Authors: Roger F. Steinert, MD, Ann Z. McColgin, MD, and Sumit Garg, MD
Figures and portions of the text were previously published in Tasman W, Jaeger EA. Duane's Ophthalmology. Philadelphia, PA: Lippincott Williams & Wilkins. 2009.
Introduction
The term keratomileusis comes from the Greek words for "cornea" and "to carve." Laser in situ keratomileusis (LASIK) combines keratomileusis with the accuracy of the excimer laser and is used worldwide for correction of a broad range of refractive abnormalities. The safety and efficacy of the procedure, combined with the quick visual recovery and minimal patient discomfort, have made LASIK the most popular refractive procedure for the treatment of all but the highest levels of myopia and astigmatism, and for high hyperopia. This chapter summarizes the background, preoperative evaluation, technique, clinical outcomes, and potential complications of LASIK.
Background
Jose I. Barraquer, MD, described corneal lamellar surgery for the correction of refractive error in 1949. In the late 1950s and early 1960s Barraquer introduced two landmark contributions in keratorefractive surgery. First, a manually advancing electric microkeratome for creating a corneal cap. Microkeratomes today employ many principles as Barraquer's original design. Second, use of a cryolathe to freeze the corneal cap for precise lenticular reshaping of corneal tissue. The cryolathe was technically difficult to use, as the freezing process and subsequent suturing often resulted in irregular astigmatism and loss of best-corrected vision.
In the mid-1980s, Swinger, Krumeich, and Cassiday developed a technique for removing corneal tissue without the cryolathe. After the microkeratome cut the corneal cap, it was stabilized with suction in the inverted position on a forming die while the refractive cut was made by a second pass of the instrument on the stromal side of the corneal cap. The reshaped free cap was sutured onto the patient's corneal stromal bed. Though avoiding the technical difficulties of the cryolathe, unpredictability and irregular astigmatism remained.
In the late 1980s, Ruiz and Rowsey introduced tissue removal from the stromal bed rather than the free corneal cap, referred to as in situ keratomileusis. The microkeratome was removed the corneal cap in a first pass, then the suction ring was changed to excise a free lenticle of tissue with a second pass. Ring dimensions used in the second pass determined the thickness, and thus the dioptric power, of the lenticle removed. This technique involved less trauma to the corneal cap, but the results remained suboptimal secondary to unpredictable refractive changes and irregular astigmatism.
In the late 1980s, Ruiz developed an automated microkeratome to improve predictability of the microkeratome cut. Automated advancement of the microkeratome head and development of an adjustable suction ring for control of the second pass, represented major advances. The lack of predictable tissue removal in the second pass and the small optical zone of the excised refractive lenticle, however, often left the patient with unacceptable optical aberrations.
In 1990, Pallikarisperformed the first LASIK procedure, using the excimer laser instead of the second microkeratome pass to remove tissue to induce the refractive change. The excimer laser produced better optical results by
- Ablating tissue with submicron accuracy
- Not deforming tissue during the refractive reshaping
- Achieving larger optical zones
The final critical step in achieving consistently excellent results with lamellar refractive surgery was modifying the microkeratome to stop the pass before creating a full free cap. Leaving a narrow hinge of tissue allowed the outer cornea to become a flap that is reflected out of the way during the laser exposure, then returned to its original position and allowed to adhere through natural corneal dehydration to the underlying stromal bed (Figure 1). Repositioning the flap in its original position and avoiding distortion induced by sutures are critical in reducing irregular astigmatism.
Ongoing improvements in microkeratomes continue to make them safer and easier to use. Advances in femtosecond scanning lasers are another important improvement in creating the LASIK flap that allow for greater accuracy and safety.

Figure 1. Schematic representation of laser in situ keratomileusis with formation of a corneal flap followed by stromal ablation. The flap is then replaced into its original position.
Instruments
Many variations of the mechanical microkeratome exist, but the basic principles of the microkeratome cutting head and role of the suction ring are the same. The suction ring's two functions are adhering to the globe, providing a stable platform for the microkeratome cutting head; and raising eye pressure to a high level, firming the cornea so it cannot move away from the cutting blade. Dimensions of the suction ring determine the diameter of the flap and size of the stabilizing hinge. The suction ring is connected to a vacuum pump typically controlled by an on-off foot pedal.
The cutting head has several key components:
- A highly sharpened disposable cutting blade; the blade is discarded after each single eye or bilateral treatment.
- An applanation plate that flattens the cornea in advance of the cutting blade; the length of blade extending beyond the applanation plate helps determine flap thickness.
- An electrical or gas-driven turbine motor, which typically oscillates the blade at 6,000 to 15,000 cycles per minute. The same motor or a second often is used to mechanically advance the cutting head, attached to the suction ring, across the cornea. The surgeon often manually controls the advance of the cutting head.
The hinge initially was located nasally in the Barraquer-designed microkeratomes. Sliding microkeratome has easiest corneal access when approaching from the temporal side, leaving the nasal area last to be cut. Popular microkeratomes were introduced that allow the head to pivot on a post, resulting in an arcing path that leaves the superior zone the last to be cut. A superior hinge was considered a better location because it resisted disturbance of the flap by the wiping motion of the upper eyelid. Nasal hinges had the drawback that many pupils are located nasal of the corneal center. The nasal hinge, even if shifted maximally to the nasal limbus, might impinge on a large treatment zone. There is evidence that a nasal hinge location spares some flap innervation from the nasal long ciliary nerves as they enter the cornea, and there may be less tendency for postoperative dry eye in LASIK patients with nasal hinge flaps compared to superior hinge flaps.
In contrast, one study found more rapid recovery of corneal sensation in patients with superior compared to nasal hinges, although all recovered sensation to the preoperative levels 6 to 12 months after surgery.
An alternative methodology for creating flaps is now widely used in clinical practice. Numerous laser platforms using femtosecond Nd-YAG laser pulse technology can create a lamellar dissection within the stroma. Each laser pulse creates a discrete area of photodisruption of the collagen. Thousands of adjacent pulses are scanned across the cornea in a controlled pattern that results in a flap where the computer is programmed for flap diameter, depth, and hinge location and size. Advocates point to potential for better depth control, avoiding complications like buttonhole perforations and epithelial defects, and precise control of flap dimensions and location.
Tanna et al. retrospectively examined the first 1,000 eyes with femtosecond flaps versus the first 1,000 with microkeratome flaps undergoing wavefront-guided LASIK, and found femtosecond flaps allowed for faster visual recovery and better uncorrected visual acuity.
Patel et al. found corneal backscatter (haze) to be initially higher in femtosecond eyes, but equal to microkeratome eyes at 3 and 6 months. High-contrast visual acuity, contrast sensitivity, and forward light scatter also did not differ between treatments at any examination.
Chan et al. showed that femtosecond and microkeratome eyes had similar visual outcomes at 1 year, but their femtosecond eyes trended to have less induced higher order aberrations (spherical aberration, coma, and trefoil).
Preoperative Evaluation
The preoperative evaluations for patients prior to LASIK and photorefractive keratectomy (PRK) are similar. This chapter is limited to the differences in evaluating the LASIK patient.
As with PRK, careful attention must be given to pre-existing systemic or ocular conditions that may interfere with healing. However, because LASIK violates the corneal surface less than PRK and does not cause the epithelium and superficial keratocytes to heal more slowly, a history of keloid formation is not necessarily a contraindication to LASIK. In addition, a history of quiescent herpes simplex keratitis or autoimmune disease is less of a contraindication to LASIK compared to PRK.
Early reports indicated that postoperative dry eye difficulties were more common with LASIK than PRK. Implementation of femtosecond laser flaps has seen a lower incidence of dry eye, which may be attributed to the creation of thinner flaps and results in a greater residual stromal bed and decreased corneal denervation. Assessment of history, the tear meniscus, Rose Bengal, or lissamine green staining, and Schirmer testing (if indicated) are particularly important. Patients with dry eyes preoperatively should have aggressive artificial tear supplementation to limit keratopathy that can lead to irregularity of the flap postoperatively. These patients can be considered for more aggressive treatments like topical cyclosporine and prophylactic placement of occlusive punctal plugs placed prior to LASIK
When evaluating the cornea before LASIK, look for signs of an anterior basement membrane dystrophy that could predispose to epithelial defects with the microkeratome pass. These patients usually benefit from PRK or creating the LASIK flap by a femtosecond scanning laser.
Corneal topography must be performed to quantitate corneal cylinder and rule out
- Forme fruste keratoconus
- Early stages of frank ectasia, such as the pattern of pellucid marginal degeneration
- Contact lens–induced corneal warpage
Keratometry is important because corneas steeper than 48 D could have thin flaps or frank "buttonholes" (central perforation), and flatter than 40 D could have a smaller diameter flap and are at increased risk for free caps because of hinge transection. When there is increased risk of a thin flap, reusing the microkeratome blade in the second eye typically results in creating a second flap 10 to 20 µ thinner than the first. These issues may be reduced or eliminated by using a femtosecond laser to create a lamellar flap. Compared to microkeratome flaps, femtosecond flaps have an exceptionally low rate of epithelial defects or sloughing, and incomplete, irregular, or buttonhole flaps. Additionally, femtosecond flaps are more uniform than meniscus shaped microkeratome flaps.
One of the most significant advantages of femtosecond laser flaps is the accuracy of flap thickness. A study comparing the IntraLase femtosecond laser to two popular microkeratomes showed that the mean achieved flap thickness was more reproducible with the laser, reducing the comparative risk of overly thick flaps. Sutton and Hodge reviewed accuracy and precision of LASIK flaps with the IntraLase femtosecond laser in 1,000 eyes, and 87.3% were within +/- 20 µm of the intended result. Ninety-eight percent of caps created with the 30-KHz laser were within +/- 20 µm compared to 74.8% in the 15-KHz group. This compares favorably with microkeratome flaps that have shown to be inaccurate.
Measuring corneal thickness using pachymetry is important before LASIK, because to avoid corneal ectasia, an adequate stromal bed must remain after subtracting the thickness of the lamellar flap and ablated tissue. Most practitioners use a minimum residual corneal bed thickness of 250 µ and at least 50% of the original corneal thickness, though this figure is clinically derived and no absolute determinations have been made. Even 250 µ remaining in the stromal bed after ablation does not guarantee that postoperative corneal ectasia will not develop.
