The links to each individual chapter of the Color Atlas of Gonioscopy are available at the Chapters link, below.
Primary Open-Angle Glaucoma
Primary open-angle glaucoma is the most prevalent form of glaucoma. It is mentioned here only to note that there are no characteristic gonioscopic abnormalities in primary open-angle glaucoma, ocular hypertension, or normal-tension glaucoma.
Material Deposited in the Angle
Pigment
Pigment dispersion syndrome In the pigment dispersion syndrome there is an accumulation of pigment on structures throughout the anterior segment. The pigment originates from the iris pigment epithelium, which is abraded against the lens zonules (Campbell, 1979). As the pigment circulates throughout the anterior segment, it is deposited at many sites. The syndrome is most common in young myopic individuals. These patients are at increased risk for retinal detachment. Although the pigment dispersion syndrome is equally prevalent in both sexes, pigmentary glaucoma develops more commonly in males.
With the slit lamp, pigment deposited by aqueous convection currents can be seen as a vertical band on the corneal endothelium—the Krukenberg spindle (9‑1). The dusting of pigment on the surface of the iris (9‑2) can occasionally be sufficiently dense to cause heterochromia (9‑3). Characteristic spoke-like defects of the iris pigment epithelium are seen by transillumination (9‑4). These can be identified more easily with infrared transillumination when available.
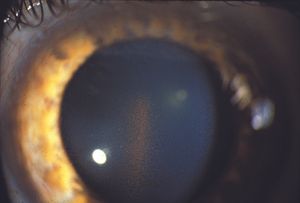
9-1 Krukenberg spindle in the pigment dispersion syndrome. Pigment released from the pigment epithelium of the posterior iris is deposited by convection currents on the corneal endothelium in a vertical band.
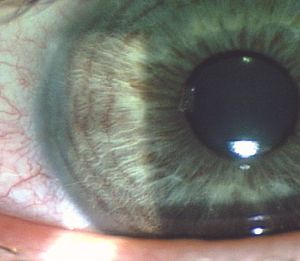
9-2 Pigment dusting on the iris associated with pigment dispersion syndrome.
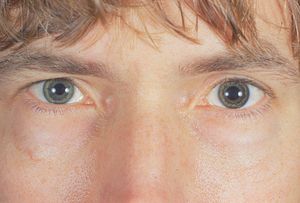
9-3 Iris heterochromia due to pigment dispersion in the left edge of a patient with the pigment dispersion syndrome. Also note that the left pupil is larger than the right: anisocoria can be seen in the pigment dispersion syndrome, the larger pupil generally being on the side with more transillumination defects. (Reprinted from American Journal of Ophthalmology, Vol 112, Haynes WL, Asymmetric Pigmentary Dispersion, pages 463– 464, copyright © 1991, with permission from Elsevier.)
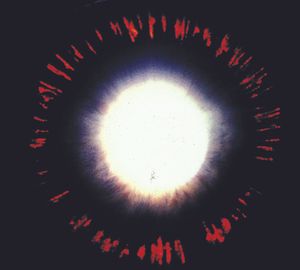
9-4 Characteristic spoke-like transillumination defects in the iris of a patient with the pigment dispersion syndrome. These defects are caused when the iris pigment epithelium is rubbed off by contact with packets of lens zonules.
Gonioscopic examination reveals dense black pigmentation of the trabecular meshwork and throughout the angle (9‑5). Pigment is frequently deposited anterior to Schwalbe’s line as a Sampaolesi’s line, which is a nonspecific sign of increased pigmentation. There may be a concavity of the mid-peripheral iris, which probably contributes to iris-zonule contact (9‑6). Peripheral iridotomy frequently resolves this iris configuration, although the role for iridotomy in the treatment of the condition is controversial. After dilation pigmentation may be seen where the zonules attach to the posterior lens capsule. Pigment in this area is called a Scheie’s stripe. Scheie’s stripe can occasionally be seen with the slit lamp (9‑7) but is best seen by gonioscopic examination (9‑8).
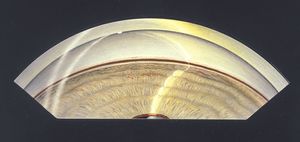
9-5 Patient with the pigment dispersion syndrome. The angle demonstrates a dense band of black pigment in the posterior trabecular meshwork.
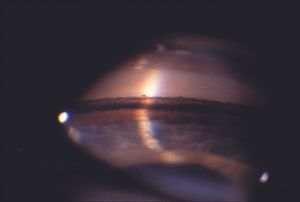
9-6 Marked posterior bowing in a patient with the pigment dispersion syndrome. Although a concave iris may be seen in the syndrome, this dramatic degree of concavity is unusual.
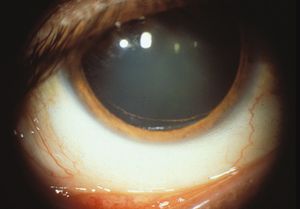
9-7 Scheie’s stripe—a band of pigment at the junction of zonules and posterior lens capsule in the pigment dispersion syndrome seen at the inferior border of the pupil. The stripe is usually seen only gonioscopically (arrow).

