Author: Roger F. Steinert, MD
Figures and portions of the text were previously published in Steinert RF, Puliafito CA: The Nd:YAG laser in ophthalmology: principles and clinical applications of photodisruption, Philadelphia, 1985, WB Saunders.
Introduction
The neodymium:yttrium-aluminum-garnet (Nd:YAG) laser is a solid-state laser with a wavelength of 1064 nm that can disrupt ocular tissues by achieving optical breakdown with a short, high-power pulse. Optical breakdown results in ionization, or plasma formation, in the ocular tissue. This plasma formation then causes acoustic and shock waves that disrupt tissue.
The development of the Nd:YAG laser as an ophthalmic instrument and its application in discussion of the posterior capsule coincided with the conversion from intracapsular to extracapsular surgical techniques in cataract surgery. Before the introduction of the Nd:YAG laser, only surgical cutting or polishing of the posterior capsule could manage opacification of the posterior capsule following extracapsular cataract extraction. Nd:YAG laser posterior capsulotomy introduced a technique for closed-eye, effective, and relatively safe opening of the opacified posterior capsule, and laser capsulotomy rapidly became the standard of care.
Capsular Opacification
Postoperative opacification of initially clear posterior capsules occurs frequently in patients after extracapsular extraction of senile cataracts. Time to opacification is highly variable. Time from surgery to visually significant opacification varies from months to years in adults. Almost 100% opacification occurs within 2 years after surgery in younger groups. Rate of opacification declines with increasing age.
The incidence of posterior capsule opacification varies with different studies. Sinskey and Cain reported that 43% of their patients require discission, with an average follow up of 26 months and a range from 3 months–4 years. Another study found opacification in 28% of their patients with 2–3 years of follow-up. Opacification after 3–5 years has been reported to be approximately 50%. Incidence of opacification is lower if a posterior chamber intraocular lens (IOL) is inserted with a convex posterior configuration in close apposition to the posterior capsule. Phacoemulsification is associated with lower rates of opacification than extracapsular cataract extraction.
A study of posterior capsule opacification in 5416 postmortem pseudophakic eyes identified 6 factors associated with reduced posterior capsule opacification:
- Hydrodissection-associated cortical cleanup
- In-the-bag IOL fixation
- Continuous circular capsulorhexis diameter slightly smaller than the IOL optic
- IOL material associated with reduced cellular proliferation: Hydrogel IOLs are associated with the highest rate of posterior capsule opacification. Polymethylmethacrylate (PMMA) is intermediate and silicone and acrylic optic material is the lowest.
- Maximal IOL optic to posterior capsule opacification
- IOL optic geometry with a square, truncated edge
Diabetes mellitus may reduce the rate of posterior capsule opacification compared with nondiabetic patients. Posterior capsule opacification occurs as a result of
- Formation of opaque secondary membranes by active lens epithelial proliferation;
- Transformation of lens epithelial cells into fibroblasts with contractile elements; and
- Collagen deposition. Anterior lens epithelial cells proliferate onto the posterior capsule at the site of apposition of the anterior capsule flaps to the posterior capsule. The contraction caused by the myoblastic features of the lens epithelial cells produces wrinkling of the posterior capsule.
Collagen deposition results in white fibrotic opacities. Mitotic inhibitors instilled into the anterior chamber after extracapsular cataract extraction dramatically reduce capsular opacification. Pharmacologic inhibition of capsular opacification has yet to be successfully introduced into clinical practice.
Posterior capsule opacification results from lens epithelial cells proliferating onto the posterior capsule at the site of apposition of the anterior capsule flaps, explaining the inability of polishing the capsule at surgery to delay the onset or reduce the frequency of late capsular opacification. Polishing the posterior capsule cannot remove the epithelial cells from the anterior capsule flaps. A peripheral ring in the capsular bag may reduce opacification.
Clinical evidence has been presented that a convex posterior chamber IOL can inhibit posterior capsule opacification and close apposition of peripheral anterior and posterior capsule flaps leads to posterior capsule opacification. An unusual form of early central posterior capsule fibrosis occurred when a posteriorly vaulted biconvex optic IOL was positioned with the optic anterior to a capsulorhexis opening smaller than the optic diameter. This positioning, usually with haptic fixation in the ciliary sulcus, allowed the anterior capsule flaps to be apposed to the posterior capsule and the IOL not to be in close apposition to the central posterior capsule. Migration of lens epithelial cells onto the posterior capsule then resulted in early central opacification.
The edge profile of the IOL is considered the dominant factor in the rate of posterior capsule opacification. Truncated edge design has been associated with reduced rates of posterior capsule opacification for both silicone and acrylic IOL optics. Several studies indicate posterior capsule opacification rate is lower when the anterior capsulorrhexis edge overlies the the optic for 360 degrees,but not all studies have shown this. A sharp-edge truncated optic increases the risk of undesireable optical phenomena after surgery.
Optical degradation of initially clear posterior capsules takes several forms clinically.
- Fibrosis connotes a gray-white band or plaquelike opacity that may be recognized in the early postoperative period or may occur later.
- Fibrosis present in the first days to weeks postoperatively likely represents cortical lamellae left at the time of surgery (Figure 1).
- Fibrosis that develops months to years postoperatively is caused by migration of anterior lens epithelium, fibroblastic metaplasia, and collagen production.
- Figure 2 shows a dense fibrinous plaque. Heavy fibrosis occurs frequently at the edge of a posterior chamber IOL placed in the bag with apposition of anterior and posterior capsules (Figure 3).