Most cases of "unexpected ectasia" have been associated with LASIK flaps thicker than the nominal expected thickness. More surgeons are using intraoperative pachymetry to determine the actual flap thickness. To calculate the likely residual stromal thickness, use the value for the intended total correction, not the nomogram-adjusted refractive error in the computer. True tissue ablation is closer to the value needed to achieve the refractive shift. A nomogram adjustment to a lower refractive error that becomes the laser input does not mean less tissue is removed, but reflects consistent overcorrection in the absence of the adjustment.
Nomogram reduction of the programmed input does not mean less tissue is removed.
After LASIK, excess corneal flattening reduces the quality of the optical performance of the eye and increases aberrations. Postoperative central power below 33 to 35 D should be avoided because of the high amount of spherical aberration of an excessively flat cornea, made worse by a large pupil under mesopic and scotopic conditions. Ideally, patients are measured preoperatively with a wavefront error detection device and, when appropriate, laser ablation is modified to reduce postoperative aberrations.
A recent survey by the American Academy of Cataract and Refractive Surgeons (ASCRS) showed 90% of refractive surgeons use wavefront technology, with 56% using wavefront technology on 76-100% of patients.
Operative Technique
Preparation
Before LASIK, the excimer laser beam is tested for proper homogeneity and fluence in an identical fashion to PRK preparation. The microkeratome and vacuum unit are assembled, inspected, and tested to ensure proper function. The importance of meticulous maintenance of the microkeratome cannot be overemphasized. A new blade is placed, and, in models that permit different heads for different thickness flaps, the desired head size verified.
If using a femtosecond laser, the unit should be warmed up and allowed to run through its self-testing procedure. Flap parameters should be reviewed, including flap diameter, flap depth, and the degree of the side cut angle.
The patient may receive a mild sedative such as oral diazepam 5 to 10 mg approximately 30 minutes prior to the procedure. Topical anesthetic drops are instilled and skin is prepped with povidone iodine or another antiseptic.
Some surgeons drape the skin or lashes with a plastic drape or Steri-Strips, whereas others feel this is unnecessary and could cause material to jam the microkeratome. A lid speculum is placed that can accommodate the suction device and microkeratome path (Figure 2). The cornea may be marked with an optical zone marker for proper centration of the suction ring, and one or more periradial lines are usually placed for proper realignment of the flap (Figure 3).
If using a femtosecond laser, a lid speculum is usually not used until after the flap is created. The femtosecond laser uses a docking platform to couple the laser to the patient's eye. This keeps the eyelids from entering the surgical field during flap creation. Generally, periradial ink lines are unnecessary when a femtosecond is used for flap creation.
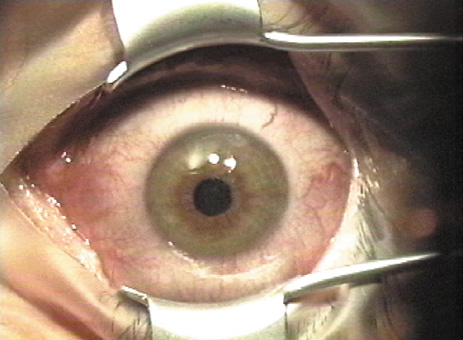
Figure 2. A lid speculum is placed that has a shape to accommodate the path of the microkeratome. Eyelids, lashes, and drape must be kept outside the path of the microkeratome as well.
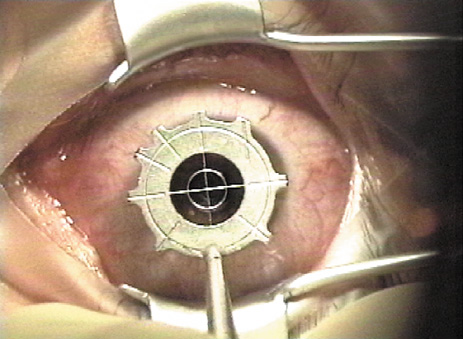
Figure 3. The cornea is marked with periradial lines to assist in realignment of the flap.
Creation of the Flap
Microkeratome suction rings are usually centered over the entrance pupil, but when using one that creates a flap of less than 9.5 mm in diameter, some surgeons prefer skewing the suction ring toward the hinge to ensure that it is not located within the laser treatment zone. The ring is selected to create a diameter larger than the ablation zone. This is particularly important for hyperopic and astigmatic corrections, as well as wavefront-guided treatments, which all typically involve large areas of ablation, sometimes as much as a 10-mm diameter. Once the ring is properly positioned, suction is activated. The intraocular pressure should be raised to over 65 mm Hg and preferably verified with a pneumotonometer (Figure 4) rather than the less precise plastic Barraquer applanator, as low pressure can result in a poor quality, thin, or incomplete flap.
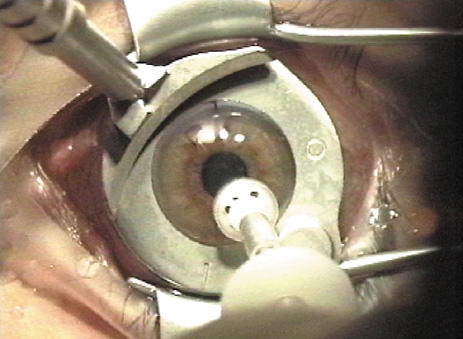
Figure 4. The suction ring is placed, the vacuum activated, and the intraocular pressure is checked (a pneumotonometer is illustrated) to assure the IOP is above 65 mm Hg.
Before making the lamellar cut, the cornea surface is moistened with proparacaine containing glycerin or with nonpreserved artificial tears. Balanced salt solution is avoided because of the possibility of creating mineral deposits within the microkeratome that could interfere with its proper function. The microkeratome is placed on the suction ring with the cornea moistened (Figure 5), and its path is checked to ensure it is free of obstacles such as speculum, drape, or overhanging eyelid.
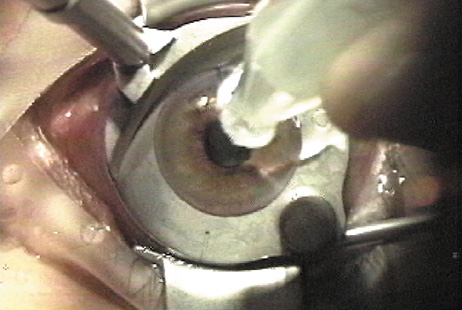
Figure 5. Moisture is applied to lubricate the epithelium prior to the microkeratome pass
The microkeratome is activated, passing over the cornea (Figure 6) until halted by the hinge-creating stopper, after which the head is reversed off the cornea. Epithelial defects may be reduced by lowering the vacuum or discontinuing suction during the reversal. Some models require the vacuum to remain at full pressure during reversal.
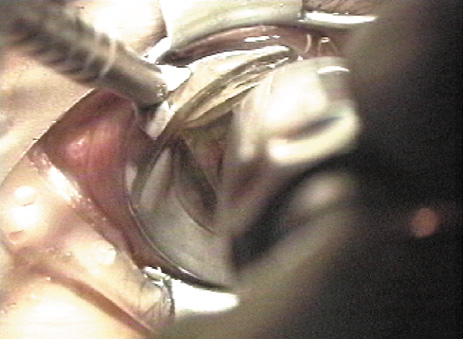
Figure 6. After moistening the corneal surface the microkeratome is placed on the suction ring, the path is checked to assure that it is free of obstacles, and the microkeratome cut is made.
With a femtosecond laser, the docking platform suction ring also is typically centered over the pupil. After docking the cone and applanating the cornea, the surgeon can electronically further shift centration to the desired position.
The IntraLase system allows the surgeon to verify that appropriate applanation pressure has been achieved when a green light appears. If a red light appears, the applanation pressure is too high and the laser will not fire.
Ensure that there is no meniscus peripherally, thereby achieving equal applanation of the cornea and a planar cut. The laser scanning process is then activated, and the patient is verbally encouraged to maintain a steady upward gaze.
Excimer Laser Ablation System
The excimer laser system is focused and centered over the pupil, and the patient is asked to look at the fixation light. The flap is reflected (Figure 7) and the patient is asked to continue fixating.
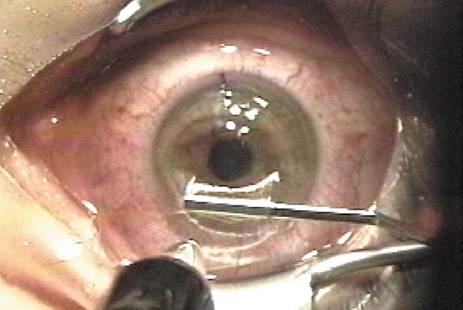
Figure 7. A blunt spatula is used to reflect the flap upon its hinge.
An ultrasonic pachymeter is applied at this point to check bed thickness (Figure 8).
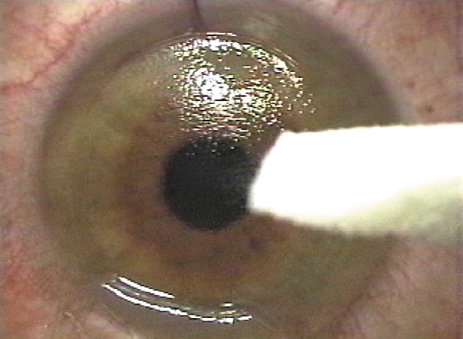
Figure 8. Some surgeons choose to perform pachymetry on the stromal bed to determine actual flap thickness achieved and the amount of tissue available for ablation.
Lights in the room and laser may need adjusting for the patient to continue fixating through the irregular stromal surface after the flap is lifted. If moist, the stromal bed is dried with a microsurgical debris-free sponge (Figure 9).

Figure 9. The stromal bed is dried with a LASIK sponge.
The pupil tracking and iris registration systems, if present, are activated, followed by application of the excimer laser ablation. With or without a pupil tracking device, the surgeon must monitor the patient to ensure fixation is maintained (Figure 10).
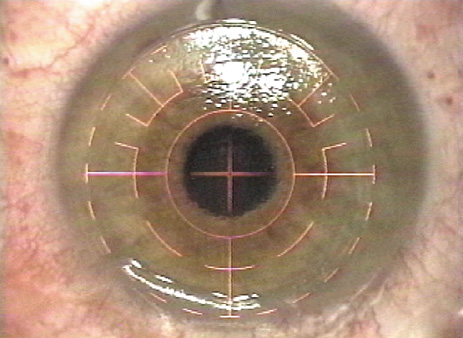
Figure 10. The patient is asked to look at the fixation light while the centration system is centered over the pupil, the tracker (if employed) is activated, and the ablation is performed.