9-8 Scheie’s stripe in the pigment dispersion syndrome. Pigment accumulates at the junction of the zonules and the posterior lens capsule (arrow). The trabecular meshwork is moderately pigmented.
Pseudoexfoliation (exfoliation syndrome, glaucoma capsulare) In pseudoexfoliation there is a deposition of basement membrane-like material throughout the anterior segment of the eye. Similar material has been identified at many other sites throughout the bodies of patients with pseudoexfoliation (Streeten et al, 1992). There are conflicting reports as to whether there is any association between pseudoexfoliation and cardiovascular disease (Shrum et al, 2000, and Citirik et al, 2007). Eyes with pseudoexfoliation show an increased incidence of open-angle glaucoma and, occasionally, angle-closure glaucoma. This process can occur in one or both eyes and is a major cause of unilateral glaucoma. It occurs primarily among the elderly and is most prevalent among those of Scandinavian heritage. Certain haplotypes of the gene LOXL1 have been shown to be responsible for a large percentage of pseudoexfoliative glaucoma (Thorleifsson et al, 2007).
On examination a granular or flaky deposit is observed on the anterior lens capsule, often with a clear zone where the iris has rubbed some of the deposited material off the lens (9‑9). The edges of this material can curl at the pupillary border or can be seen there as dandruff-like debris. With transillumination patchy iris defects are often apparent at the pupillary border. Pigment may collect on the iris or corneal endothelium. The zonules are frosted with pseudoexfoliative material (9‑10). The zonules are fragile, making zonular dehiscence a risk in extracapsular cataract extraction (Skuta et al, 1987). Even after cataract surgery, the intraocular lens/capsular complex is prone to subluxation or dislocation (9‑11). In aphakic eyes pseudoexfoliative material may be seen on the ciliary processes (9‑12) or on the face of the vitreous body (9‑13).
Marked pigmentation of the angle is found on gonioscopic examination. The pigment typically has a more granular, brown character than the dense black pigment that is seen in the pigment dispersion syndrome (9‑14 to 9‑16).
Oculodermal melanocytosis In oculodermal melanocytosis there is abnormal pigmentation of the periorbital skin and globe. The involvement of the skin takes the form of a deep, dermal pigmentation in the ophthalmic and maxillary distributions of the trigeminal nerve. If only the eye is affected, the condition is termed “melanosis oculi” (9‑17). Ocular pigmentation most commonly affects the episclera but may also involve the iris, fundus, conjunctiva, and trabecular meshwork (9‑18). The disorder is more common in women than in men. It is seen most often in darkly pigmented individuals. Glaucoma has been reported in 10% of a large sample of patients with the condition (Teekhasaenee et al, 1990).
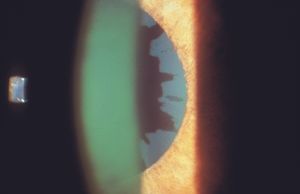
9-9 Slit-lamp photograph of pseudoexfoliative material on the anterior lens capsule. This material has a ground glass-like appearance and has areas that have been rubbed away by the motion of the iris against the lens capsule
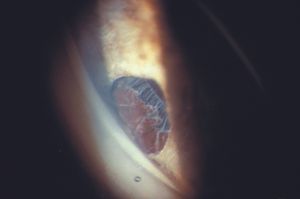
9-10 Gonioscopic view of pseudoexfoliative material on zonules. This view is through a peripheral iridectomy after a trabeculectomy. Note the frosted appearance of the zonules.

9-11 Subluxed IOL in patient with pseudoexfoliation
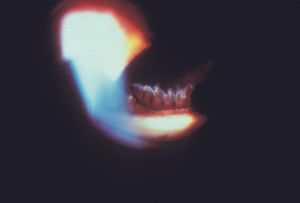
9-12 Gonioscopic photograph of pseudoexfoliative material on the ciliary body of an aphakic patient. (Courtesy of Richard K. Parrish II, MD, Bascom Palmer Eye Institute.)
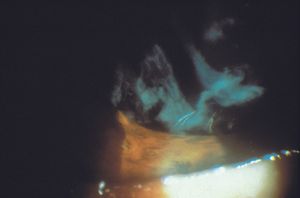
9-13 Pseudoexfoliation on the anterior vitreous face of an aphakic patient.
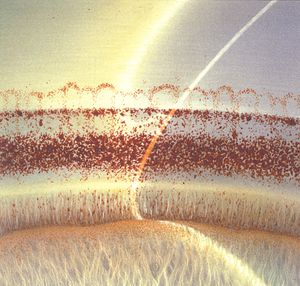
9-14 The angle in pseudoexfoliation. Note the clumped brown pigment over the pigmented trabecular meshwork. There is also a line of pigment along Schwalbe’s line and another, wavy line of pigment anterior to this line.

9-15 Pseudoexfoliation with a dense pigmentation of the angle that obscures most angle structures. The corneal wedge identifies Schwalbe’s line.
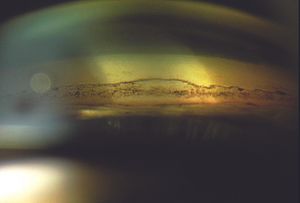
9-16 Gonioscopic view of the angle in pseudoexfoliation. There is extensive brown pigmentation of the angle, as well as additional angle pigmentation.
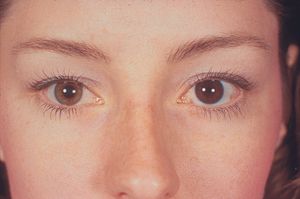
9-17 Heterochromia from melanosis oculi.
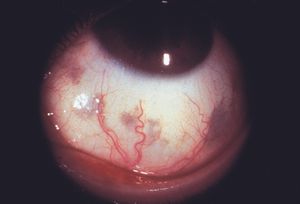
9-18 Deep slate-gray pigmentation of the episclera in a patient with melanosis oculi. There is also a dense pigmentation of the iris. (Same patient as 9‑17.)
Inspection of the angle of patients with glaucoma in oculodermal melanocytosis shows the trabecular meshwork to be covered by pigment or to be obscured by many pigmented iris processes (9‑19 and 9‑20). The ciliary body band is generally dark. The mechanism of glaucoma development is thought to be melanocytic infiltration of the trabecular meshwork and the dispersion of pigment. Evidence for the pigment dispersion mechanism is the finding of endothelial deposits of pigmentation with a Krukenberg spindle-like distribution (Teekhasaenee et al,1990).
Other causes of increased angle pigmentation There may be a release of pigment after surgery. This can occur after incisional surgery (9‑21) or after laser surgery. A malpositioned intraocular lens can rub against the posterior iris and release pigment into the angle. Peripheral laser iridotomy is a common cause of increased angle pigmentation, particularly of the inferior angle. Some patients with severe iridocyclitis may show a marked release of pigment into the anterior chamber (9‑22). With angle closure there can be pigmentation of the angle from chronic iris contact. Melanomalytic glaucoma results from the release of pigment by a large melanoma, usually of the choroid. This is a very uncommon cause of increased angle pigmentation.