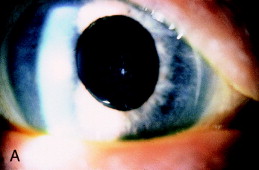
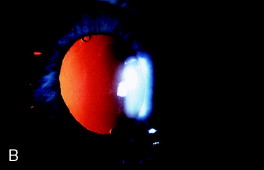
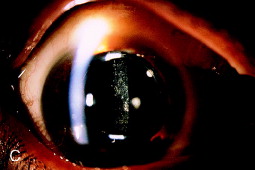 Figure 1. Fine fibrosis of the posterior capsule seen at the second postoperative examination represents cortical lamellae left at the time of surgery. The fibrosis is evident with oblique slit lamp illumination (A) but is optically insignificant when viewed with a red reflex (B). Fine fibrosis (C) may also develop months or years after cataract surgery on an initially clear capsule. This eye is shown 2.5 years after phacoemulsification cataract extraction with implantation of a one-piece polymethylmethacrylate IOL within the capsular bag. From Steinert RF, Puliafito CA: The Nd:YAG laser in ophthalmology: principles and clinical applications of photodisruption, Philadelphia, 1985, WB Saunders, p 74.
Figure 1. Fine fibrosis of the posterior capsule seen at the second postoperative examination represents cortical lamellae left at the time of surgery. The fibrosis is evident with oblique slit lamp illumination (A) but is optically insignificant when viewed with a red reflex (B). Fine fibrosis (C) may also develop months or years after cataract surgery on an initially clear capsule. This eye is shown 2.5 years after phacoemulsification cataract extraction with implantation of a one-piece polymethylmethacrylate IOL within the capsular bag. From Steinert RF, Puliafito CA: The Nd:YAG laser in ophthalmology: principles and clinical applications of photodisruption, Philadelphia, 1985, WB Saunders, p 74.
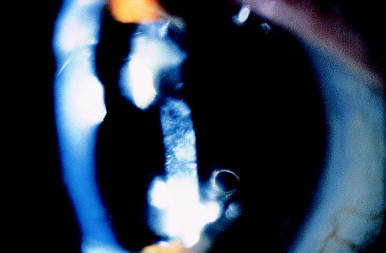
Figure 2. Heavy diffuse fibrosis of a posterior capsule behind a posterior chamber IOL. (From Steinert RF, Puliafito CA: The Nd:YAG laser in ophthalmology: principles and clinical applications of photodisruption, Philadelphia, 1985, WB Saunders, p 74.)
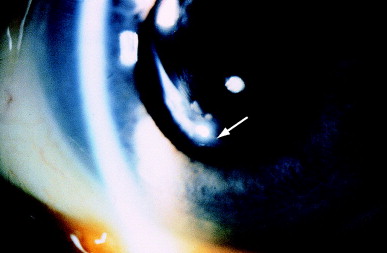
Figure 3. Dense fibrosis at the edge of a posterior chamber IOL optic placed in the bag (arrow) in which an anterior capsular flap is apposed to the posterior capsule. (From Steinert RD, Puliafito CA: The Nd:YAG laser in ophthalmology: principles and clinical applications of photodisruption, Philadelphia, 1985, WB Saunders, p 75.)
The second major form of opacity, formation of small Elschnig pearls and bladder cells, (Figure 4) occurs months to years after surgery. Lens epithelial cells proliferate, which can form layers several cells thick.
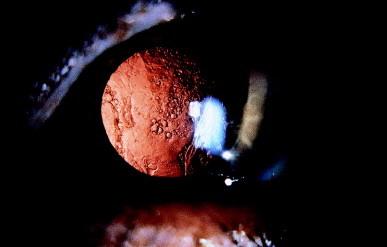
Figure 4. Red reflex view shows formation of multiple small epithelial pearls after anterior epithelial cells migrate centrally from peripheral areas of apposition of anterior capsular flaps to the posterior capsule. (From Steinert RF, Puliafito CA: The Nd:YAG laser in ophthalmology: principles and clinical applications of photodisruption, Philadelphia, 1985, WB Saunders, p 75.)
Capsular wrinkling can have two manifestations:
- Broad undulations of clear capsule are common in the early postoperative period before the capsule tenses. Posterior chamber lens haptics may induce broad wrinkles along the axis of the haptic orientation. A posterior chamber lens may also flatten broad wrinkles if the optic body presses on the capsule. Fibrotic contraction can also induce wrinkles (Figure 5). Broad, undulating wrinkles of clear capsule are rarely visually disturbing. On occasion, a patient may perceive linear distortion or shadows that correspond to the wrinkles and that are relieved by capsulotomy.
- Fine wrinkles or folds in the capsule, caused by myoblastic differentiation on the migrating lens epithelial cells, can result in marked optical disturbance (Figure 6).

Figure 5. Broad wrinkles of the clear posterior capsule (arrow) are seen on red reflex, with numerous small epithelial pearls. (From Steinert RF, Puliafito CA: The Nd:YAG laser in ophthalmology: principles and clinical applications of photodisruption, Philadelphia, 1985, WB Saunders, p 76.)
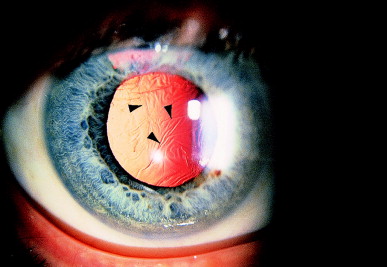
Figure 6. Fine wrinkles in the posterior capsule are evident on red reflex (arrowheads). These wrinkles alone can be visually disturbing and can reduce acuity by several lines or cause Maddox rod light streaks. (From Steinert RF, Puliafito CA: The Nd:YAG laser in ophthalmology: principles and clinical applications of photodisruption, Philadelphia, 1985, WB Saunders, p 76.)
If the iris forms synechiae to the capsule, reactive pigment epithelial hyperplasia and migration onto the capsule may occur. Adhesions occur if large amounts of cortex are left at the time of surgery, which is particularly common with traumatic cataracts. Figure 7 shows dense melanin deposition on a pupillary membrane after an old traumatic cataract. Localized pigmented precipitates on the capsule and IOL can occur spontaneously or after hemorrhage or inflammation.

Figure 7. Pigment from proliferating uveal melanocytes has covered a large portion of this dense pupillary membrane, which formed after a traumatic cataract 40 years previously. The border of the pigment has a sharp scalloped configuration (arrow). (From Steinert RF, Puliafito CA: The Nd:YAG laser in ophthalmology: principles and clinical applications of photodisruption, Philadelphia, 1985, WB Saunders, p 77.)