Stromal ablation must be promptly initiated before excessive stromal dehydration occurs. If centration is lost the ablation should be halted and fixation regained prior to finishing treatment. The ablation amount depends on the nomogram each surgeon develops by monitoring outcomes. Major variables that some surgeons find important include: specific laser, individual surgeon, amount of correction, gender, and age.
Flap Replacement
After ablation is completed, the flap is reflected (Figure 11).
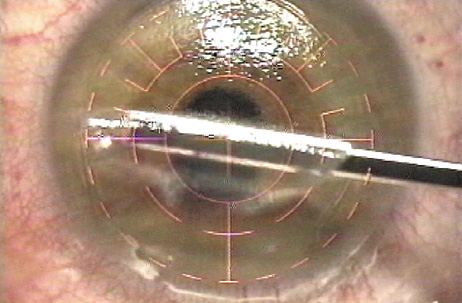
Figure 11. The flap is stroked back into place.
The interface is irrigated (Figure 12) until debris is eliminated, best visualized with oblique rather than coaxial illumination.
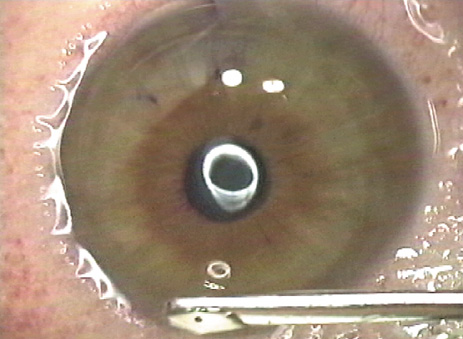
Figure 12. The interface is irrigated under oblique illumination to assure that all debris is removed.
The flap surface is stroked with a smooth instrument (such as the irrigation cannula) from hinge to periphery to ensure that wrinkles are eliminated and that the flap settles into its original position, as indicated by the earlier radial marks and illustrated by applying prednisolone acetate 1% suspension (Figure 13).
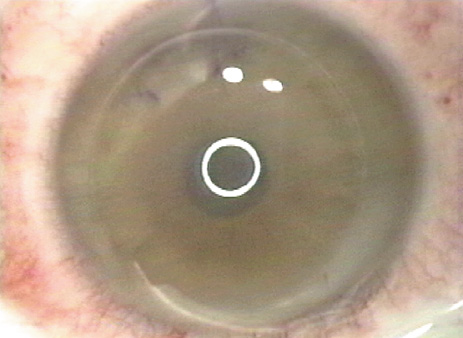
Figure 13. After stroking the flap to remove excess fluid from the interface, the flap is inspected to assure that the alignment marks are in good position and that the "gutter" is uniform. Application of prednisolone acetate 1% suspension, with its white appearance, is very helpful in showing the gutter width.
Within several minutes, the endothelial pump will begin securing the flap in position.
A bandage contact lens should be placed if a significant epithelial defect is present. Once the flap adheres, the lid speculum is removed, taking care not to move the flap.
At the procedure's conclusion, place a drop of antibiotic and steroid over the eye and place a clear shield or protective goggles.
Recheck the flap at the slit-lamp 15 to 30 minutes later to ensure proper alignment.
Instruct patients to use topical antibiotics and steroids postoperatively for approximately 5 days, and advise that the flap surface must be kept well lubricated in the early postoperative period.
Postoperative day 1 day, ensure flap is in proper alignment and there is no evidence of infection or excessive inflammation. If no complications, patients are typically examined 1 week; 1 month; and 3, 6, and 12 months postoperatively.
Outcomes
LASIK studies vary considerably in techniques employed, degree of refractive error treated, length that patients are followed, and variables analyzed. This section summarizes the findings of some larger LASIK studies using modern techniques to treat different degrees of myopia, astigmatism, and hyperopia. This section cites a few historically significant studies and emphasizes the typical range of outcomes encountered in current clinical practice.
Conventional versus Wavefront Treatments
A significant improvement in refractive surgery has been development and implementation of wavefront-guided and optimized treatments. Conventional (traditional) treatments are spherocylindrical only. Wavefront error measurement technology can be employed diagnostically to quantitatively define aberrations affecting vision quality. This technology creates ablation profiles customized per patient to reduce pre-existing aberrations and induction of new aberrations. Wavefront-guided treatments address higher-order aberrations that cause symptoms like loss of contrast sensitivity and nighttime haloes and glare, and also are reported to improve the accuracy of correcting low-order aberrations of spherical error and astigmatism. Aberrations are commonly expressed mathematically as Zernicke polynomials, whose terms can be arranged in a pyramidal fashion (Figure 14).
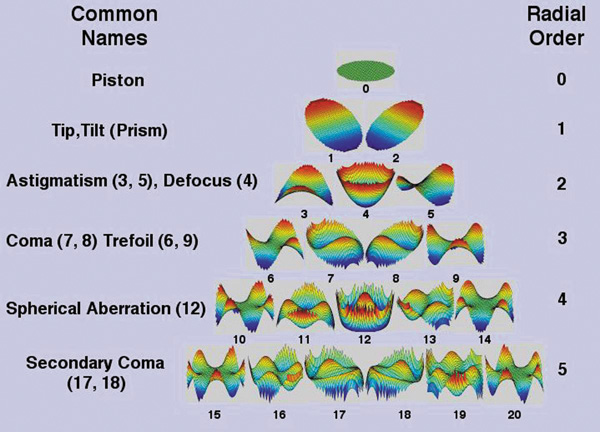
Figure 14. The Zernicke polynomial terms organized as a pyramid.
Two important aberrations are spherical aberration (Figure 15), which often causes a perception of glare and halo, particularly at night, and coma, which causes a smear in a direction like a comet tail (Figure 16).

Figure 15. (A) Spherical aberration shown as a three-dimensional deformation of the wavefront in a sombrero shape. (B) The resultant simulated blur around the letters of a vision chart. Image courtesy Sarver Associates.
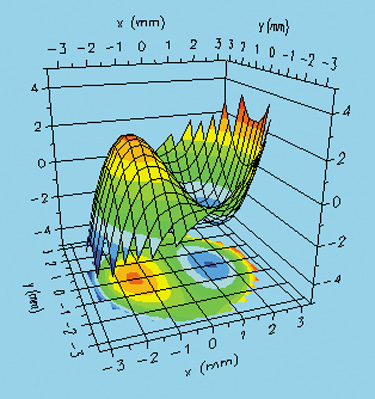
Figure 16. (A) Coma shown as a three-dimensional deformation of the wavefront in a potato chip shape. (B) The resultant simulated directional streak extending from the letters (to the right in this case) of a vision chart. Image courtesy of Sarver Associates.
Wavefront-guided lasers currently approved in the U.S. include the Alcon LADARVision CustomCornea, AMO VISX S4 CustomVue, and the Bausch & Lomb Technolas 217z Zyoptix.
Wavefront-optimized lasers use theoretical wavefront data to determine the best overall tissue ablation plan rather than patient-specific measurements. Wavefront-optimized treatments treat spherocylinder errors while reducing induced spherical aberration.
The Alcon Allegretto Wave Light offers both wavefront-optimized and guided treatments.
Many trials of wavefront analysis-guided and optimized treatments report improvements in visual acuity and quality compared to conventional excimer treatments.Traditional wavefront analysis measures the total wavefront error from the eye. The majority of wavefront aberrations come from the cornea.
Topography-guided treatments are being evaluated. Topography-based treatments are calculated from an ideal reference spherical cornea using videokeratography. Topography-guided treatments evaluate more data points than wavefront-guided, and could benefit patients with highly aberrated corneal topographies and irregular astigmatism.
Low Myopia
Because PRK was judged as adequate for low-to-moderate myopia and did not include creating a corneal flap, LASIK was initially viewed as a procedure for myopia greater than 6 D. As LASIK techniques and microkeratome designs improved, however, many studies reported safety and efficacy of conventional LASIK at a level at least as high as PRK to correct low myopia. Eyes treated with conventional LASIK for low myopia report 45% to 83% achieve uncorrected visual acuity of 20/20 or better, 85% to 100% achieved 20/40 or better, and 73% to 100% obtained a postoperative refraction within 1.0 D of the intended refraction. In most studies no eyes lose two or more lines of best-corrected vision.
Several early studies comparing conventional LASIK to PRK for low myopia show similar safety and predictability, but higher patient satisfaction after LASIK. El-Maghraby and coauthors found that in patients who had conventional LASIK in one eye and PRK in the other to correct -2.5 to -8.0 D, almost twice as many were highly satisfied with their LASIK compared to their PRK eye 1 year after treatment. LASIK eyes had less postoperative pain, more rapid visual recovery, were more likely to achieve uncorrected visual acuity of 20/20 or better, and had more regular postoperative corneal topography. As techniques and technology continue to develop, reported results also have improved. Nevertheless, PRK results are often equal to or superior to LASIK for low myopia.
Alcon Autonomous's premarket approval application to the Food and Drug Administration compared the effectiveness of 139 eyes with wavefront-guided Custom ablations to 47 conventionally treated at 6 months after surgery. All eyes had a preoperative myopic manifest refractive error under -7.0 D sphere and under 0.5 D cylinder. For wavefront-treated eyes, 79.9% achieved uncorrected visual acuity (UCVA) of 20/20 or better, 91.5% 20/25 or better, and 98.6% 20/40 or better. The manifest refraction spherical equivalent (MRSE) was +/- 0.5 D in 74.8% and +/- 1.0 D in 95.7%.
Aberrations were smaller at 6 months than preoperatively for 38% of Custom eyes, compared to 14% of a healed flap edge by RMS increased by 0.08 µm (20%) in the Custom group, and 0.33 µm (82%) in the conventional group. Spherical aberrations increased by 0.04 µm (22%) for Custom eyes and 0.23 µm (108%) for conventional eyes. No Custom patients reported "significantly worse" glare or haloes postoperatively, and only one patient reported significantly worse night driving difficulty. Mean contrast sensitivity for Custom eyes improved by 0.1 to 0.2 log units relative to conventional eyes. Best spectacle-corrected vision (BSCVA) and low-contrast acuity were both slightly better for Custom eyes than conventionally treated eyes. Custom eyes showed more clinically significant increases than decreases, and conventional eyes showed more clinically significant decreases than increases in both measures.
