunders: Philadelphia, 2001.
References
- Duke-Elder, Stewart. "Part 2. Congenital deformities". System of Ophthalmology. Vol. III. Duke-Elder, Stewart (ed.). London: Kimpton, 1964.
- Hoskins HD Jr, Shaffer RN, Hetherington J. Anatomical classification of the developmental glaucomas. Arch Ophthalmol. 1984 Sep;102(9):1331-6.
- Shaffer, R., Weiss, D. Congenital and Pediatric Glaucomas. Saint Louis: Mosby, 1970.
- Yeung HH, Walton DS. Clinical classification of childhood glaucomas. Arch Ophthalmol. 2010 Jun;128(6):680-4.
- Freedman SF, Lynn MJ, Beck AD, Bothun ED, Orge FH, Lambert SR. Glaucoma-related adverse events in the first five years after unilateral cataract removal in the Infant Aphakia Treatment Study. Arch Ophthalmol 2015, 2015 May 21.
- Gordon MO, Gao F, Beiser JA, Miller JP, Kass MA. The 10-year incidence of glaucoma among patients with treated and untreated ocular hypertension. Arch Ophthalmol. 2011 Dec;129(12):1630-1.
- Beck AD, Chang TCP, Freedman SF. "Definition, Classification, Differential Diagnosis." Childhood Glaucoma: Consensus Series 9. Weinreb RN et al. Amsterdam: Kugler, 2013.