Posterior Capsulotomy
Indications
Nd:YAG laser capsulotomy is indicated for treatment of opacification of the posterior capsule resulting in decreased visual acuity or visual function, or both, for the patient. Confirmation that posterior capsule opacification is the cause of decreased visual acuity is necessary. Patients may complain of glare despite the appearance of minimal capsular opacification. Glare testing can be helpful in validating these symptoms.
Contraindications
Nd:YAG laser capsulotomy is contraindicated if
- Corneal scars, irregularity, or edema preclude adequate visualization of the target aiming beam or degrade the Nd:YAG laser beam optics, preventing reliable and predictable optical breakdown.
- The patient cannot fixate adequately, with the threat of inadvertent damage to adjacent intraocular structures.
- A glass IOL is involved due to the possibility of a complete fracture in the glass optic. The merits of surgical discission should be carefully weighed.
- Known or suspected active cystoid macular edema (CME), given evidence regarding a beneficial effect of the barrier function of an intact posterior capsule and rare cases of clinical CME that occur after Nd:YAG laser capsulotomy. Avoidance of capsulotomy in an eye with active inflammation is suggested until the visual impairment becomes functionally unacceptable to the patient.
- Though rare, Nd:YAG laser posterior capsulotomy may be complicated by a retinal tear or detachment. Clinical data establishing a correlation between the number or energy level of laser pulses and retinal detachment is lacking.
For eyes at high risk for retinal detachment, the least amount of energy and the lowest possible number of shots should be used to accomplish the capsulotomy, and only a small opening should be made (Table 1). Repolishing the capsule may be considered in high-risk patients.
Table 1. Contraindications to laser capsulotomy.
|
Absolute Contraindications
|
Relative Contraindications
|
- Corneal scars, irregularities, or edema that interfere with target visualization or make optical breakdown unpredictable
- Inadequate stability of the eye
|
- Glass intraocular lens
- Known or suspected cystoid macular edema
- Active intraocular inflammation
- High risk for retinal detachment
|
Technique
Preoperative Assessment
All patients require a complete ophthalmic history and examination before treatment.
The contribution of a capsular opacity to a patient's overall visual deficit may be difficult to judge. Table 2 lists useful techniques. Capsular opacities generally cause little visual difficulty. Capsular opacities may be impressive in oblique slit-lamp illumination but insignificant against the red reflex. Direct ophthalmoscopy is the most reliable technique for assessing capsular opacity. The surgeon's view of retinal details generally correlates with what the patient sees. Retinoscopy and the red reflex also reveal significant optical disturbances. The fundus view with the Hruby lens or 90-diopter lens may allow accurate assessment of capsular clouding. Indirect ophthalmoscope can penetrate significant capsular opacity.
Table 2. Assessment of significance of capsular opacity.
- Direct ophthalmoscopic visualization of fundus structures
- Retinoscopy
- Red reflex evaluation by slit-lamp exam, direct/indirect ophthalmoscopic exam
- Hruby lens view of fundus
- Laser interferometer evaluation
- Potential acuity meter evaluation
- Fluorescein angiography
|
Laser interferometer and the potential acuity meter should penetrate mild to moderate capsular opacity and predict macular function. Both instruments may give false-positive ("good") acuity prediction in the presence of CME,which is the most common cause of postcataract visual impairment besides capsular opacity. False-negative acuity predictions may also occur because of diffuse posterior capsule opacification, poor pupillary dilation, poor patient posture at the slit-lamp examination, communication problems, alphabet illiteracy, nystagmus, tremor, senility, poor patient cooperation, and fatigue.
Adequate visualization may be present for fluorescein angiography or angioscopy. Posterior segment optical coherence tomography is possible in mild levels of posterior capsule opacification but more advanced levels may disrupt the scan to clinically unacceptable levels. To avoid unnecessary capsulotomy, CME should be considered for patients where capsular opacity seems inadequate to explain the quality of vision.
Preparation of the Patient
Explain the purpose and nature of the procedure and obtain informed consent. Remind the patient that
- The procedure is painless.
- Small clicks or pops may be heard, but the patient is to maintain steady fixation.
- The procedure is completed in a matter of minutes.
Brimonidine, apraclonidine, or a beta-blocking agent should be administered in the eye immediately on completion of the Nd:YAG laser posterior capsulotomy to minimize a postoperative intraocular pressure (IOP) spike. If contraindicated, a topical or systemic carbonic anhydrase inhibitor, prostaglandin analogue, or, in a case of an extremely vulnerable optic nerve, oral hyperosmotic agent may be used to prevent or treat any IOP elevation following laser therapy.
Dilation of the pupil facilitates visualization of the capsule over a broad expanse. Dilation is helpful for inexperienced surgeons, except in cases of an iris-clip lens. In the absence of a miotic pupil, dilation may be omitted for an experienced surgeon.
Sketch the landmarks of the pupillary zone of the capsule before dilating the pupil. Pupils are often eccentric or may dilate eccentrically, as shown in Figure 8. Inattention may result in an eccentric capsulotomy, necessitating a second laser session. Before dilation, and a single "marker" shot can be placed in the capsule near the middle of the pupillary axis. When the pupil is dilated, the marker shot is a reminder of the patient's true visual axis.
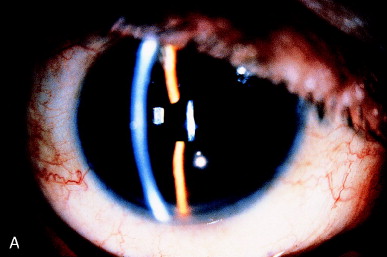
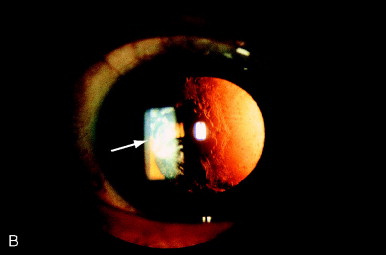
Figure 8. (A) Typical capsular opacity before dilation. (B) Capsulotomy appears eccentric because of uneven pupillary dilation caused by posterior synechia to the capsule (arrow). The capsular opening is properly centered for the undilated pupil. (From Steinert RF, Puliafito CA: The Nd:YAG laser in ophthalmology: principles and clinical applications of photodisruption, Philadelphia, 1985, WB Saunders, p 81.)