Report results at 1 month of a single site comparison of the Alcon Custom Cornea and Bausch and Lomb Zyoptix systems showed similar excellent results, such as 93% and 90% rates of 20/20 UCVA and 80% and 70% 20/16 UCVA, respectively. A single site comparison of Alcon Custom Cornea and VISX CustomVue for eyes under -8 D and cylinder under 1.5 D yielded 98% and 95%, respectively, within 0.5 D of target. A contralateral eye study showed that, after a nomogram adjustment, the Alcon Custom Cornea and VISX CustomVue results were 80% and 100%, respectively, within 0.5 D of goal.
Studies evaluating wavefront-guided and optimized LASIK continue to show safety and efficacy. Schallhorn and Venter reviewed initial results of 32,569 eyes with low to moderate myopia treated with wavefront-guided LASIK using the VISX STAR S4 excimer laser, and found 94% of eyes within 0.5 D of emmetropia, UCVA of 20/20 in 92% and UCVA of 20/40 in 99%. An American Academy of Ophthalmology (AAO) report concluded that wavefront-guided LASIK is safe and effective in myopia and myopia with astigmatism. Compared to conventional LASIK, wavefront-guided LASIK improved outcomes, specifically contrast sensitivity, night vision, and visual symptoms.
A review comparing the VISX Star S4, LADARVision 4000, and WaveLight Allegretto wavefront versus conventional treatments showed LASIK to be safe and effective for myopia and myopic astigmatism. VISX Star S4 wavefront treatments improved results compared to VISX Star S4 conventional treatments, but LADARVision wavefront treatments did not improve LADARVision conventional results. The Allegretto had the best results in terms of visual acuity in myopic astigmatism eyes, and the VISX Star S4 induced the least higher order aberrations in myopic and myopic astigmatism eyes.
A contralateral comparison study of myopic astigmatism patients showed that wavefront-guided and optimized treatments both had excellent, predictable refractive results. Wavefront-guided LASIK induced less change in 18 of 22 higher-order Zernike aberrations compared to wavefront-optimized, which only change spherical aberration. Wavefront-guided treatments result in better contrast sensitivity compared to wavefront-optimized.
A comparison of wavefront-guided versus optimized LASIK on the Allegretto platform in myopic astigmatism eyes showed no statistically significant difference in visual acuity or refractive outcomes. The study did note noted that Wavefront-guided treatments could be considered for patients with preoperative root-mean-square higher order aberrations greater than 0.35 mm. However, the majority of eyes (83%) in the study population (374 eyes), had RMS higher order aberrations of less than 0.3 mm.
Moderate Myopia
Large published series of conventional LASIK in patients with moderate myopia (approximately -6.0 to -12.0D) report that
- 26% to 57% of eyes achieve uncorrected postoperative visual acuity of 20/20 or better
- 55% to 85% reach at least 20/40
- 41 to 72% are within 1.0 D of intended correction
The percentage of patients losing 2 or more lines of best-corrected vision ranges from 0% to 3.5%.
Alió et al. reported excellent stability and efficacy of LASIK at 10 years; 73% of eyes were within +/- 1.00 D, and 92% within +/- 2.00 D with a mean myopic regression of -0.12 +/- 0.16 D per year. No eyes developed ectasia.
Studies comparing LASIK to PRK for moderate myopia show that the long-term results are remarkably similar. LASIK eyes, however, have faster visual recovery with better uncorrected visual acuity at 1 month. A large multicenter randomized prospective trial showed that more PRK eyes lost best-corrected vision than LASIK-treated eyes, despite average outcomes appearing similar.
Wavefront treatments of moderate myopia have also shown excellent efficacy, predictability, and safety. One study showed that, in moderate myopes, wavefront-guided LASIK significantly improved mean night driving visual performance compared to conventional LASIK.
The VISX Star S4 laser was evaluated in moderate myopes and resulted in a mean postoperative spherical equivalent of – 0.18 +/- 0.43 D at 3 months. 27.9% of eyes were 20/15 or better, 58% were 20/20 uncorrected, 82% were with 0.50 D, and 97.6% were within 1.00 D of emmetropia. No eyes lost more that one line of BSCVA. Similar results were achieved with the VISX Star S4 with postoperative spherical equivalent of -0.33 +/- 0.55 D at 12 months: 64.0% were within +/- 0.50 D and 90% within +/- 1.00 D of intended correction at 12 months.
High Myopia
High myopia is frequently defined as myopia greater than -12.0 D, with several studies reporting results of patients treated with up to -29.0 D. Conventional LASIK's predictability is markedly reduced in this range, with 26% to 50% achieving at least 20/40 uncorrected vision and 32% to 53% a postoperative refraction within 1.0 D of the intended correction. Treating high degrees of myopia also meant there was a higher incidence of best-corrected vision loss than in correction of lower myopia levels. Patients with high myopia, however, often gain best-corrected vision after LASIK, likely due to decreased image minification compared to preoperative spectacles. PRK studies for high myopia generally report unacceptable haze and regression of effect compared to LASIK.
Alió et al. reported results of 196 eyes with a mean preoperative spherical equivalent of -13.95 +/- 2.79 D treated with a conventional profile. At 10 years, 42% were within +/- 1.00 D and 61% within +/- 2.00 D.Additionally, 27.5% underwent retreatments for under correction and/or regression. Eyes that were not retreated had a regression rate of -0.25 +/- 0.18 D per year, 5% lost more that 2 lines of BSCVA, and 1% developed corneal ectasia (> 15 D myopes).
As experience with LASIK increases, however, LASIK and PRK are rarely, if ever, considered appropriate for corrections above -12 D because required ablation depths for high corrections may leave an inadequate stromal bed (less than 250 µ at a minimum) for long-term structural stability of the cornea and it is difficult to predict if high corrections will achieve good high contrast uncorrected visual acuity. High corrections have an unacceptably high level of vision compromises, including glare, halo, and loss of contrast sensitivity. These difficulties are due to the induction of high-order aberrations, particularly spherical, by high corrections that must markedly flatten the central cornea compared to the midperipheral. Many surgeons offer refractive lens exchange or phakic IOLs for patients with extreme myopia.
Myopia with Astigmatism
The results of conventional toric ablations have not been as predictable as results of spherical LASIK ablations. The procedure often undercorrects the cylinder, which could indicate the need for improved nomograms or an inaccuracy of the axis ablated. Scanning spot lasers have improved results, possibly because these instruments achieve the desired complex ablation pattern without excess tissue removal.
Fewer studies are available on the efficacy of conventional toric LASIK.
Most published studies have a limited number of subjects with a wide range of refractive errors, and results of patients having either toric or spherical ablations can be combined, making the results difficult to interpret. Zaldivar et al. reported a series of 84 eyes with 3 to 6 months follow-up after having conventional toric ablations for spherical equivalent refractions ranging from -5.5 to -11.5 D with less than 4.0 D of cylinder. 22% of eyes achieved at least 20/20 uncorrected vision, 77% at least 20/40, and 83% were within 1.0 D of intended correction.
El Danasoury et al. found that 12 months postoperatively, 55% of 56 patients attained 20/20 vision and 91% were within 1.0 diopter of intended correction after conventional toric ablations for spherical equivalents from -2.25 to -5.0 D with 0.5 to 3.0 D of astigmatism.
Fraenkel et al. reported 43 patients with a preoperative spherical equivalent of -1.5 to -15.0 and astigmatism ranging from 0.75 to 7.0 D. 35% of patients attained uncorrected vision of at least 20/20 postoperatively, 79% attained 20/40, and 91% were within 1.0 D of intended correction after conventional LASIK.
Villarrubia et al. compared wavefront and conventional LASIK in myopes with up to 4 D of astigmatism. 99% of eyes in the wavefront and 92% in the conventional group were within 0.50 D of the intended correction.
Wavefront treatment induced significantly fewer higher-order aberrations and resulted in significantly higher contrast sensitivity than conventional treatment. An AAO report concluded that wavefront-guided LASIK is safe and effective in myopia and myopia with astigmatism.
Hyperopia
PRK or LASIK treatments for myopia flatten the central cornea, whereas for hyperopia, the treatments steepen the central cornea by preferentially ablating the midperiphery. Initial problems with hyperopic treatments included diminished predictability and stability in comparison to myopic treatments and loss of best-corrected visual acuity (BCVA), partially secondary to decentrations with small ablation zones.
With enlargement of the optical and peripheral blend zones, and improved centration with tracking devices, studies of hyperopic LASIK with longer follow-up periods have shown improved outcomes. Predictability and stability were better for corrections of +1.0 to +4.0 D than for treating higher degrees of hyperopia. The increased use of scanning spot lasers over broad-beam ones could also be improving hyperopic corrections, as the former create correct exposure patterns without using masks, parallel blades, or other beam manipulators.
Llovet et al. showed treatment of hyeropia up to +6.25 diopters to be safe and effective using a small spot scanning laser and microkeratome. At one year, the mean cycloplegic spherical equivalent improved from +2.5 +/- 0.8 D to +.01 +/- 0.5 D in eyes with ≤ +3.50 D, and +4.5 +/- 0.6 D to +0.4 +/- 0.6 D in eyes with ≥ +3.60 D. 87.8% of eyes maintained or gained 1 or more lines of BSCVA.
Results with wavefront-optimized excimer laser for hyperopia showed stability, with 93.7% (119/127) of eyes within +/- 1.00 D at ≥ 3 years compared to the 6 month follow-up. Regression of effect was noted in 4.7%, with a trend toward more regression with greater amounts of treatment.
Using a wavefront-guided excimer laser, Desai et al., reported safety, efficacy, and predictability at 5 years for hyperopic LASIK. The mean spherical equivalent reduced from +2.53 +/- 1.25 D to +0.53 +/- 0.81 D in all eyes, with no significant difference between low (< +2.00 D), medium (+2.00 to +4.00 D), and high (> +4.00 D) hyperopia groups. 82.4% of low, 75.0% of medium, and 66.3% of high hyperopes were within +/- 1.00 D of emmetropia at 5 years. As seen with conventional treatments, there was a regression of effect, with +0.18 D, +0.30 D, and +0.55 D respectively between 1–5 years postoperatively.