9-19 Gonioscopic view of the eye of a patient with melanosis oculi. There are abundant iris processes and marked pigmentation within the angle. (Courtesy of Robert Ritch, MD, New York Eye and Ear Infirmary.)

9-20 Normal fellow eye of patient shown in 9‑21. (Courtesy of Robert Ritch, MD, New York Eye and Ear Infirmary.)
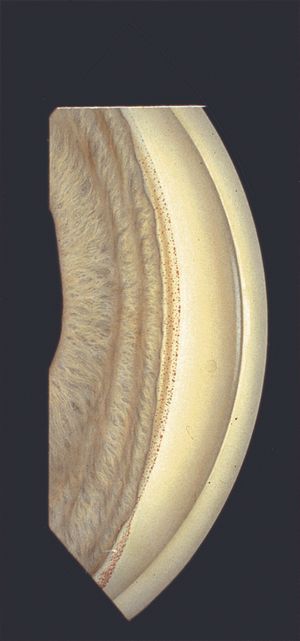
9-21 Gonioscopic view of temporal angle after cyclodialysis procedure, which is now rarely performed. Patients often have scattered pigmentation throughout the angle following various forms of intraocular surgery. The pigmentation is usually densest inferiorly.

9-22 Dense trabecular pigmentation of the superior angle in a patient following herpes zoster ophthalmicus.
Blood
Blood may be observed in the anterior chamber secondary to a range of etiologies, such as trauma, surgery, and neovascularization. Blood settles into the inferior angle under the influence of gravity. Small amounts of blood can be seen only by gonioscopy (9‑23 and 9‑24). Small hyphemas do not usually result in an increase in intraocular pressure. Large hyphemas are more likely to cause problems with intraocular pressure as the trabecular meshwork fills with blood. Very large hyphemas that extend into the posterior chamber can occlude the pupil and cause elevated pressure secondary to pupillary block. In some patients with recurring hyphemas—especially following surgery—gonioscopy can be used to locate bleeding vessels and to treat them with a laser. Small balls of pigment may be seen in the angle long after a hyphema has resolved (see 9‑50). It is important to remember that gonioscopy must be performed very gently when a hyphema is present, due to the risk of rebleeding early after a traumatic hyphema occurs. It is often best to defer gonioscopy to a later time to look for angle recession.
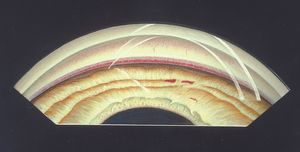
9-23 Gonioscopic view of inferior angle in a 16-year-old boy who was struck in the eye. There is blood lying on the trabecular meshwork.

9-24 A higher-magnification gonioscopic view of the inferior angle of the eye of a 17-year-old boy shortly after he was struck in the eye. There is a small amount of blood anterior to the trabecular meshwork and also in Schlemm’s canal.
Ghost-cell glaucoma is an unusual disorder that is seen in patients with vitreous hemorrhage. Red blood cells denature within the vitreous body and take on a spherical form, in contrast to the normal biconcave shape (9‑25). The cells are inflexible and pass through the trabecular meshwork with difficulty (Campbell et al, 1976). Such cells are more likely to raise intraocular pressure than red blood cells. Ghost erythrocytes may be identified by their characteristic khaki color on slitlamp (9‑26) and gonioscopic examination. Glaucoma is most likely to develop in aphakic eyes or in eyes with zonular breaks through which the ghost cells can gain access to the anterior chamber.
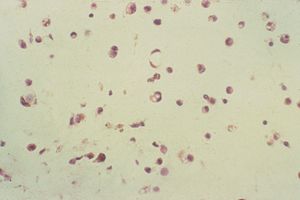
9-25 Ghost erythrocytes from the vitreous body of a patient with chronic intraocular hemorrhage. The cells are spherical with marginated denatured hemoglobin (arrow).
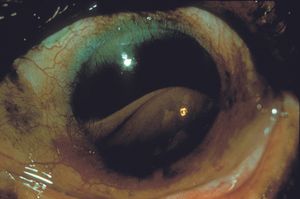
9-26 Ghost cells in the anterior chamber of an aphakic patient. Note the candy stripe of bright red cells within the degenerated khaki-colored cells.
Inflammation
In the inflamed eye one can see precipitates on the internal cornea and on the trabecular meshwork. Sometimes the precipitates are revealed only by gonioscopic examination (9‑27). A small hypopyon may be visible in the inferior angle with gonioscopy. Peripheral anterior synechiae are frequently observed after chronic inflammation as large precipitates consolidate (9‑28 and 9‑29) and cause the iris to become adherent to the trabecular meshwork. Closure of the angle by synechiae is discussed in Chapter 8.
Fuchs heterochromic iridocyclitis is a form of inflammatory glaucoma with a unique constellation of findings. Examination reveals frequent heterochromia (9‑30 and 9‑31), mild inflammation of the anterior chamber, diffuse fine stellate keratic precipitates (9‑32 and 9‑33), cataract (9‑34), and neovascularization of the iris (9‑35) and angle (9‑36 and 9‑37). These fine angle vessels do not often lead to the development of synechiae. The vessels are very fragile and can bleed on paracentesis of the anterior chamber (Amsler and Verrey, 1946) and even on gonioscopy (Begg, 1969). The most consistent feature of Fuchs heterochromic iridocyclitis is a flat, featureless iris (9‑31 and 9‑38) (Kimura et al, 1955). The disease appears to be associated with rubella virus, as intraocular antibodies to rubella virus have been isolated from the aqueous of patients with Fuchs heterochromic iridocyclitis (de Groot-Mijnes et al, 2006). Its prevalence has decreased in the United States since vaccination has become commonplace (Birnbaum, 2007).
Glaucomatocyclitic crisis is an uncommon, usually unilateral, mild inflammation accompanied by strikingly elevated intraocular pressures of 40 to 60 mm Hg. There are often a few small, colorless keratic precipitates over the inferior corneal endothelium. There may be slight pupillary dilation and iris hypochromia is occasionally observed on the affected side. The high pressure can cause corneal edema. The angle is open and has normal pigmentation. Synechiae do not develop (Posner and Schlossman, 1948). The sudden, strikingly high intraocular pressure rises can lead to a mistaken diagnosis of acute angle closure unless gonioscopy is performed.
A rare syndrome of precipitates on the trabecular meshwork was described by Chandler and Grant in 1965. This presents like primary open-angle glaucoma but demonstrates a paradoxical increase in intraocular pressure with cholinergic agents. On examination fine inflammatory precipitates are seen on the trabecular meshwork (as in 9‑27). The eye is otherwise generally quiet. The pressure usually responds to corticosteroids and aqueous suppressants, but recurrences are the rule and patients frequently require indefinite therapy. Some cases develop full-blown inflammation of the anterior chamber, while others never experience inflammation beyond the trabecular meshwork. Synechiae are a common development.