A single drop of 2.5% phenylephrine is recommended for routine dilation. Weak dilation prevents iris capture of a posterior chamber IOL (Figure 9), which may be difficult to properly reposition. If inadequate, a drop of 0.5% or 1% tropicamide may be added.
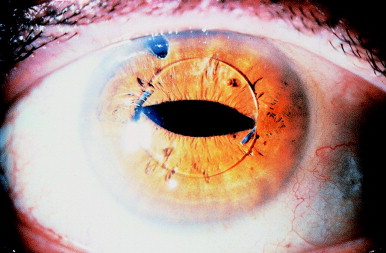
Figure 9. Iris capture of a ciliary sulcus–fixated planar haptic posterior chamber IOL. This phenomenon can occur after wide dilation for posterior capsulotomy. If dilation is necessary at all, weak mydriatics and cycloplegics should be employed. (From Steinert RF, Puliafito CA: The Nd:YAG laser in ophthalmology: principles and clinical applications of photodisruption, Philadelphia, 1985, WB Saunders, p 81.)
Capsulotomy generally does not require anesthesia unless a contact lens is used. If a contact lens is used, a drop of topical anesthetic is applied to the cornea immediately before the procedure. A retrobulbar injection to establish akinesia may be helpful in rare circumstances, such as nystagmus. If a topical anesthetic is applied in advance of the procedure, instruct the patient to keep their eyes closed during the interim to maintain the surface integrity and optical quality of the corneal epithelium.
The patient must be seated comfortably with properly adjusted stool, table, and chin rest heights and a footrest when appropriate. A strap that passes from the headrest behind the patient's head counteracts a patient's tendency to move back during the treatment. Surgeon's visualization of the target is usually improved in a darkened room. If a patient is expected to fixate with the other eye, an illuminated fixation target should be provided. Table 3 summarizes the steps in patient preparation.
Table 3. Preparation of the patient.
|
Before Treatment Session
|
At the Laser
|
- Complete ophthalmic history and examination
- Discussion of proposed procedure, including risks, benefits, and alternatives; signing of informed consent form
- Apraclonidine or beta-adrenergic blocking agent
- Pupillary dilation (optional)
- Determination of visual axis and normal pupillary size: sketch and preliminary laser marker shot
- Weak mydriatic and cycloplegic agents: 2.5% phenylephrine or 0.5% or 1% tropicamide
|
- Review of the procedure, the expected pop or click, and the importance of fixation
- Application of topical anesthetic if contact lens is to be used
- Adjustment of stool, table, chin rest, and footrest for optimal patient comfort
- Application of head strap to maintain forehead position
- Darkening of the room (optional)
- Provision of fixation target for fellow eye
- Illumination of target if room is darkened
|
Procedure
A Peyman or central Abraham contact lens may be used to stabilize the eye, improve the laser beam optics, and facilitate accurate focusing. The Abraham Nd:YAG laser:
- Increases the convergence angle to 24 degrees from 16 degrees.
- Decreases the area of laser at the posterior capsule to 14 µm from 21 µm.
- Increases the beam diameter at both the cornea and the retina.
Use the Abraham Nd:YAG laser lens with care because it is a modified posterior pole lens. If the Nd:YAG laser is not sent through the lens button, but rather the peripheral "carrier" portion of the lens, the Nd:YAG laser may be focused on the retina and cause damage.
The minimal amount of energy necessary to obtain breakdown and rupture the capsule is desired. With most lasers, a typical capsule can be opened by using 1 to 2 mJ/pulse.
The capsule is examined for wrinkles that indicate tension lines. Shots placed across tension lines result in the largest opening per pulse because the tension causes the initial opening to widen.
Figure 10 shows an actual capsulotomy, photographed sequentially and drawn from the photographs, showing the opening as it develops and the location of the next laser shot. Table 4 outlines the basic technique.

Figure 10. Artist drawing based on sequential capsulotomy photographs. The capsulotomy is developed in a cruciate pattern. (A) The first shot is made superiorly in the location of some fine tension lines. (B) The second shot is aimed inside the inferior edge of the initial opening. (C) The next shot again is made at the 6 o'clock position of the capsulotomy border. (D) The fourth shot is made across inferior tension lines to allow the capsulotomy to widen. (E) The opening is nearly 3 mm wide. It is widened by a shot at the 3 o'clock capsulotomy margin. (F) The opening now needs to be directed to the left, with a shot at the 9 o'clock position. (G) The cruciate opening has been accomplished, but a triangular flap extends into the pupillary space from the 7:30 region in the left inferior pupil. A shot is applied to the flap both to cut it and to push it toward the periphery. (H) The capsulotomy is complete, and the pupil will be clear of capsule after the dilation wears off. From Steinert RF, Puliafito CA: The Nd:YAG laser in ophthalmology: principles and clinical applications of photodisruption, Philadelphia, 1985, WB Saunders, pp 84–85.
Table 4. Posterior capsulotomy technique.
- Use minimum energy: 1 mJ if possible.
- Identify and cut across tension lines.
- Perform a cruciate opening: Begin at 12 o'clock in the periphery, progress toward 6 o'clock, and cut across at 3 and 9 o'clock.
- Clean up any residual tags.
- Avoid freely floating fragments.
|
Create a cruciate opening, beginning superiorly near the 12 o'clock position and progressing downward toward the 6 o'clock position.
Unless a wide opening has already developed, shots are then placed at the edge of the capsule opening, progressing laterally toward the 3 and 9 o'clock positions.