Hyperopic Astigmatism
Treatment of hyperopic astigmatism has been encouraging. Arbelaez and Knorz reported results of patients treated for spherical hyperopia (60 eyes) and hyperopic astigmatism (50 eyes) with 12 months of follow-up. Ninety-one percent of low spherical hyperopia patients (+1.0 to +3.0 D) and 83% of low toric ablations attained a postoperative refraction within ±1.0 D of the intended correction. Of eyes treated for +3.1 to +5.0 D, 83% of spherical and 58% of toric ablations achieved refraction within 1.0 D of intended correction. Of eyes with more than 5.0 D of hyperopia, 50% of spherical and 17% of toric ablations achieved a postoperative refraction within 1.0 D of intended correction 12 months postoperatively.
Ismail showed a reduction of mean cylindrical refraction from +3.15 +/- 0.70 D to +1.27 +/- 0.78 D at 24 months using a conventional treatment. Partial regression of astigmatic effect was found starting at 4 to 6 months, but stabilizing by 1 year.
Kanellopoulos et al. found 71% of hyperopes > +5.25 D or cylinder > or = +1.25 D to be within +/- 0.50 D of the refractive goal at 12 months when using a wavefront-optimized ablation.
Mixed Astigmatism
Mixed astigmatism is defined as refractive error with cylinder greater than sphere and of opposite sign. LASIK is FDA-approved for mixed astigmatism of up to 6.0 D of sphere and cylinder, with the cylinder greater than the sphere and of opposite sign. The outcomes of LASIK for mixed astigmatism are similar to hyperopia and hyperopic astigmatism.
Retreatment
Although LASIK reduces refractive error and improves uncorrected visual acuity in almost all cases, some patients have residual refractive error requiring retreatment. Degree of refractive error warranting retreatment varies based on patient's lifestyle and expectations. Retreatment rates for conventional LASIK are 5.5% to 36%, depending partially on the degree of myopia treated, the laser and nomograms used, and the patient population's expectations.
One study showed that retreatment rates are higher with higher initial corrections, residual astigmatism, and patients over 40.
Randleman et al. reported a 6.3% retreatment rate when using a wavefront-optimized platform for PRK and LASIK. Of the 855 eyes studied, 8.2% were hyperopic and 91.8% myopic. Age, sex, corneal characteristics, and environmental factors did not affect retreatment rate, and there was no significant difference in retreatment rate between PRK and LASIK. A higher retreatment rate was found in hyperopic eyes and eyes with greater than 1 diopter of astigmatism.
One advantage of LASIK over PRK is refractive stability generally occurs earlier and allows earlier enhancements, typically within the first half-year after LASIK. With PRK, the ongoing activation of keratocytes and risk of haze after enhancement usually requires at least 6 months before an enhancement PRK can be safely performed. Retreatment rates are typically higher in hyperopia and higher myopia.
Retreatment after LASIK is usually performed by lifting the pre-existing lamellar flap and applying additional ablation to the stromal bed. The flap can often be lifted several years after the original procedure. If a strong Bowman's layer scar has formed, a new flap can be created with the microkeratome or femtosecond laser.
If a recut flap intersects the original flap cut, a loose piece of corneal tissue will occur that is difficult to replace properly. There is a high risk of severe optical disturbance that will require rigid contact lens use and potentially corneal transplantation.
When the rare recut is performed, depth control guided by OCT imaging is critical. It is safer to handle a strongly healed flap edge using a femtosecond laser to create a new side cut. The new side cut is made smaller than the original flap diameter and entirely inside the original side cut. The new side cut gives access to the original stromal bed, which usually can be separated for an indefinite period of time postoperatively.
Minimize epithelial disruption when lifting a pre-existing flap. Jeweler's forceps or 27-gauge needle can be used to localize the edge of the previous flap. The flap edge can be more easily seen at the slit lamp than with the laser's operating microscope. A flap lift is most easily begun at the slit lamp and completed at the excimer laser. A circumferential epithelial dissection is then performed so the flap can be lifted without tearing the epithelial edges (Figure 17A).
Alternatively, a healed flap edge can be found by placing pressure on the midperiphery, where a slight discontinuity in the corneal tension causes an arc-shaped light reflection at the flap edge, allowing it to be lifted cleanly with the diffuse illumination of the operating laser microscope (Figure 17B). Once ablation is performed the flap is repositioned and the interface is irrigated as in initial LASIK procedures. Confirm no loose epithelium is trapped beneath the flap edge that would cause epithelial ingrowth.
A 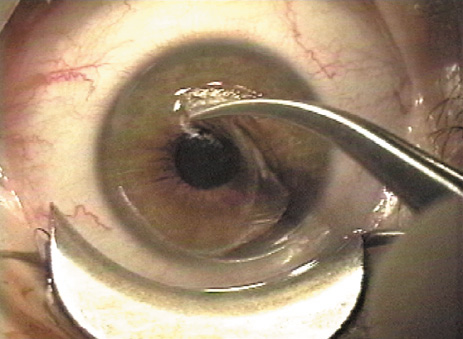
B 
Figure 17. (A) To lift a flap for an enhancement, some surgeons use an instrument such as a jeweler's forceps at the slit-lamp. (B) Other surgeons visualize the flap edge under the laser microscope by depressing the cornea and observing for an arc shape light reflection a the flap edge.
In retreatments performed with flap lifting, complications of laser ablation apply, and there is, increased risk of epithelial ingrowth into the interface and risk of developing flap striae. PRK is considered to occasionally enhance a previous primary LASIK treatment. It was thought that PRK on a LASIK flap carried an increased risk of haze formation and irregular astigmatism., but this concern has been questioned. PRK is an appealing alternative when the residual stromal bed is insufficient for further ablation, or in situations such as a buttonhole flap. The concomitant use of anti-scarring agents like mitomycin-C to improve the results of PRK performed on LASIK flaps has been investigated and may not be necessary on a routine basis.
A final note of caution: Many refractive surgeons have performed enhancements for apparent myopic regression after LASIK when the true cause was subtle early myopic shifting due to nuclear cataract.
Wavefront-Guided PRK and LASIK Outcomes
Wavefront-guided treatments are FDA-approved only for primary treatments, but are an option for patients with vision disturbances attributable to aberrations.Studies show efficacy, predictability, and safety of wavefront-guided retreatments for residual refractive errors and regression. Alió and Montés-Mico found wavefront-guided enhancements to be superior to conventional ones.
Figure 18A shows the corneal topography of a decentered ablation with loss of BCVA and complaints of glare. The wavefront measurement showed a large amount of coma, consistent with the decentration (Figure 18B). After receiving wavefront-guided enhancement, the patient experienced marked improvement, improved BCVA, resolution of most vision complaints, improvement in the corneal topography (Figure 18C), and reduction in the amount of aberration (Figure 18D).
A 
B 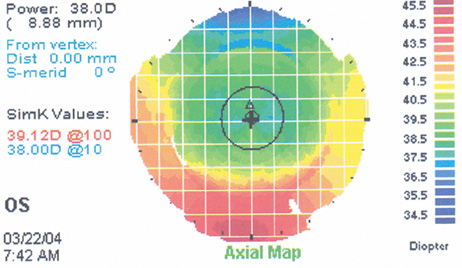
C 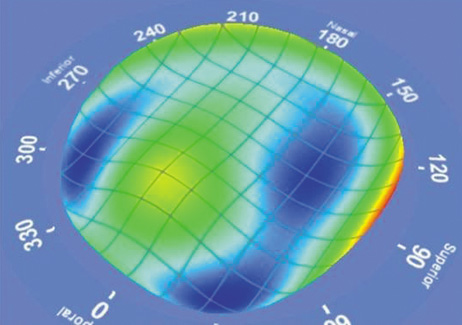
Figure 18. (A) Corneal topography of a superiorly decentered myopic ablation. (B) The three-dimensional wavefront shows a large amount of coma oriented vertically (90 degrees). (C) After a wavefront-guided enhancement centered on the pupil, the corneal topography shows a marked improvement in centration. (D) The postoperative wavefront is not perfectly flat but the coma is markedly reduced.
Complications
In the past, severe complications associated with LASIK were related to microkeratome problems. Microkeratome technology advances have significantly reduced severe, sight-threatening complications. Meticulous care in the cleaning and assembly of the microkeratome is still essential to performing a smooth, uninterrupted keratectomy.
Microkeratome Complications
An overview of microkeratome complications is gained from a synopsis of 47,094 LASIK cases.
Epithelial Defects
The most common microkeratome complication epithelial defects created by the friction of the applanation plate sliding across the surface of the cornea that is stiffened by elevated intraocular pressure. Even in the absence of visible anterior basement membrane disease preoperatively, patients may have inadequate epithelial adhesion structures to resist the frictional stress. A central or inferior defect may result, with either frank sloughing of a sheet of epithelium (Figure 19) or a stretched loosened epithelium ("epithelial slider"). Another epithelial defect is localized abrasion near the hinge where the flap is maximally compressed at the end of the cut. This defect results from the structure of the microkeratome itself.
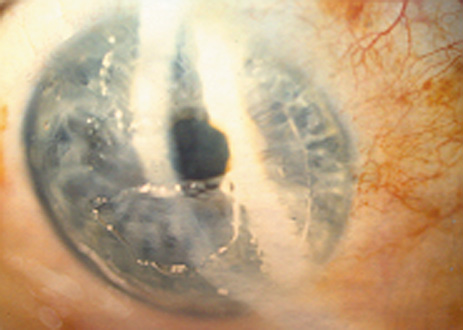
Figure 19. A large epithelial defect is present after retracting the microkeratome in a middle-aged patient with no clinical evidence of anterior basement membrane dystrophy.
Prevention of epithelial defects is preferable to treatment. Key steps are
- Preoperative screening for anterior basement membrane disease
- Avoiding toxic damage to the epithelium from anesthetics and drying
- Frequent administration of lubricants, especially immediately prior to the microkeratome pass (Figure 5)
The best course of action for a defect is usually applying a bandage soft contact lens after completing the laser treatment and repositioning the flap. A potent broad-spectrum antibiotic as infection prophylaxis and intense topical steroids to discourage diffuse lamellar keratitis (DLK) is advisable. The bandage contact lens is retained until the epithelial defect heals.