9-27 Gonioscopic view of an angle showing inflammatory precipitates (arrow) on the trabecular meshwork. (Reprinted with permission from R. Rand Allingham. Shields’ Textbook of Glaucoma. 2005. Courtesy of Lippincott Williams & Wilkins.)
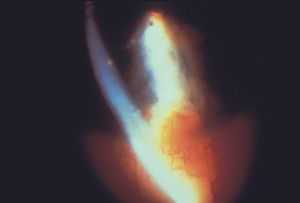
9-28 Slit-lamp photograph showing a large sarcoid granuloma in the inferior angle. This inflammatory mass can consolidate and pull the iris over the trabecular meshwork. (Reprinted from Clinical Atlas of Glaucoma, 1st ed, E. Michael Van Buskirk, copyright © 1986, with permission from Elsevier.)

9-29 Gonioscopic view of a sarcoid granuloma of the inferior angle. Same patient as in 9‑28.
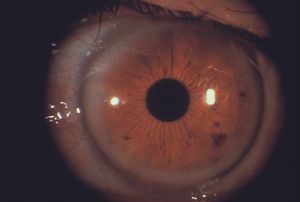
9-30 Normal right eye of a patient who has Fuchs heterochromic iridocyclitis in the left eye.
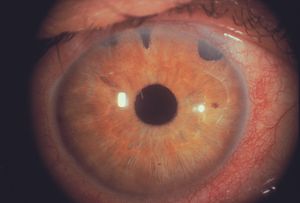
9-31 Left eye of the patient in 9‑30. This eye has Fuchs heterochromic iridocyclitis. Note the surgical iridectomies from filtration surgery and also the hypochromia of this iris when compared with the right eye. The iris is flat and featureless and lacks the normal crypts seen in the other eye.
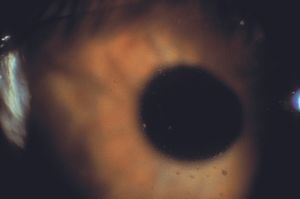
9-32 Same patient as 9‑30 and 9‑31. A closer view of the left eye demonstrates diffuse, small, keratic precipitates over the entire corneal endothelium.
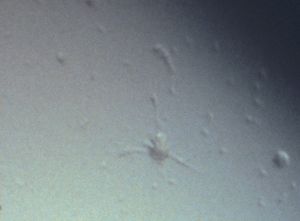
9-33 A example of a keratic precipitate in Fuchs heterochromic iridocyclitis. Note that the precipitate is clear with dendritic projections. (Reprinted with permission from Tasman W. Jaeger. Duane’s Clinical Ophthalmology, “Uveitic Glaucoma” by W. L. M. Alward. 1989. Courtesy of Lippincott Williams & Wilkins.)
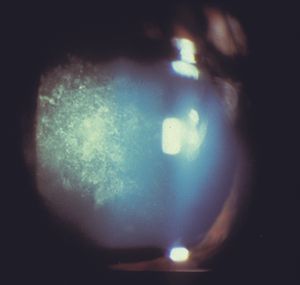
9-34 Posterior subcapsular cataract in Fuchs heterochromic iridocyclitis.
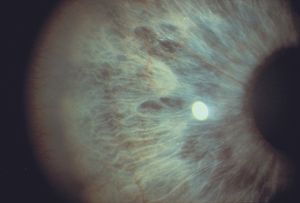
9-35 Diffuse neovascularization of the iris in Fuchs heterochromic iridocyclitis.

9-36 Neovascularization of the angle in Fuchs heterochromic iridocyclitis. These vessels are extremely fragile and bleed with minor trauma. Unlike other forms of neovascularization, the tendency to form synechiae is minimal.

9-37 More substantial neovascularization of the angle in Fuchs heterochromic iridocyclitis.