If any capsular flaps remain in the pupillary space, the laser is fired specifically at the flaps to cut them and cause them to retract and fall back to the periphery. The goal is to achieve flaps based in the periphery inferiorly. Free-floating fragments should be avoided because they may remain and cause visual interference. Cutting in a circle ("can-opener" style) tends to create large fragments that may not sink from the visual axis or that may settle against the endothelium or angle structures. A large "vitreous floater" of residual capsule may bother the patient.
Beginning the cruciate opening in the superior periphery has several advantages:
- If the patient startles and an adjacent IOL is marked, the mark appears in the periphery.
- Both patient and surgeon are more relaxed before the critical central area is treated.
- As the flaps develop, gravity aids in pulling them toward the inferior periphery. It can be difficult to cause a flap that is hanging down from above to retract.
An IOL may be marked in the course of the capsulotomy. This is particularly true for posterior chamber lenses for which there is little or no separation of the capsule from the IOL. Figure 11 shows a capsulotomy without damage to an overlying posterior chamber IOL.

Figure 11. Posterior capsulotomy performed on a capsule in direct apposition to a lathe-cut posterior chamber IOL. Figure 6 is the pretreatment photograph of the same eye. Note the eccentric location of the optic caused by the displacement of the inferior haptic in the bag and the superior haptic in the ciliary sulcus. The capsulotomy is properly located in the visual axis, but care is taken not to extend the opening beyond the edge of the optic to avoid vitreous herniation around the optic (arrow). (From Steiner RF, Puliafito CA: The Nd:YAG laser in ophthalmology: principles and clinical applications of photodisruption, Philadelphia, 1985, WB Saunders, p 86.)
Visually significant pits and cracks can be minimized and avoided through careful techniques, as outlined in Table 5. The minimal amount of energy must be employed. With a typical capsule and careful focusing, 1 to 2 mJ is usually adequate.
Table 5. Minimizing IOL laser marks.
- Use minimum energy.
- Use contact lens to stabilize the eye, improve laser beam optics, and facilitate accurate focusing.
- If lens making is occurring, make a Christmas-tree opening from 12 o’clock to 4:30 and from 12 o’clock to 7:30 without placing any shots in the central optical zone.
|
- Identify any areas of intraocular lens–capsule separation and begin treatment there.
- Use deep focus techniques: Optical breakdown occurs in the anterior vitreous. The shock wave radiates forward and ruptures the capsule. Higher energy (2 mJ or more) must be used.
|
The capsule should be carefully examined for an area of separation from the IOL in which to begin the capsulotomy. Once the capsulotomy has begun, further areas of separation usually develop.
Beginning the capsulotomy in the 12 o'clock periphery gives an indication of the tendency for IOL marking in a noncritical area.
If there is a tendency for unavoidable repeated marks, the usual cruciate pattern should be modified. Instead of progressing from the 12 o'clock to the 6 o'clock position across the visual axis, the cut should be made nasally and temporally, staying in the periphery of the optical zone. The capsule can be opened "Christmas-tree" fashion, based inferiorly, without any shots in the central visual axis.
To avoid IOL marks, the laser can be intentionally focused posterior to the capsule, causing optical breakdown in the anterior vitreous. The shock wave radiates forward and ruptures the capsule. Optical breakdown just at the capsule and IOL surface, with resultant IOL marking, is avoided. The breakdown threshold is higher in the anterior vitreous than at an optical interface so higher energy, usually a minimum of 2 mJ, is required. Focus consistently at an area posterior to the capsule so the breakdown is not allowed to come up to the back of the IOL, which would result in a larger mark. This technique traumatizes the vitreous, so preference is to reserve the deep focus technique for cases in which IOL marks are occurring with focus directly on the capsule.
In aphakic eyes, deliberate focus anterior to the capsule has been advocated as a mechanism for opening the capsule while leaving the anterior hyaloid intact.
Capsulotomy Size
The capsulotomy should be as large as the pupil in isotopic conditions, such as driving at night, when glare from the exposed capsulotomy edge is most likely. A small opening might be preferred for a patient at high risk of retinal detachment. A small opening in a dense membrane results in excellent optics, analogous to those of a small pupil (Figure 12).
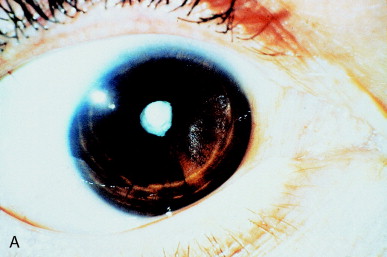
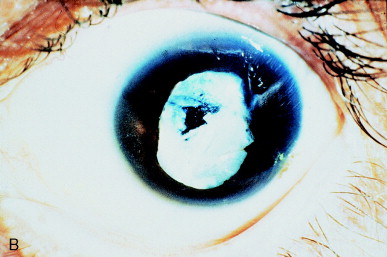
Figure 12. (A) Dense retropupillary membrane after complicated extracapsular cataract extraction. (B) An adequate membrane opening is well centered on the pupillary axis. (From Steinert RF, Puliafito CA: The Nd:YAG laser in ophthalmology: principles and clinical applications of photodisruption, Philadelphia, 1985, WB Saunders, p 92.)
When the capsule is only hazy and transmits images to the retina, a small opening is an improvement but is still suboptimal. The hazy membrane continues to transmit a poor quality image that mixes at the retina with the image transmitted through the clear opening. The patient may experience symptoms of blur, glare, or decreased contrast sensitivity.
Figure 13 shows an example of a posterior capsulotomy performed without dilation. As the patient looks up, down, left, and right, the laser can be applied to capsular edges behind the sphincter so that the capsulotomy can be perfectly centered. Slit-lamp illumination should be with a narrow beam, angled obliquely, to minimize miosis and indicate average pupillary size with ambient dim lighting.