For patients with a "slider" area of loose but intact epithelium, the size and location of the sliding sheet can help determine whether to debride the loosened sheet or allow it to remain and try to re-establish adhesion. Patients with epithelial defects need to be informed about the condition and the expected slower recovery of vision. More intensive treatment may be needed to re-establish integrity of the epithelium, even with the application of PTK.
Free Cap
Sporadically, the microkeratome transects the hinge, creating a free cap rather than the desired flap. Two principal causes of free caps are the surgeon selecting incorrect settings for the "stop" that controls flap creation and an unusually small or flat cornea. Flat corneal curvature is a risk factor for developing a free cap. Consider adjusting the microkeratome to create a slightly larger hinge or smaller diameter flap when the preoperative keratometry reading is 41 D or less in the hinge meridian or the corneal diameter is less than 11 mm.
When free caps occur, the critical first step is recognizing the absence of the flap and stopping the assistant from taking the microkeratome; the free cap almost always adheres to the microkeratome head, and it must be identified and recovered before it is lost (Figure 20). The cap is placed in a sterile container, preferably closed, to reduce desiccation. The laser procedure is completed if the exposed stromal bed is large enough for laser ablation, and then the cap is repositioned. Using gentian violet ink alignment marks helps identify the epithelial side and correct repositioning of the flap.
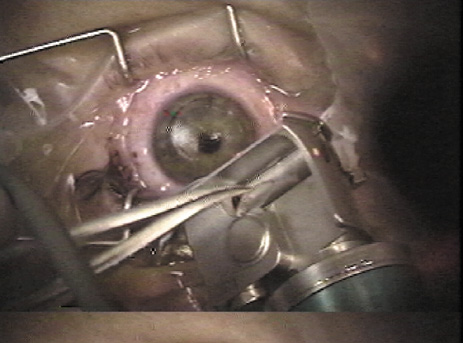
Figure 20. In a free flap, when the hinge is transected, the surgeon must find and retrieve the flap from the microkeratome before the flap is lost.
Some surgeons have success allowing a free cap to adhere naturally, but many prefer placing a suture to perform the function of the hinge. Usually a 10-0 nylon suture is employed as a single interrupted stitch. The suture cannot be tight, as tension distorts the flap and creates irregular astigmatism. If the flap is secure on the first postoperative day, it is usually safe to remove it then. Some surgeons also use a bandage soft contact lens to help stabilize the cap.
Irregular, Thin, and Buttonhole Flaps
Blade defects, poor suction, or uneven microkeratome progression across the cornea can produce an irregular, thin, or buttonholed flap (Figure 21) that could cause irregular astigmatism with loss of best-corrected vision. A steep corneal curvature is a risk factor for developing intraoperative flap complications.
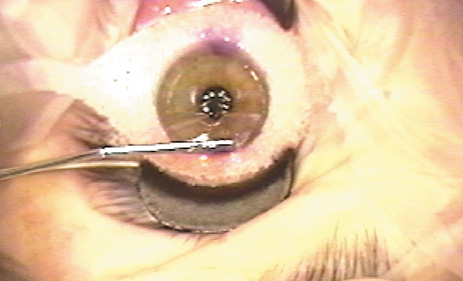
Figure 21. A buttonhole within the LASIK flap occurred due to loss of suction during the microkeratome pass. Note the semicircular central defect seen as the flap is reflected through the middle of the buttonhole.
When thin or buttonholed flaps are recognized, they should be replaced without performing ablation. A bandage soft contact lens is applied for flap stabilization, typically for several days to 1 week. A new flap can be cut after at least 3 months of healing, preferably with a different microkeratome head designed to produce a deeper cut, and ablation can be applied at that time. Application of PRK over the flap to correct the residual optical deficit, with or without topical mitomycin C to discourage scar formation, is another option.
Corneal Perforation
Corneal perforation is a rare but devastating intraoperative complication that can occur if the microkeratome is not properly assembled, or its depth plate is not properly placed in an older model. Surgeons must double-check that the microkeratome has been properly assembled before proceeding. Most modern microkeratomes have a prefixed depth plate so this source of error is eliminated. Corneal perforation also can occur with LASIK on an excessively thin cornea. Corneal thickness must be measured with pachymetry before LASIK, especially in patients being retreated.
Femtosecond Laser Complications
Overall, LASIK flaps created with femtosecond lasers over mechanical microkeratomes are more stable, consistent, and accurate.Binder's study of 1,000 consecutive IntraLase femtosecond LASIK flaps found no decentered or irregular flaps, epithelial defects, or flap perforations.
Haft et al. found that in 4,772 eyes, less than 1% had direct or indirect complications due to IntraLase femtosecond laser flap creation. None of the complications (0.42% deep lamellar keratitis, 0.25% transient light sensitivity syndrome, 0.17% premature breakthrough of gas through the epithelium in the flap margins, 0.06% incomplete to suction loss, and 0.02% irregular flap due to previous corneal scar) led to BSCVA loss.
Though deep lamellar keratitis seems to occur more frequently after LASIK with a femtosecond laser, this is rarely visually significant.
Dry Eye and Corneal Sensation
The occurrence and etiology of dry eye syndromes after laser ablation procedures, particularly LASIK, is discussed in the preceding section on preoperative evaluation. Carefully monitor patients postoperatively for signs of punctate keratitis or more severe manifestations of neurotrophic epitheliopathy. Intensive tear film support with nonpreserved artificial tears, gels and ointments, and adding punctal occlusive plugs or cyclosporine drops if lubricants are inadequate, are the mainstay of therapy while awaiting innervation to return.
Many patients seek laser vision correction due to contact lens intolerance, and dry eyes are a common reason for that intolerance. Dryness persists and briefly worsens while recovering from the flap denervation The patient may notice the long-term persistent dryness has worsened and blame LASIK.Dry eyes commonly cause brief BCVA loss after LASIK.The implementation of femtosecond laser flaps has seen a lower incidence of dry eye, which could be due to creating thinner flaps, resulting in a greater residual stromal bed and decreased corneal denervation.
Striae
Postoperatively, the flap can slip, resulting in prominent wrinkles and a widened peripheral gutter (Figure 22). The earlier the flap malposition is recognized and treated, the easier it is to remove potentially visually significant folds.
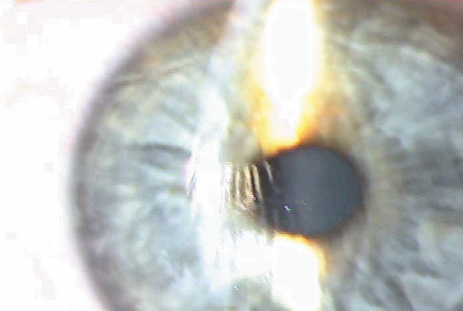
Figure 22. Macrostriae 1 day after uneventful LASIK caused by flap slippage.
Current approaches to smoothing the flap and avoiding striae at the procedure's end vary widely. With every technique used, once the flap is repositioned, carefully examine for the presence of striae. Coaxial and oblique illumination should be used at the operating microscope. Checking the patient at the slit-lamp 15 to 30 minutes postoperatively to detect early flap slippage. Striae should be immediately corrected by refloating and stroking the flap smooth. A protective plastic shield is often used for the first 24 hours to discourage touching of the eyelids and inadvertent flap disruption.
Macrostriae represent full-thickness, undulating stromal folds (Figure 22). These occur because of initial flap malposition or postoperative flap slippage. Examination discloses a wider gutter on the side with the most prominent folds. Flap slippage should be rectified immediately, because the folds rapidly become fixed. A lid speculum is placed under the operating microscope or at the slit-lamp, the flap lifted, copious irrigation used in the interface, and the flap is stroked repeatedly until the striae resolve. Using hypotonic saline or sterile distilled water as the irrigating solution swells the flap and may reduce striae, but swelling reduces flap diameter, which widens the gutter, delays flap adhesion because of prolonged endothelial dehydration time, and could worsen striae after the flap dehydrates.
If macrostriae are present for 24 hours, the folds tend to fix into position through reactive epithelial hyperplasia in the valleys and hypoplasia over the elevations. The central 6 mm of the flap over the macrostriae should be de-epithelialized to remove this impediment to smoothing wrinkles. A bandage soft contact lens should always be used to stabilize the flap and protect the surface until full re-epithelialization occurs. In severe cases of intractable macrostriae, a tight 360-degree antitorque running 10-0 nylon suture or multiple interrupted sutures may be placed for several days, but irregular astigmatism may be present after suture removal.
Microstriae are fine, hairlike optical irregularities best seen on red reflex illumination or light reflected off the iris. Microstriae are fine folds in the Bowman's layer. This anterior location of the microstriae accounts for the BCVA disruption they cause (Figure 23A).
Computer topography color maps usually do not show these irregularities, but disrupting the surface contour is usually heralded by disrupting the regularity of the topographer's placido mires. Applying dilute fluorescein reveals so-called "negative striae," in which the elevated striae disrupt the tear film with loss of fluorescence over the elevated striae (Figure 23B).
A 
B 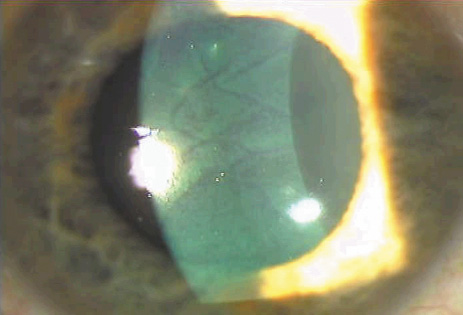
Figure 23. (A) Microstriae become prominent when the corneal surface dries. (B) Microstriae are revealed by "negative staining" when the tear film with fluorescein dye is disrupted over the elevated microstriae.
A few striae may not be visually significant. Mild acuity loss or other optical symptoms, like ghost images, gradually improve as the epithelial thickness adjusts to the folds and restores a more regular anterior tear film. Use nonpreserved artificial tears or a bandage soft contact lens to encourage remodeling of a smooth corneal surface.
Persistent optically significant striae must be addressed. Many interventions have been recommended with variable results. Hydrating the Bowman's layer has been advocated. This occurs with prolonged stroking of the epithelial surface with a moistened surgical spear sponge or irrigating cannula, but microstriae usually disappear within minutes of deliberate de-epithelialization of the area, followed by several drops of sterile distilled water. Hypotonic solution applied directly to the Bowman layer speeds the disappearance of the microstriae.