9-38 Gonioscopic view showing flat, featureless iris with neovascularization in Fuchs heterochromic iridocyclitis.
Lens material
When the lens is ruptured, the fragments may cause obstruction of the aqueous outflow. In lens-particle glaucoma, a fine glistening can be seen in the angle due to the presence of lens fragments (9‑39 and 9‑40). Fragments of lens are identified on tapping the anterior chamber.
Phacolytic glaucoma is seen in eyes with mature or hypermature cataracts (9‑41). The affected lenses leak denatured protein through an intact capsule into the anterior chamber. Macrophages engulf the material and are seen floating in the anterior chamber amid a heavy flare. White material observed on the anterior lens capsule probably represents collections of macrophages. The trabecular meshwork becomes obstructed by macrophages (9‑42 and 9‑43) and protein (Epstein et al, 1978). Pressure elevation is often abrupt and can reach very high levels. The eye is usually injected and the cornea is frequently edematous.
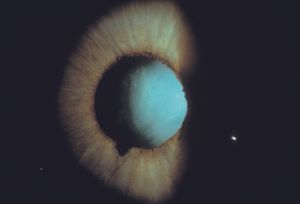
9-39 Patient with recent trauma demonstrating a tear of the sphincter. The patient had a marked elevation of his intraocular pressure.
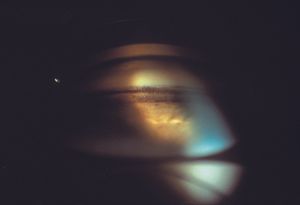
9-40 Gonioscopic view of the eye shown in 9‑39. The patient had glistening material on the surface of the trabecular meshwork. An anterior chamber tap demonstrated lens particles. Note that the angle is probably recessed.
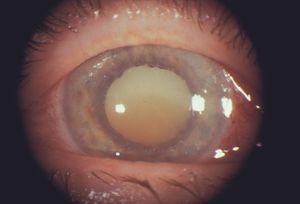
9-41 A hypermature cataract with a liquefied cortex and a brunescent nucleus that has sunk to the inferior portion of the lens.
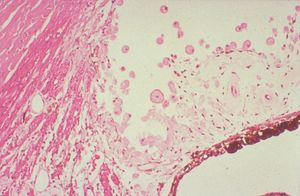
9-42 Phacolytic glaucoma in an eye with traumatic angle recession and a hypermature lens. Lens protein-filled macrophages are identified in the recessed angle. (Courtesy of the National Museum of Health and Medicine, Armed Forces Institute of Pathology.)
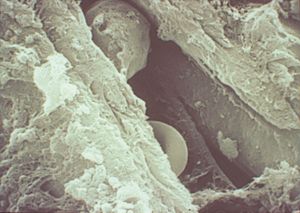
9-43 Scanning electron micrograph of intratrabecular space from an eye with phacolytic glaucoma. A red blood cell (white arrow) and a macrophage (black arrow) are seen between the trabecular pillars. Note the ruffled surface of the macrophage. (Courtesy of Robert Folberg, MD, University of Iowa.)
Foreign bodies
Foreign bodies can be found lodged in the anterior chamber angle after trauma (9‑44 and 9‑45). They are usually located in the inferior angle and may be found on routine examination. Most are inert and damage the eye only if they traumatize the corneal endothelium. If a patient presents with focal corneal edema, gonioscopy may reveal that a foreign body has lodged in the angle. When such bodies are removed, the edema usually clears (McDonald and Ashodian, 1959). Some metallic and vegetable foreign materials are poorly tolerated. Siderosis is a late effect of a retained foreign body composed of iron or steel. The iron oxidizes and ions are dispersed through the eye, resulting in toxicity to the trabecular meshwork, lens, cornea, and retina. The trabecular meshwork may take on a rusty hue (9‑46), and the eye may develop late glaucoma. The most striking slit-lamp finding is a rust-colored anterior cataract (9‑47). The most serious effects of siderosis arise from toxicity to the retina.
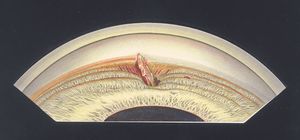
9-44 Glass in the inferior angle after trauma. The patient had broken his glasses while working in a sawmill. A fragment of glass was removed earlier. The patient presented with discomfort and injection. The chip of glass is wedged between the trabecular meshwork and the iris, distorting both structures. There is a small tear in the iris and clotted blood under the fragment. Some blood is present in Schlemm’s canal.
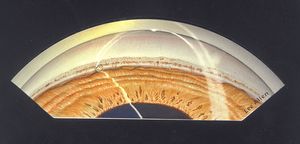
9-45 Sand in the inferior angle after a land-mine explosion.

9-46 The angle of a 28-year-old man who was struck in the eye by a metal clip from a concrete form. He sustained an intraocular iron foreign body. Note the rusty discoloration of the trabecular meshwork from siderosis.
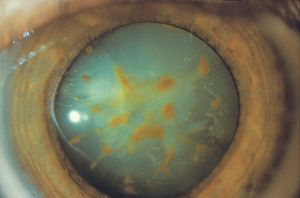
9-47 Cataract from siderosis in an eye with a retained iron foreign body.
Traumatic Angle Changes
Iridodialysis
Iridodialyses, or tears in the iris (9‑48 and 9‑49), occur as the result of blunt trauma. Bleeding from the tears can result in hyphemas. By themselves iridodialyses generally cause no problems. Occasionally there will be some flattening of the pupil in the quadrant of the tear. Some patients may notice extra images through the iris defect. An iridodialysis mostly serves as an indicator that the eye has previously sustained a substantial impact and should alert one to the possibility of angle recession or retinal damage. Some iridodialyses occur so far towards the periphery that they can be appreciated only by gonioscopy.
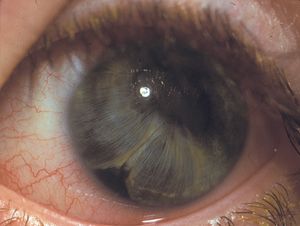
9-48 Inferior iridodialysis following trauma.
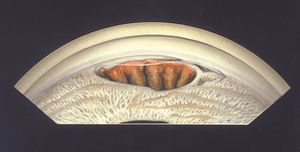
9-49 Gonioscopic view of traumatic iridodialysis in the inferior angle found on routine examination. The patient remembered having sustained blunt trauma to the eye several years previously. Ciliary processes can be seen through the iris defect.
Angle recession
A recessed angle occurs as a result of severe blunt trauma in which there is a tear in the face of the ciliary body, usually between the longitudinal and circular muscles. The tear can occur over a limited area or may involve the entire angle. Angle recession is important because of the accompanying increased risk of glaucoma. Glaucoma occurs in about 9% of patients with recessed angles and may develop months or years after the injury (Kaufman and Tolpin, 1974). Most patients with significant hyphemas will be found to have angle recession after the hemorrhage clears (Tönjum, 1966).
Gonioscopic examination shows a wide ciliary body band and a deep anterior chamber (9‑50 and 9‑51). The ciliary face may appear lighter in the recessed area because there is little ciliary tissue overlying the sclera (9‑50 and 9‑52). The presence of torn tissue may give the ciliary face a cobweb appearance, and the ciliary body face may have a linear defect suggesting that a cyclodialysis cleft had almost formed with the injury (9‑53). Iris processes will be broken and the scleral spur may appear whiter than normal because the uveal meshwork has been torn from the surface. It is useful to compare eyes to determine if one eye has an abnormally wide ciliary body band and/ or broken ciliary processes (9‑54 and 9‑55).
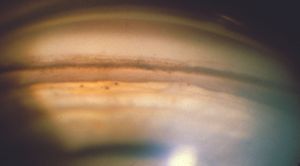
9-50 Angle of a 45-year-old man who was hit with a BB as a child. He later developed unilateral glaucoma from angle recession. Note the very deep anterior chamber and the large amount of ciliary body face visible below the scleral spur. The ciliary body band is a light color because it consists of only a thin layer of tissue over sclera. The tiny black balls on the iris are residuals from old hemorrhage.