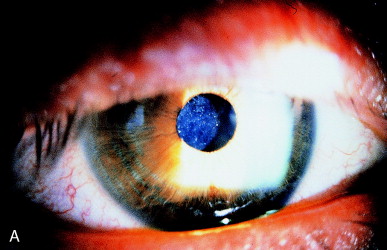
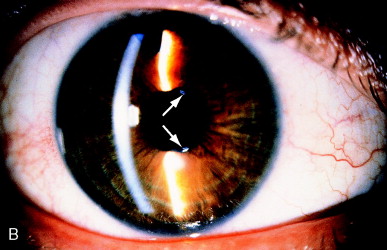
Figure 13. Posterior capsulotomy performed without pupillary dilation. (A) Hazy capsule before treatment. (B) After laser application, the pupillary zone is clear. Two tags of capsule at the edge of the pupil can be seen (arrows). These could be easily exposed to the laser by having the patient look up and down. From Steinert RF, Puliafito CA: The Nd:YAG laser in ophthalmology: principles and clinical applications of photodisruption, Philadelphia, 1985, WB Saunders, p 82.
Capsulotomies may spontaneously enlarge postoperatively. Capsulotomies may increase in mean area by 32% within 6 weeks with capsular enlargement tending toward sphericity with capsular tag retention. Tension created by contractile properties of myofibroblastic lens epithelial cells, IOL haptics, or both, may cause this alteration in capsulotomy contour.
A capsule with residual haze impairs vision and produces glare. A clinical study of glare after extracapsular cataract extraction substantiated the deleterious effect of capsular opacification. Glare and haze remain a problem for 1‑ and 2‑mm capsular openings, decrease with a 3-mm opening, and fully resolve only with a 4‑mm capsular opening.
Postoperative Care
After Nd:YAG laser posterior capsulotomy in all patients, brimonidine, apraclonidine, or a beta-blocker should be administered topically to minimize any IOP increase. For high-risk patients, IOP may be measured again 1 hour following laser treatment. IOP should be remeasured at 4 hours for patients with significant preexisting glaucomatous disc damage or if the IOP is increased 5 mm Hg or more at 1 hour.
Increased IOP may be treated with brimonidine, apraclonidine, topical beta-adrenergic antagonists, prostaglandin analogue, topical pilocarpine, topical or systemic carbonic anhydrase inhibitor, or hyperosmotic agents. Patient's medical history, allergies, and current ocular therapy should be reviewed before determining the appropriate acute antiglaucoma therapy. Continue antiglaucoma therapy for at least 1 week to prevent a delayed pressure elevation. Measure IOP again about 1 week after laser surgery and sooner if indicated by a pressure increase or preexisting glaucomatous optic nerve damage or visual field loss.
Treatment following laser therapy (Table 6) with topical steroids and cycloplegic agents varies based on the individual surgeon's experience. Many patients require no therapy following laser treatment. For patients with iritis, topical steroids can be used four times daily for one or more postoperative weeks. Some patients may require a tapered dosage.
Table 6. Care following capsulotomy.
|
Medication
|
Minimual Suggested Follow-Up Protocol
|
- Apraclonidine immediately following capsulotomy
- Optional additional antiglaucoma therapy (beta-adrenergic antagonist, pilocarpine, carbonic anhydrase inhibitor, hyperosmotic agents) as needed for IOP control
- Optional: cycloplegics (1% cyclopentolate at time of treatment); steroids (1% prednisolone or 0.1% dexamethasone 4 times a day tapered as needed)
|
- 1 hour–4 hours
- Pressure rise to 5 mm Hg: Treatment should be given at 1 day, 1 week, 1 month, 3 months, and 6 months.
|
Results
Nd:YAG laser posterior capsulotomy results in improved visual acuity in 83% to 96% of eyes.
Failure of vision to improve is often due to preexisting ocular disease, including age-related macular degeneration, CME, other macular disease, retinal detachment, corneal edema, glaucoma, ischemic optic neuropathy, and amblyopia.
Complications
Complications causing decreased vision are uncommon but include elevated extraocular pressure, CME, retinal detachment, IOL damage, endophthalmitis, iritis, vitritis, macular holes, and corneal edema.
Intraocular Pressure Elevation
Elevated IOP is the most common, although usually transient, complication following Nd:YAG laser capsulotomy. IOP elevations greater than 10 mmHg have been observed in 15% to 67% of eyes. IOP typically begins to rise immediately after the laser capsulotomy, peaks at 3 to 4 hours, decreases but may remain elevated at 24 hours, and usually returns to baseline at 1 week. Rarely, IOP remains persistently elevated, causing visual field loss requiring glaucoma surgery, or both. Acute IOP increase may cause loss of light perception vision.
Elevated IOP has been associated with preexisting glaucoma, capsulotomy size, lack of a posterior chamber IOL, sulcus fixation of a posterior chamber IOL, laser energy required for the capsulotomy, myopia, and preexisting vitreoretinal disease.
Reliable in-the-bag fixation of posterior chamber IOLs has vastly reduced the incidence of clinically significant elevation of IOP after Nd:YAG laser capsulotomy.
Increased IOP following Nd:YAG laser capsulotomy is associated with a reduced facility for aqueous humor outflow. This reduction has been attributed to capsular debris, acute inflammatory cells, liquid vitreous, and shock-wave damage to the trabecular meshwork. Acute inflammatory cells and capsular debris cause increased IOP by demonstrating pigment granules, erythrocytes, fibrin, lymphocytes, and macrophages within the trabecular meshwork after laser capsulotomy. Glaucomatous eyes may have increased frequency and magnitude of IOP elevation as they already have a reduced outflow facility.
Liquid vitreous as the cause of outflow obstruction is supported by the clinical association between increased IOP following laser treatment and myopia, preexisting vitreoretinal disease, lack of a posterior chamber IOL, and sulcus-fixated posterior chamber IOLs. A capsule-fixated posterior chamber IOL and a smaller capsulotomy may provide a barrier effect, preventing liquid vitreous from reaching the anterior chamber and trabecular meshwork. Liquid vitreous injected into the anterior chamber in owl monkey eyes was found to increase IOP.