If striae persist, the flap should be lifted and interface irrigated with balanced salt solution for the flap to reposition. In severe cases, traction with fine tooth forceps or suturing as described for macrostriae could be helpful. Care must be taken not to tear the fragile flap. A bandage soft contact lens is then applied with antibiotic and mild steroid until re-epithelialization is established.
An alternative is phototherapeutic keratectomy (PTK). Pulses from a broad-beam laser, set to a maximal diameter of 6.5 mm, are applied initially to penetrate the epithelium in about 200 pulses. The epithelium acts as a masking agent, exposing the elevated striae before the valleys between them. After the transepithelial ablation, additional pulses are applied with the administration of a thin film of medium viscosity artificial tears every 5 to 10 pulses, with a maximum of 100 additional pulses. These guidelines ensure that little to no haze results, and an average hyperopic shift of under +1 diopter occurs, because of the minimal tissue removal.
Steinert et al. demonstrated significant improvement in BCVA and lack of optically significant haze with PTK, and stable results and minimal complications were found at 2 years follow-up.
Central Islands and Decentration
Irregular astigmatism can be induced by complications related to the laser ablation. Laser fluence and homogeneity must be checked before the procedure to ensure a smooth ablation profile without "hot spots" or "cold spots." Central islands can occur after LASIK and PRK, but the incidence after LASIK is significantly lower. Etiology of central islands after LASIK is not fully understood, but evidence points to vortex currents in the broad beam ablation center, central accumulation of fluid or debris during ablation, or patient healing responses.
Central islands can sometimes be avoided by using anti-island software where more pulses are delivered to the ablation zone center. If a central island occurs and does not resolve, an excimer laser can treat it by lifting the flap and applying a central ablation to the elevated island. Treating central islands with topography-guided excimer treatments and using customized ablation algorithms have shown positive results.
Another potential source of irregular astigmatism related to laser application is a decentered ablation. Mild decentrations are often asymptomatic, but larger decentrations can lead to significant optical aberrations difficult to treat. Decentration also leads to an increase in aberrations including tilt, oblique astigmatism, vertical coma, and spherical aberration. Until recently, decentration has been addressed by further ablation with opposite decentration, or through arcuate keratotomies and limbal relaxing incisions.
Wavefront measurements are sometimes more able to precisely direct enhancement retreatments to improve optical disruptions caused by decentrations, which tend to induce the coma aberration. Wavefront- and topography-guided treatments of decentration have shown improvement in visual acuity with decreases in higher order aberrations. In the U.S., wavefront-guided retreatments remain FDA off-label and topography-guided treatments are not yet FDA-approved. See earlier discussion on outcomes and enhancements in the preceding section.
Interface Inflammation
Sterile interface inflammation has been referred to as "Sands of the Sahara,", diffuse lamellar keratitis (DLK), or, diffuse interface keratitis (DIK).This syndrome ranges from asymptomatic interface haze near the flap edge to marked diffuse haze with diminished best-corrected vision. DLK is clinically staged depending on severity:
Stage 1: Barely visible fine dusting of white blood cells in the interface, often just in the periphery.
Stage 2: Cells are more easily seen and often accumulate in the ridges caused by microkeratome blade chatter, giving the characteristic parallel lines resembling windswept desert sand (Figure 24A).
Stage 3: Reached when the density of inflammatory cells begins interfering with visualizing intraocular details (Figure 24B).
Stage 4: If the release of collagenase occurs with haze, softening, or loss of stroma and onset of large wrinkles, DLK is in this stage (Figure 24C).
A 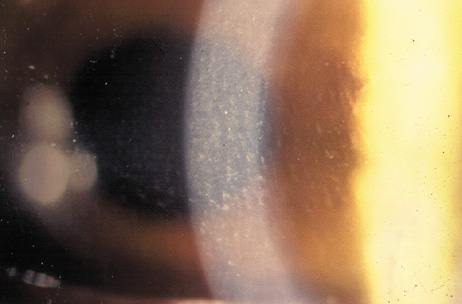
B 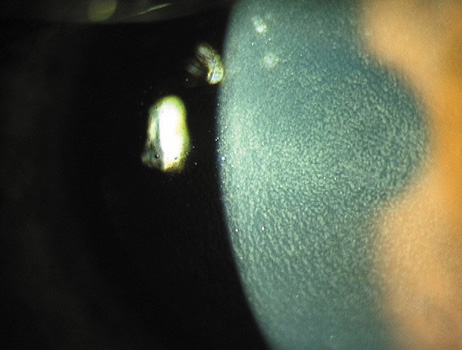
C 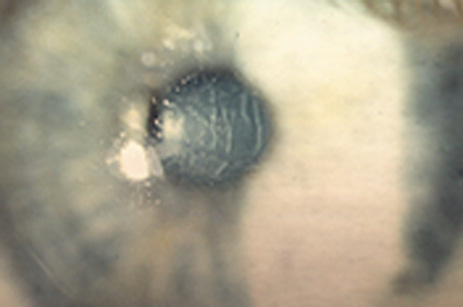
Figure 24. Diffuse lamellar (interface) keratitis. (A) In stage 2, the accumulated white blood cells often cluster along the "chatter lines" created by the microkeratome blade oscillation. (B) As the intensity progresses, in stage 3 the visualization of the anterior chamber is reduced. (C) In the most advanced stage 4, release of collagenase causes softening and even loss of corneal stroma from the flap and underlying bed, accompanied by flap wrinkling.
The inflammation generally resolves on its own without sequelae, but since severe cases can lead to scarring or flap melting, many surgeons treat inflammation with aggressive topical steroids in stages 1 and 2 and irrigation of the interface with balanced salt solution and intense topical steroids in stages 3 and 4. A brief course of systemic steroids may also be employed in severe cases.
Initial attempts to identify a single cause of interface keratitis focused on debris or solution on microkeratome blades, but subsequent cases documented inflammation after flap-lift retreatments without microkeratome use. One case showed interface inflammation after epithelial disruption without lifting the flap at all. Inflammation is likely a nonspecific reaction to corneal trauma, and the interface becomes a potential space where white blood cells can aggregate. A common source of a DLK cluster is a toxic biofilm that builds up in the steam sterilizer reservoir. Though DLK seems to occur more frequently after LASIK with a femtosecond laser, this is rarely visually significant.
Infectious Keratitis
Differentiating sterile interface inflammation from potentially devastating infectious inflammation is important. Interface infection can lead to flap melting, severe irregular astigmatism, and corneal scarring requiring penetrating keratoplasty. If infection is suspected, the flap should be lifted and the interface cultured and irrigated with antibiotics. The most common infections are from gram-positive organisms, then by atypical mycobacteria. Infection from nearly all organisms has been reported, and fungal infection is particularly difficult to treat. If an infection does not respond, the flap may need amputating to improve antibiotic or antifungal penetration. Fourth-generation fluoroquinolones, gatifloxacin, and moxifloxacin are effective against most bacteria causing post-LASIK infections, including mycobacteria.
The ASCRS Cornea Clinical Committee published an algorithm for treating infectious keratitis following LASIK. For any keratitis the flap should be elevated and a culture and scraping performed. For rapid-onset (within 2 weeks) infectious keratitis the committee recommended a fourth-generation fluoroquinolone alternating with fortified cefazolin every 30 minutes. If the patient is hospitalized, fortified vancomycin should be substituted for cefazolin to provide methicillin-resistant staphylococcus aureus coverage.
If the onset is delayed (greater than 2 weeks), atypical bacteria, nocardia, and fungi are often causative. In this case, the committee recommended a fourth-generation fluoroquinolone alternating with fortified amikacin every 30 minutes. Additionally, in all patients, corticosteroids should be stopped and oral doxycycline started to inhibit collagenase productions. Obviously, treatment should be tailored to culture results. Figure 20 shows an infected LASIK flap after a recurrent abrasion.
Epitheal Ingrowth
Epithelial ingrowth into the lamellar interface is first recognized anytime from 2 days to 12 months postoperatively (Figure 25A). Isolated nests of epithelial cells that are not advancing do not need treatment. However, in the following cases, the epithelium should be removed by lifting the flap and scraping the epithelium from the underside and stromal bed prior to flap repositioning:
- The epithelium is advancing toward the visual axis (Figure 25B).
- The epithelium is associated with irregular astigmatism.
- The epithelium triggers overlying flap melting.
A 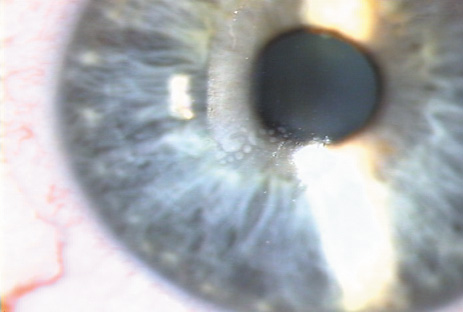
B 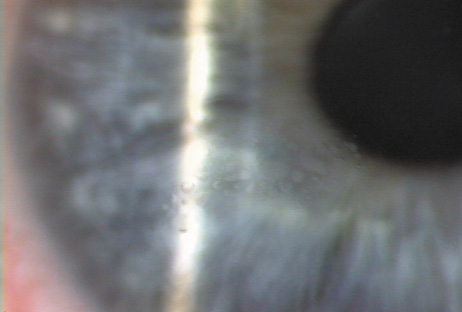
Figure 25. Epithelial ingrowth within the interface. A. Peripheral nests of epithelial cells are often creamy white. B. Occasionally a clear cystic appearance occurs, such as this track of cells extending toward the center.
Two series reported an incidence of epithelial ingrowth requiring intervention after approximately 2% of LASIK procedures. Incidence is greater in patients who develop an epithelial defect during the procedure and those undergoing a retreatment with lifting of a pre-existing flap.
Special care should be taken to ensure that no epithelium becomes caught under the flap edge when it is repositioned. Placing a bandage contact lens at the procedure's conclusion could decrease the incidence of epithelial ingrowth for patients at higher risk. Placing a suture may help seal the peripheral flap and discourage recurrent ingrowth.
PRK could be a good option when epithelial ingrowth is challenging and recurrent. Epithelial ingrowth after femtosecond laser LASIK flaps is an infrequent complication, and happens less frequently than with microkeratome flaps. The side cut anatomy and creation of less peripheral trauma when the flap is created may be responsible for the lower incidence.