9-51 Angle recession in an 11-year-old who was struck in the eye. Note the step-off below the scleral spur and the deep ciliary body face. There is blood in Schlemm’s canal.
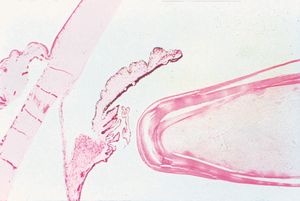
9-52 Histopathology of angle recession showing a tear in the anterior face of the ciliary body between the longitudinal and circular fibers (small arrow). The ciliary body is still attached to the scleral spur (large arrow). The space between sclera and the ciliary body (*) is an artifact. (Courtesy of Robert Folberg, MD, University of Iowa.)

9-53 Gonioscopic view of a patient with angle recession who has a linear defect in the ciliary body band suggestive of a cyclodialysis cleft. This patient had elevated pressures indicating the defect was not a complete cleft.
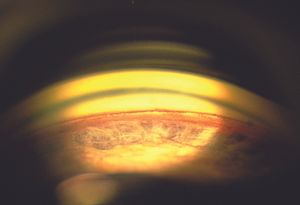
9-54 Angle recession, affected eye. Note the ciliary body band is not particularly wide and could potentially be considered normal.
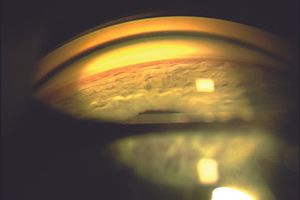
9-55 Fellow eye of patient in 9‑53, showing the normal width of the ciliary body band in this patient, and confirming the diagnosis of angle recession in the eye shown in 9‑53.
Cyclodialysis cleft
A cyclodialysis cleft is the disinsertion of the ciliary body from the scleral spur. This permits free access of aqueous into the suprachoroidal space and usually results in decreased intraocular pressure, sometimes resulting in hypotony. Cyclodialysis clefts can occur as a result of trauma or can be created surgically. Surgical cyclodialysis is rarely performed now because of problems with hemorrhage and hypotony and the failure to provide long-term control.
A cyclodialysis cleft appears as a very deep angle with a cleft through which the white color of the sclera is visible (9‑56 and 9‑58). As in angle recession, there will be rupture of any iris processes in the area of the cleft and the scleral spur may appear to be whiter than in other areas.
A cyclodialysis cleft is a detachment of the ciliary body from the scleral spur (9‑57), while a recessed angle is a tear into the face of the ciliary body. A comparison of the histopathology shown in 9‑52 and 9‑57 illustrates this difference clearly. Angle recession is associated with a normal or elevated intraocular pressure, whereas a cyclodialysis cleft is associated with a normal or decreased intraocular pressure.
Cyclodialysis clefts may close spontaneously or they can be closed surgically. In both cases there may be a dramatic elevation of intraocular pressure as aqueous outflow is suddenly diverted back to the trabecular meshwork.
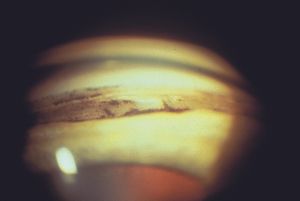
9-56 Gonioscopic view of the angle of a patient following trauma. A cyclodialysis cleft is seen as a white band below the trabecular meshwork. The chamber angle is very deep. Unlike the patient in 9‑53, this patient suffered from hypotony, which resolved following argon-laser treatment to the area around the cleft.
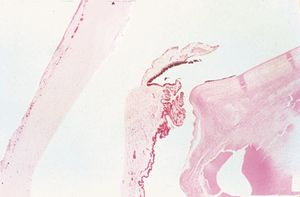
9-57 Histopathologic section of a cyclodialysis cleft showing the dis-insertion of the ciliary body (CB) from the scleral spur (arrow). (Courtesy of Robert Folberg, MD, University of Iowa.)
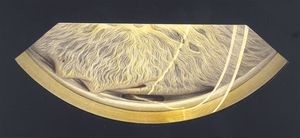
9-58 Aphakic glaucoma status after surgical cyclodialysis showing an open cleft with surrounding synechiae.
Tumors
Tumors of the posterior segment, iris, and ciliary body can elevate intraocular pressure by closing the angle, as discussed in Chapter 8. Open-angle mechanisms by which tumors can elevate pressure include tumor seeding into the angle, a ring melanoma, or shedding of pigment from a large choroidal melanoma (melanomalytic glaucoma) (Yanoff, 1970). Glaucoma occurs more frequently with tumors of the anterior segment, particularly malignant melanoma. Uveal malignant melanomas of the anterior segment lead to the development of glaucoma in over 41% of cases—compared to 14% of posterior melanomas (Yanoff, 1970). Eyes with melanomas of the anterior segment associated with glaucoma have a poorer prognosis than eyes in which glaucoma has not developed (Shields and Klintworth, 1980). Other tumors of the anterior segment can cause glaucoma, but less commonly than melanoma. A gonioscopic examination is critical in evaluating the extent of tumor involvement in eyes with malignancies of the anterior segment. The examination may reveal a direct invasion of the angle (9‑59 and 9‑60) or even peripheral cornea. Even if this is not demonstrated, diffuse invasion of the angle cannot be ruled out (9‑61 to 9‑63).