Nd:YAG laser–induced shock waves causing increased IOP resulting in damage to the trabecular meshwork has been supported clinically by the association between increased IOP and higher total laser energy used to create the capsulotomy. Photodisruption pulses in the aqueous of the midanterior chamber have not been associated with increased IOP, nor has there been microscopic evidence of damage to the trabecular cords.
Prevention of the IOP increase is appropriate. Apraclonidine, brimonidine, timolol, levobunolol, and pilocarpinedecrease the frequency and magnitude of IOP increases. Apraclonidine is most effective. Apraclonidine, timolol, levobunolol, or other beta-adrenergic antagonists are administered 1 hour before the procedure and again following the procedure. Pilocarpine should be administered only postoperatively due to its miotic effect.
Patients at high risk for IOP elevation or with vulnerable optic nerves should be carefully monitored following the procedure as prophylactic therapy may not prevent late IOP increases.
IOP following Nd:YAG laser capsulotomy may also be elevated by vitreous obstruction of a sclerostomy, the development of neovascular glaucoma, or pupillary block glaucoma.
Patients with existing glaucoma or where high IOP developed acutely after capsulotomy may have long-term elevated IOP.
Cystoid Macular Edema
CME develops in 0.55% to 2.5% of eyes following Nd:YAG laser posterior capsulotomy. CME may occur between 3 weeks and 11 months after the capsulotomy. Incidence of new CME is low following laser capsulotomy, although some patients may acquire CME at a later date. Risk of CME could be lowered by a longer interval between extracapsular cataract extraction and laser capsulotomy, although other studies have not confirmed this. Treatment of CME following Nd:YAG laser posterior capsulotomy is identical to its treatment following cataract extraction and is discussed in Phototherapeutic Keratectomy.
Retinal Detachment
Retinal detachment may complicate Nd:YAG laser posterior capsulotomy in 0.08% to 3.6% of eyes.
Retrospective review of Medicare claims found the cumulative probability of retinal detachment over 36 months following cataract surgery was 1.6% to 1.9% in patients who had laser capsulotomy versus 0.8% to 1% in patients undergoing cataract surgery alone. This review could not distinguish if the same or fellow eye had cataract surgery, capsulotomy, and retinal detachment, nor could it determine the sequence.
Retinal detachment may occur early after the laser capsulotomy or more than a year later. Asymptomatic retinal breaks were found at a rate of 2.1% within 1 month of posterior capsulotomy in one study. Myopia, a history of retinal detachment in the other eye, younger age,and male sex are risk factors following Nd:YAG laser posterior capsulotomy.
In uncomplicated phacoemulsification and PC IOL implantation, a rate of retinal detachment after laser capsulotomy of 0 to 0.4% over 1 to 8 years has been reported in 2 series. In one of these series, no retinal detachments occurred in eyes with axial lengths under 24.0 mm. A study found no increased risk of retinal detachment after Nd-YAG laser capsulotiomy in eyes that did not have a posterior capsule tear at the time of cataract surgery.
Intraocular Lens Damage
Pitting of IOLs occurs in 15% to 33% of eyes during Nd:YAG laser posterior capsulotomy. Pitting is usually not visually significant, although rarely the damage may cause sufficient glare and image degradation that the damaged IOL must be explanted.
The type and extent of lens damage depend on the material used in the IOL. Glass IOLs may be fractured by the Nd:YAG laser. PMMA IOLs sustain cracks and central defects with radiating fractures. Molded PMMA IOLs are more easily damaged than higher-molecular-weight lathe-cut lenses. Damage to silicone lenses is characterized by blistered lesions and localized pits surrounded by multiple tiny pits. The damage threshold is lowest for silicone, intermediate for PMMA, and highest for acrylic materials.
Frequency of damage depends on IOL style. IOLs designed with a ridge separating the posterior capsule from the IOL sustain less damage than lenses with a convex posterior surface and close apposition between the posterior chamber IOL and the posterior capsule.
Endophthalmitis
Propionibacterium acnes endophthalmitis has been reported following Nd:YAG laser posterior capsulotomy. Patients have decreased vision caused by posterior capsular opacification and an otherwise quiet eye. Following laser capsulotomy, the eyes developed significant uveitis and loss of vision. The capsulotomy is presumed to have created opportunity for organisms within the capsule to reach the vitreous and develop into endophthalmitis.
Other Complications
Other complications include
- Iritis persisting for 6 months after laser capsulotomy has been reported in less than 1% of eyes.
- Macular holes have rarely been reported to develop after capsulotomy.
- Specular microscopic studies have reported corneal endothelial cell loss of 2.3% to 7% following Nd:YAG laser posterior capsulotomy.
Suggested Reading
- Aron-Rosa D, Aron JJ, Griesemann M, Thyzel R. Use of the neodymium-YAG laser to open the posterior capsule after lens implant surgery: a preliminary report. J Am Intraocul Implant Soc. 1980;6(4):352-354.
- Baratz KH, Cook BE, Hodge DO. Probability of Nd:YAG laser capsulotomy after cataract surgery in Olmsted County, Minnesota. Am J Ophthalmol. 2001;131(2):161-166.
- Kraff MC, Sanders DR, Lieberman HL. Total cataract extraction through a 3-mm incision: a report of 650 cases. Ophthalmic Surg. 1979;10(2):46-54.
- Frezzotti R, Caporossi A. Pathogenesis of posterior capsular opacification. Part I. Epidemiological and clinico-statistical data. J Cataract Refract Surg. 1990;16(3):347-352.
- Apple DJ, Peng Q, Visessook N, et al. Surgical prevention of posterior capsule opacification. Part 1: Progress in eliminating this complication of cataract surgery. J Cataract Refract Surg. 2000;26(2):180-187.
- Wang MC, Woung LC. Digital retroilluminated photography to analyze posterior capsule opacification in eyes with intraocular lenses. J Cataract Refract Surg. 2000;26(1):56-61.