Recent software upgrades on the IntraLase femtosecond laser allow a reverse bevel side-cut, which may further lower the incidence of epithelial ingrowth.
Interface Debris
Debris in the interface is occasionally seen postoperatively. The principal indication for intervention—with flap lifting, irrigation, or manual removal of debris—is when foreign material causes an inflammatory reaction. Small amounts of lint, nondescript particles, or tiny metal particles from stainless steel surgical instruments are usually well tolerated. Although a small amount of blood in the interface from transected peripheral vessels may be tolerated, any significant amount usually elicits an inflammatory cell response and should be irrigated. Using a topical vasoconstrictor like epinephrine to facilitate coagulation at the time of flap replacement helps minimize this problem.
Ectasia
The importance of an adequate residual stromal bed to prevent structural instability and postoperative corneal ectasia is discussed in the preoperative evaluation section. Current standards recommend a minimum residual stromal bed of at least 250 µ after completing the ablation.
Keratectasia is usually associated with LASIK for higher myopic corrections, but in thin corneas or patients with multiple laser ablations, cases of ectasia have been reported in corrections as low as -4 D where the residual stromal was believed thicker than 250 µ. Later examinations showed that the microkeratome created a flap thicker than expected, resulting in a thinner residual stromal bed. Anterior segment optical coherence tomography and confocal microscopy are useful in evaluating flap thickness and residual stromal bed in suspected cases of keratectasia.
In other cases, preoperative subtle keratoconus or other ectasias may have been present. Multiple risk factors can lead to keratectasia, but no single or defined combination of risk factors is as an absolute predictor of post-LASIK ectasia. Ectasia can also occur in patients without any recognized risk factors.
Randleman et al. published an Ectasia Risk Score System, which assigns points in a weighted fashion to topographic pattern, predicted residual stromal bed thickness, age, preoperative corneal thickness, and manifest refraction spherical equivalent (Figure 26). In a validation study, 92% of eyes correctly classified as being high risk.
Figure 26. LASIK Ectasia Risk Factor Score Point System A. Risk factors with corresponding point value. B. Cumulative Score with corresponding recommendation for surgery.
Often, good vision can be restored with a rigid gas permeable contact lens. Implanted intrastromal polymethylmethacrylate segments have successfully treated coma-like aberrations and astigmatism in LASIK-induced keratectasia. Collagen crosslinking with riboflavin and ultraviolet A light has been used to arrest and partially reverse the progression of LASIK-induced keratectasia. Extreme cases may require penetrating keratoplasty, including femtosecond laser enabled keratoplasty.
Conclusions
For most levels of refractive error, LASIK is the procedure of choice. The limits of the procedure, particularly for higher levels of myopia (greater than -12.0) and hyperopia (greater than +4.0), must be respected and alternative refractive options considered.
Although LASIK has rapidly surpassed PRK in popularity to treat many refractive errors, visual results are similar among the procedures. A study of patients treated for myopia of -1.00 to -9.50 D showed equal refractive outcomes. Another study of treatment of myopia between -6.00 and -15.00 D showed a slightly lower tendency for postoperative optical symptoms in LASIK compared to PRK eyes. Similar trends are found for wavefront treatments. LASIK preference often relates more to comfort level and early recovery of vision than to ultimate visual acuity and function.
The incidence of LASIK complications decreases with surgeon experience, and complications are best managed with avoidance. When recognized and properly treated, most complications do not result in loss of best-corrected vision. As understanding of risk factors for ectasia improves, incidence should decline. Technological advances, including femtosecond laser flaps, wavefront-guided and optimized, and topography-guided treatment profiles have improved outcomes and allowed for successful treatment of iatrogenic aberrations.
Suggested Reading
- Duffey RJ, Leaming D: US Trends in Refractive Surgery: The 2009 ISRS Survey. American Academy of Ophthalmology Refractive Subspeciality Day. San Francisco. October 24, 2009.
- Ruiz LA, Rowsey JJ: In situ myopic keratomileusis. Invest Ophthalmol Vis Sci 29:S39, 1988
- Lee KW, Joo CK: Clinical results of laser in situ keratomileusis with superior and nasal hinges. J Cataract Surg 29:457, 2003.
- 16. Chan A, Ou J, Manche EE: Comparison of the Femtosecond Laser and Mechanical Keratome for Laser In Situ Keratomileusis. Arch Ophthalmol 126:1484, 2008.
- Talamo JH, Meltzer J, Gardner J: Reproducibility of flap thickness with IntraLase FS and Moria LSK-1 and M2 microkeratomes. J Refract Surg 22:556, 2006.
- Nuijts RM, Nabar VA, Hament WJ, Eggink FA: Wavefront-guided versus standard laser in situ keratomileusis to correct low to moderate myopia. J Cataract Refract Surg 28:1907, 2002.
- Maldonado Bas A, Onnis R: Results of laser in situ keratomileusis in different degrees of myopia. Ophthalmology 105:606, 1998.
- Awwad ST, El-Kateb M, Bowman RW, Cavanagh HD, McCulley JP: Wavefront-guided laser in situ keratomileusis with the Alcon CustomCornea and the VISX Custom Vue: Three month results. J Cataract Refract Surg 20:S606, 2004.
- Padmanabhan P, Mrochen M, Basuthkar S, et al. Wavefront-guided versus wavefront-optimized laser in situ keratomileusis: Contralateral comparative study. J Cataract Refract Surg 34:389, 2008.
- Steinert RF, Hersh PS, the Summit Technology PRK-LASIK Study Group: Spherical and aspherical photorefractive keratectomy and laser in-situ keratomileusis for moderate to high myopia: Two prospective, randomized clinical trials. Trans Am Ophthalmol Soc XCVI:197, 1998.
- Kulkamthorn T, Silao JNI, Torres LF: Wavefront-guided Laser In Situ Keratomileusis in the Treatment of High Myopia by Using the CustomVue Wavefront Platform. Cornea 27:787, 2008.
- Perez-Santonja JJ, Bellot J, Claramonte P, Ismail MM, Alio JL: Laser in situ keratomileusis to correct high myopia. J Cataract Refract Surg 23:372, 1997.
- El Danasoury MA, Waring III GO, El Maghraby A, Mehrez K: Excimer laser in situ keratomileusis to correct compound myopic astigmatism. J Refract Surg 13:511, 1997.
- El Danasoury MA, Waring GO, El Maghraby E, Mehrez K: Excimer laser in situ keratomileusis to correct compound myopic astigmatism. J Refract Surg 13:511, 1997.
- Davidorf JM, Eghbali F, Onclinx T, Maloney RK: Effect of varying the optical zone diameter on the results of hyperopic laser in situ keratomileusis. Ophthalmology 108:1261, 2001
- Kezerian GM,Moore CR, Stonecipher KG: Four-year postoperative results of the US ALLEGRETTO WAVE clinical trial for the treatment of hyperopia. J Refract Surg 24:S431, 2008.
- Durrie DS, Aziz AA: Lift-flap retreatment after laser in situ keratomileusis. J Refract Surg 15:150, 1999.
- Netto MV, Wison SE: Flap lift for LASIK retreatment in eyes with myopia. Ophthalmology 111:1362, 2004.
- Srinivasan S, Drake A, Herzig S: Photorefractive keratectomy with 0.02% mitomycin C for treatment of residual refractive errors after LASIK. J Refract Surg. 24:S64, 2008.
- Kashani S, Rajan M, Gartry D: Wavefront-Guided Retreatment After Primary Wavefront-Guided Laser In Situ Keratomileusis in Myopes and Hyperopes: Long-term Follow-up. Am J Ophthalmol 147:417, 2009.
- Lin RT, Maloney RK: Flap complications associated with lamellar refractive surgery. Am J Ophthalmol 127:129, 1999.
- Binder PS: One Thousand consecutive IntraLase laser in situ keratomileusis flaps. J Cataract Refract Surg 32:962, 2006.
- Tanaka M, Takano Y, Dogru M, et al. Effect of preoperative tear function on early functional visual acuity after laser in situ keratomileusis. J Cataract Refract Surg 30:2311, 2004.
- Oshika T, Klyce SD, Smolek MK, McDonald MB: Corneal hydration and central islands after excimer laser photorefractive keratectomy. J Cataract Refract Surg 24:1575, 1998
- Lim-Bon-Siong R, Williams JM, Steinert RS, Pepose JS: Retreatment of decentered excimer photorefractive keratectomy ablations. Am J Ophthalmol 123:122, 1997.
- Alió J, Galal A, Montalbán R, et al. Corneal wavefront-guided LASIK retreatments for correction of highly aberrated corneas following refractive surgery. J Refract Surg 23:760, 2007.
- Steinert RF, McColgin AZ, White A, Horsburgh GM: Diffuse interface keratitis after LASIK: A non-specific syndrome. Am J Ophthalmol 129:380, 2000.
- Asano-Kato N, Toda I, Hori-Komai Y, Takano Y, Tsubota K: Epithelial ingrowth after laser in situ keratomileusis: Clinical features and possible mechanisms. Am J Ophthalmol 134:801, 2002.
- Rojas MC, Lumba JD, Manche EE: Treatment of epithelial ingrowth after laser in situ keratomileusis with mechanical debridement and flap suturing. Arch Ophthalmol 122:997, 2004.
- Randleman JB, Russell B, Ward MA, Thompson DP, Stulting RD: Risk factors and prognosis for corneal ectasia after LASIK. Ophthalmology 110:267, 2003.
- Ou RJ, Shaw EL, Glasgow BJ: Keratectasia after laser in situ keratomileusis (LASIK): Evaluation of the calculated residual stromal bed thickness. Am J Ophthalmol 134:771, 2002.
- Randleman BJ: Post-laser in-situ keratomileusis ectasia: current understanding and future directions. Curr Opin Ophthalmol 17:406, 2006.
- Randleman JB, Woodward M, Lynn MJ, et al. Risk assessment for ectasia after corneal refractive surgery. Ophthalmology 115:37, 2008.
- Randleman JB, Trattler WB, Stulting RD: Validation of the Ectasia Risk Score System for preoperative laser in situ keratomileusis screening. Am J Ophthalmol 145:813, 2008.
- Pinero DP, Alio JL, Uceda-Montanes A, et al. Intracorneal Ring Segment Implantation in Corneas with Post-Laser In Situ Keratomileusis Keratectasia. Ophthalmology 116:1665, 2009.