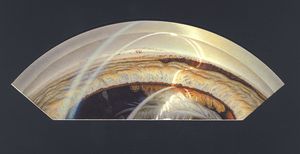
9-59 Gonioscopic view of malignant melanoma of the ciliary body invading through the iris into the angle.
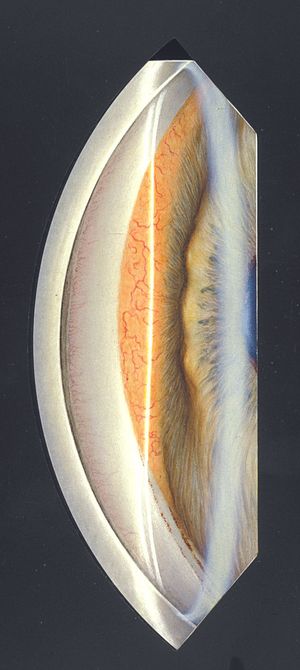
9-60 Large malignant melanoma of the ciliary body extending into the anterior segment and covering the nasal angle.
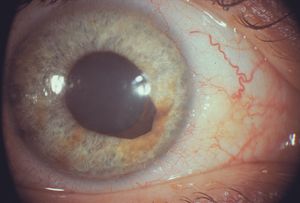
9-61 Slit-lamp photograph of inferonasal mass of the iris with ectropion uvea. This patient suffered from elevated intraocular pressure and very poor vision due to glaucoma.
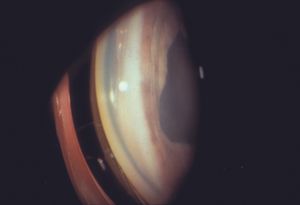
9-62 On gonioscopy the mass seen in 9‑61 does not appear to extend into the chamber angle.
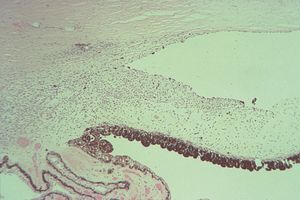
9-63 On histopathology, the mass seen in 9‑61 and 9‑62 is found to be a malignant melanoma affecting the iris root, trabecular meshwork and ciliary body through 360º.
Blood in Schlemm’s canal
The presence of blood in Schlemm’s canal (9‑64 and 9‑65) may be caused by an abnormal flow relationship between the anterior chamber and the episcleral venous channels. The normal episcleral venous pressure is 8 to 10 mm Hg and the normal intraocular pressure is 10 to 21 mm Hg. The higher intraocular pressure normally causes aqueous flow to be directed towards the venous system. If the episcleral venous pressure is higher than the intraocular pressure, blood flows into Schlemm’s canal. This can occur idiopathically, or with intraorbital or intracranial vascular abnormalities, such as carotidcavernous fistulas, dural-sinus fistulas, and Sturge-Weber syndrome. In these conditions conjunctival and episcleral vessels are usually dilated and tortuous (9‑66). These vessels are “arterialized” and remain largely dilated all the way to the limbus.
If the eye is hypotonus from any cause, blood can reflux into Schlemm’s canal. If a Goldmann lens is pressed too tightly against the globe, episcleral venous outflow will be impeded and blood will appear in the canal. Blood can also be seen in Schlemm’s canal in some normal eyes.
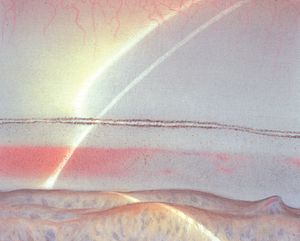
9-64 Gonioscopic view of an angle showing blood in Schlemm’s canal. There is, incidentally, a prominent Schwalbe’s line.
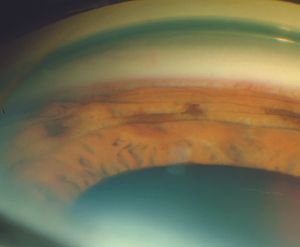
9-65 Blood in Schlemm’s canal in a patient with idiopathic episcleral venous pressure elevation. Note the very faint red hue seen through the pigmented trabecular meshwork, not easily seen without suspecting its presence.
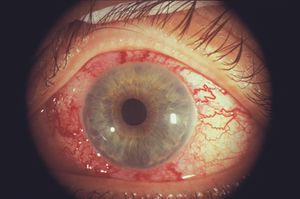
9-66 Engorged, tortuous blood vessels in a patient with elevated episcleral venous pressure due to a low-flow duralsinus fistula. Note the dilated vessels reach the limbus, which is not seen in other conditions in which episcleral vessels may also be dilated.
Postsurgical Changes
Many changes occur in the angle following surgery to the anterior segment. After surgery for glaucoma, gonioscopy is a valuable tool for examining the surgical site. A patent internal sclerostomy should be visible after a trabeculectomy. Gonioscopy can identify blockage of the sclerostomy by blood, vitreous, or lens capsule. In some cases, the iris or ciliary body may become incarcerated (9‑67). A blood clot or bleeding site can be identified. In iridencleisis some iris tissue is intentionally pulled into the edges of the wound (9‑68). This procedure is no longer routinely performed. Setons are usually visible with the slit lamp, but they may be located so far into the angle that gonioscopy is necessary to evaluate the location and patency of the tube (9‑69). Gonioscopy is particularly helpful in identifying the location of the tube following corneal transplantation, as the view of the tube is often obscured by an edematous peripheral cornea on the host side.
After cataract extraction the anterior chamber is generally quite deep because of the loss of lens thickness (9‑70). The cataract wound can be visualized internally (9‑71). Depending on the type and location of a cataract wound, many ocular structures may become incarcerated in the wound, including zonules (9‑71), iris (9‑72), or vitreous (9‑73). One may see evidence of traumatic changes resulting from surgery, such as a scroll of detached Descemet’s membrane (9‑74). In an eye with an anterior chamber intraocular lens, gonioscopy can be helpful in evaluating the placement of the haptics (9‑75 and 9‑76).
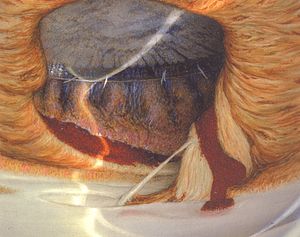
9-67 Angle after trabeculectomy with incarceration of uveal material into the filtering sclerostomy.