- Roy FH. After-cataract: clinical and pathologic evaluation. Ann Ophthalmol. 1971;3(12):1364-1366
- Cobo LM, Ohsawa E, Chandler D, Arguello R, George G. Pathogenesis of capsular opacification after extracapsular cataract extraction. An animal model. Ophthalmology. 1984;91(7):857-863.
- Clark DS, Emery JM, Munsell MF. Inhibition of posterior capsule opacification with an immunotoxin specific for lens epithelial cells: 24 month clinical results. J Cataract Refract Surg. 1998;24(12):1614-1620.
- Nishi O, Nishi K. Preventing posterior capsule opacification by creating a discontinuous sharp bend in the capsule. J Cataract Refract Surg. 1999;25(4):521-526.
- Buehl W, Findl O, Menapace R, et al. Long-term effect of optic edge design in an acrylic intraocular lens on posterior capsule opacification. J Cataract Refract Surg. 2005;31(5):954-961.
- Georgopoulos M, Menapace R, Findl O, Petternel V, Kiss B, Rainer G. After-cataract in adults with primary posterior capsulorhexis: comparison of hydrogel and silicone intraocular lenses with round edges after 2 years. J Cataract Refract Surg. 2003;29(5):955-60.
- Hollick EJ, Spalton DJ, Ursell PG, Meacock WR, Barman SA, Boyce JF. Posterior capsular opacification with hydrogel, polymethylmethacrylate, and silicone intraocular lenses: two-year results of a randomized prospective trial. Am J Ophthalmol. 2000;129(5):577-584.
- Mester U, Fabian E, Gerl R, et al. Posterior capsule opacification after implantation of CeeOn Edge 911A, PhacoFlex SI-40NB, and AcrySof MA60BM lenses: one-year results of an intraindividual comparison multicenter study. J Cataract Refract Surg. 2004;30(5):978-985.
- Prosdocimo G, Tassinari G, Sala M, et al. Posterior capsule opacification after phacoemulsification: silicone CeeOn Edge versus acrylate AcrySof intraocular lens. J Cataract Refract Surg. 2003;29(8):1551-1555.
- Sacu S, Menapace R, Findl O, Kiss B, Buehl W, Georgopoulos M. Long-term efficacy of adding a sharp posterior optic edge to a three-piece silicone intraocular lens on capsule opacification: five-year results of a randomized study. Am J Ophthalmol. 2005;139(4):696-703.
- Li N, Chen X, Zhang J, et al. Effect of AcrySof versus silicone or polymethyl methacrylate intraocular lens on posterior capsule opacification. Ophthalmology. 2008;115(5):830-838.
- Davison JA. Positive and negative dysphotopsia in patients with acrylic intraocular lenses. J Cataract Refract Surg. 2000;26(9):1346-1355.
- Keates RH, Steinert RF, Puliafito CA, Maxwell SK. Long-term follow-up of Nd:YAG laser posterior capsulotomy. J Am Intraocul Implant Soc. 1984;10(2):164-168.
- Dickerson DE, Gilmore JE, Gross J. The Abraham lens with the neodymium-YAG laser. J Am Intraocul Implant Soc. 1983;9(4):438-440.
- Stark WJ, Worthen D, Holladay JT, Murray G. Neodymium:YAG lasers: an FDA report. Ophthalmology. 1985;92(2):209.
- Richter CU, Arzeno G, Pappas HR, Steinert RF, Puliafito C, Epstein DL. Intraocular pressure elevation following Nd:YAG laser posterior capsulotomy. Ophthalmology. 1985;92(5):636-640.
- Schubert HD. Vitreoretinal changes associated with rise in intraocular pressure after Nd:YAG capsulotomy. Ophthalmic Surg. 1987;18(1):19-22.
- Vine AK. Ocular hypertension following Nd:YAG laser capsulotomy: a potentially blinding complication. Ophthalmic Surg. 1984;15(4):283-284.
- Schubert HD, Morris WJ, Trokel SL, Balazs EA. The role of the vitreous in the intraocular pressure rise after neodymium-YAG laser capsulotomy. Arch Ophthalmol. 1985;103(10):1538-1542.
- Richter CU, Arzeno G, Pappas HR, Arrigg CA, Wasson P, Steinert RF. Prevention of intraocular pressure elevation following neodymium-YAG laser posterior capsulotomy. Arch Ophthalmol. 1985;103(7):912-915.
- Weinreb RN, Wasserstrom JP, Parker W. Neovascular glaucoma following neodymium-YAG laser posterior capsulotomy. Arch Ophthalmol. 1986;104(5):730-731.
- Winslow RL, Taylor BC. Retinal complications following YAG laser capsulotomy. Ophthalmology. 1985;92(6):785-789.
- Liesegang TJ, Bourne WM, Ilstrup DM. Secondary surgical and neodymium-YAG laser discissions. Am J Ophthalmol. 1985;100(4):510-519.
- Koch DD, Liu JF, Gill EP, Parke DW 2nd. Axial myopia increases the risk of retinal complications after neodymium-YAG laser posterior capsulotomy. Arch Ophthalmol. 1989;107(7):986-990.
- Shah GR, Gills JP, Durham DG, Ausmus WH. Three thousand YAG lasers in posterior capsulotomies: an analysis of complications and comparison to polishing and surgical discission. Ophthalmic Surg. 1986;17(8):473-477.
- Bath PE, Hoffer KJ, Aron-Rosa D, Dang Y. Glare disability secondary to YAG laser intraocular lens damage. J Cataract Refract Surg. 1987;13(3):309-313.
- Keates RH, Sall KN, Kreter JK. Effect of the Nd:YAG laser on polymethylmethacrylate, HEMA copolymer, and silicone intraocular materials. J Cataract Refract Surg. 1987;13(4):401-409.
- Piest KL, Kincaid MC, Tetz MR, Apple DJ, Roberts WA, Price FW Jr. Localized endophthalmitis: a newly described cause of the so-called toxic lens syndrome. J Cataract Refract Surg. 1987;13(5):498-510.