9-68 Gonioscopic view of iridencleisis showing an opened fistula into which leaves of iris are tucked.
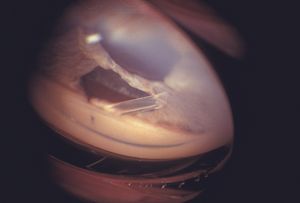
9-69 Gonioscopic view of tube from Baerveldt seton within the anterior chamber.
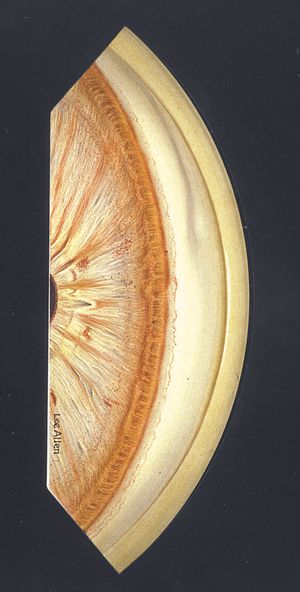
9-70 Nasal angle of aphakic eye showing a deep anterior chamber, which is common after the removal of a lens. There is deep pigmentation in the trabecular meshwork. The cataract wound can be seen in the top half of the cornea.
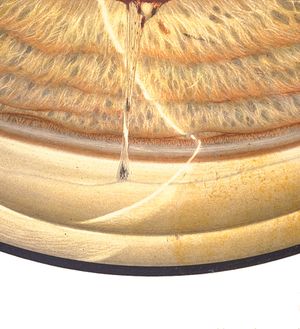
9-71 Internal view of cataract incision in eye of aphakic patient. A small strand of zonular fibers adheres to the wound.
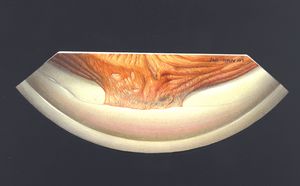
9-72 A large band of iris incarcerated in a cataract wound. The painting is positioned to show the superior angle viewed through an indirect lens. The artist’s name therefore appears upside down.
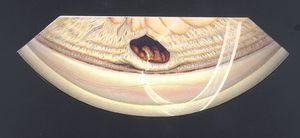
9-73 Gonioscopic view after intracapsular cataract extraction showing a sheet of vitreous that passes through the iridectomy and into the wound.
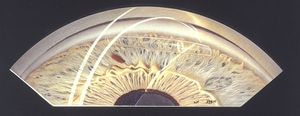
9-74 Inferior scroll of Descemet’s membrane after surgery.

9-75 Haptics of an anterior chamber lens in the anterior chamber angle.

9-76 Haptics of an anterior chamber lens in the chamber angle. Note that the haptic to the left is embedded in the anterior surface of the iris.
Miscellaneous Conditions
Iris-retraction syndrome
The rare iris-retraction syndrome occurs when a rhegmatogenous retinal detachment occurs in an eye with a secluded pupil (Campbell, 1984). Patients with secluded pupils develop iris bombé as aqueous humor trapped in the posterior chamber pushes the iris forward. When there is a rhegmatogenous retinal detachment, there is some flow of aqueous through the break. If patients with secluded pupils and rhegmatogenous retinal detachments are placed on aqueous suppressants, they may make so little aqueous humor that it is all able to flow through the retinal break, causing the iris to be pulled back—often dramatically (9‑77 and 9‑78) If aqueous suppressants are discontinued, the eye may develop iris bombé again (Campbell, 1984).

9-77 Patient with a rhegmatogenous retinal detachment and secluded pupil. When treated with acetazolamide the patient developed a marked back-bowing of the iris, typical of the iris-retraction syndrome. The markedly concave shape of the iris should be noted.
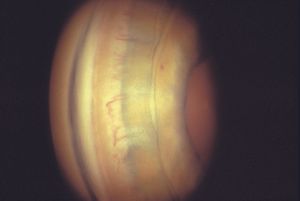
9-78 Iris-retraction syndrome. Eye of the same patient as in 9‑77, seen here with diffuse illumination and higher magnification. Note that the iris is pulled very far posteriorly and iris vessels are exposed and stretched.
Corneal disease
Some corneal diseases can be examined gonioscopically. External tumors can be seen through the cornea (9‑79) and one can evaluate for internal spread. The vasculature of interstitial keratitis can be visualized (9‑80).
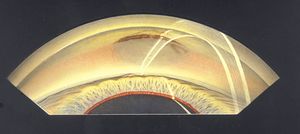
9-79 A patient with melanoma at the limbus visible by gonioscopy. The patient had had a small pigmented spot at the limbus since birth. Over the preceding nine months the spot had enlarged and grown on to her cornea. After gonioscopic examination the tumor was locally excised and found to be a malignant melanoma.
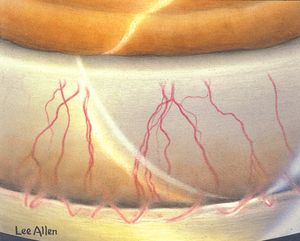
9-80 Gonioscopic view of eye of patient with interstitial keratitis showing blood vessels in the cornea that are visible only to the level of the trabecular meshwork.